
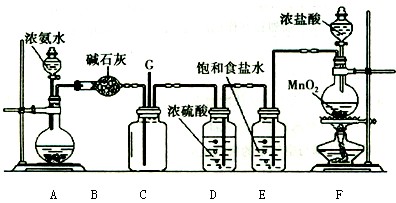
 Mn2++Cl2��+2H2O(2��)
Mn2++Cl2��+2H2O(2��) Mn2++Cl2��+2H2O��
Mn2++Cl2��+2H2O�� NH3��H2O
NH3��H2O NH4����OH��������Ҫ���ɰ�����Ӧ��ʹƽ�����淴Ӧ�����ƶ�����˿�������ʯ�һ�����������ƻ��ʯ�ҵȡ�
NH4����OH��������Ҫ���ɰ�����Ӧ��ʹƽ�����淴Ӧ�����ƶ�����˿�������ʯ�һ�����������ƻ��ʯ�ҵȡ�

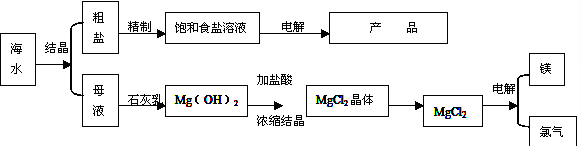
 ������������ϵ�д�
������������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
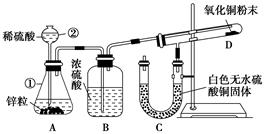
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
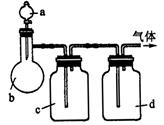
| ���� | a | b | c | d |
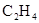 | �Ҵ� | Ũ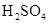 | 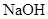 ��Һ ��Һ | Ũ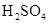 |
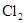 | Ũ���� | 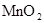 | 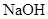 ��Һ ��Һ | Ũ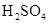 |
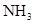 | ����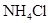 ��Һ ��Һ | ��ʯ�� | 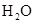 | ����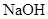 |
| NO | ϡ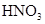 | ͭм | 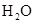 | 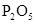 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
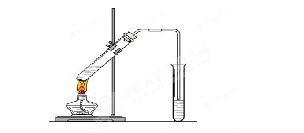
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
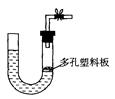
| A������ʯ��ϡ������ȡ������̼ |
| B��п����ϡ���������� |
| C��Ũ����Ͷ���������ȡ���� |
| D����ʯ��ˮ��ȡ��Ȳ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ζ����ȡ��Һ���ʱ���� |
| B����ƿ������ˮϴ��δ���� |
| C����ʽ�ζ���������ˮϴ��δ�ñ�Һ��ϴ |
| D���ζ�����Һ��dz��ɫ��δ�ȵ������������ʼ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��H2��O2��HCl | B��O2��NO2��NO | C��O2��NH3��H2 | D��CO2��O2��NH3 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com