
��ҵ����������Ϊԭ����������������β�����˺���N2��O2�⣬������SO2������SO3���� ����Ϊ�˱���������ͬʱ������Ṥҵ���ۺϾ���Ч�棬Ӧ�����ܽ�β���е�SO2ת��Ϊ���õĸ���Ʒ���밴Ҫ��ش��������⣺
����Ϊ�˱���������ͬʱ������Ṥҵ���ۺϾ���Ч�棬Ӧ�����ܽ�β���е�SO2ת��Ϊ���õĸ���Ʒ���밴Ҫ��ش��������⣺
��1����β��ͨ�백ˮ�У��ܷ��������Ӧ��д�����п��ܷ���������������ԭ��Ӧ�Ļ�ѧ����ʽ�� �� ��
��2����β���백ˮ��Ӧ���õ��ĸ�Ũ����Һ�У���һ���������백ˮ��̼����泥���ʱ��Һ���¶Ȼ����н��ͣ����������塣�ٵ�����Һ�¶Ƚ��͵�ԭ������� ;�������ľ����������ֽ��ҵ��Ҳ��������������ӰҺ����������֪�ýᾧˮ�������Է��� ����Ϊ134�����仯ѧʽΪ ����������������Ҫ����Һ�м��������ĶԱ����ӻ�Ա����������ʣ���Ŀ���� ��
����Ϊ134�����仯ѧʽΪ ����������������Ҫ����Һ�м��������ĶԱ����ӻ�Ա����������ʣ���Ŀ���� ��
��3�������ڲⶨ����β����SO2�������� ��������ĸ��
A.NaOH��Һ����̪��Һ B.KMnO4��Һ��ϡH2SO4
C.��ˮ��������Һ D.��ˮ����̪��Һ
���𰸡�
��1��2H2SO3+O2==2H2SO4 2(NH4)2SO3+O2==2(NH4)2SO4
2NH4HSO 3+O2==2NH4HSO4(��������)
3+O2==2NH4HSO4(��������)
��2���ٷ�Ӧ���� �ڣ�NH4��2SO3��H2O �۷�ֹ������隣�����
��3��BC
��������������Ҫ���黯ѧ�뻷������ѧ�빤ҵ������������Ⱦ�Ĺ�ϵ��(1)��SO2ͨ�뵽��ˮ������SO2+H2O H2SO3��Ȼ��H2SO3�백ˮ���ÿ�����(NH4)2SO3Ҳ������NH4HSO3��������������е�O2�Ӵ�ʱ������Ӧ2H2SO3+O2===2H2SO4��2(NH4)2SO3+O2===2(NH4)SO4, 2NH4HSO3+O2===2NH4HSO4�ȡ�(2)��(1)��������Һ�м���NH3��H2O��NH4HCO3��Һ�¶Ƚ��Ͳ��������壬˵����Ӧ�������������������������������ֽ��ҵ��Ư�����ֿ�����������ӰҺNa2S2O3��˵���þ���Ϊ(NH4)2SO3�Ľᾧ�壬������֪�þ���Ļ�ѧʽΪ(NH4)2SO3��H2O��(NH4)2SO3���л�ԭ���ױ��������ʼ����������ʵ�Ŀ���Ƿ�ֹ(NH4)2SO3��������(3)Ҫ�ⶨSO2�ĺ��������Լ�����SO2��Ӧ������������ָʾ��Ӧ���յ㣬����֪��ΪB��C��
H2SO3��Ȼ��H2SO3�백ˮ���ÿ�����(NH4)2SO3Ҳ������NH4HSO3��������������е�O2�Ӵ�ʱ������Ӧ2H2SO3+O2===2H2SO4��2(NH4)2SO3+O2===2(NH4)SO4, 2NH4HSO3+O2===2NH4HSO4�ȡ�(2)��(1)��������Һ�м���NH3��H2O��NH4HCO3��Һ�¶Ƚ��Ͳ��������壬˵����Ӧ�������������������������������ֽ��ҵ��Ư�����ֿ�����������ӰҺNa2S2O3��˵���þ���Ϊ(NH4)2SO3�Ľᾧ�壬������֪�þ���Ļ�ѧʽΪ(NH4)2SO3��H2O��(NH4)2SO3���л�ԭ���ױ��������ʼ����������ʵ�Ŀ���Ƿ�ֹ(NH4)2SO3��������(3)Ҫ�ⶨSO2�ĺ��������Լ�����SO2��Ӧ������������ָʾ��Ӧ���յ㣬����֪��ΪB��C��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���ʼ��仯�����й㷺Ӧ�á�
��1������ʯ����Ҫ�ɷ�Ca5��PO4��3F���ڸ������Ʊ����ף�P4�����Ȼ�ѧ����ʽΪ��4Ca5��PO4��3F��s��+21SiO2��s��+30C��s��3P4��g��+20CaSiO3��s��+30CO��g��+SiF4��g�� ��H
��������Ӧ�У����������������___________________________________��
����֪��ͬ�����£�
4Ca5��PO4��3F��s��+3SiO2��s��====6Ca3��PO4��2��s��+2CaSiO3��s��+SiF4��g�� ��H1
2Ca3��PO4��2��s��+10C��s��====P4��g��+6CaO��s��+10CO��g�� ��H2
SiO2��s��+CaO��s��====CaSiO3��s�� ��H3
�æ�H1����H2�ͦ�H3��ʾ��H����H��_____________________________��
��2�������������Ϊ����������ӣ�����ṹʽ����ͼ��֮����ȥ����ˮ���ӵIJ����ṹʽΪ________________________�����������ƣ��׳ơ����ơ����dz��õ�ˮ���������仯ѧʽΪ___________________________��

��3���������ƣ�NaH2PO2�������ڻ�ѧ������
��NaH2PO2��PԪ�صĻ��ϼ�Ϊ________________________��
�ڻ�ѧ��������Һ�к���Ni2+��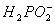 �������Ե������·���������Ӧ��
�������Ե������·���������Ӧ��
(a)__________Ni2++__________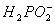 +__________
+__________ __________Ni+________
__________Ni+________ +_______
+_______
��b��6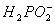 +2H+====2P+4
+2H+====2P+4 +3H2��
+3H2��
���������д������ƽ��Ӧʽ��a����
�����â��з�Ӧ�������϶Ƽ�����������Ͻ𣬴Ӷ��ﵽ��ѧ������Ŀ�ģ�����һ�ֳ����Ļ�ѧ�ơ�������·���Ƚϻ�ѧ�����ơ�
�����ϵIJ�ͬ�㣺_______________��ԭ���ϵ���ͬ�㣺_______________����ѧ�Ƶ��ŵ㣺_______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
X��Y��Z��WΪ������ͬ�������ķ��ӻ����ӣ�����ԭ������С��10��Ԫ����ɣ�X��5��ԭ�Ӻˡ�ͨ��״���£�WΪ��ɫҺ�塣
��֪:X+Y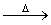 Z+W
Z+W
(1)Y�ĵ���ʽ��_________________________��
(2)Һ̬Z��W�ĵ������ƣ����ɵ������������ͬ���������ӣ�Һ̬Z�ĵ��뷽��ʽ��_________________________________��
(3)��ͼʾװ���Ʊ�NO����֤�仹ԭ�ԡ���������Ҫ������
a.����ƿ��ע��������NaOH��Һ����ʢ��ͭƬ��С�ձ�����ƿ�С�
b.ֹˮ�У���ȼ���ף�����ƿ�У����ý�����
c.�����׳��ȼ�գ�һ��ʱ����Һ©�����������ձ��е�������ϡ���ᡣ
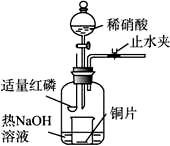
�ٲ���c��ȱ�ٵ�һ����Ҫ������_______________________________________��
�ں��׳��ȼ�յIJ�����NaOH��Һ��Ӧ�����ӷ���ʽ�� ______________________________________��
�۲���c����ϡ������ձ��е�������______________________________________
____________________________________________________________________��
��Ӧ�����ӷ���ʽ��____________________________________________________��
(4)һ���¶��£���1 mol N2O4�����ܱ������У�����ѹǿ���䣬�����¶���T1�Ĺ����У���������ɫ��Ϊ����ɫ���¶���T1�������ߵ�T2�Ĺ����У�������Ϊ��ɫ��������T2������ѹǿ��������Ϊ����ɫ����������ʵ���n���¶�T�仯�Ĺ�ϵ��ͼ��ʾ��
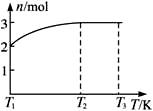
���¶���T1��T2֮�䣬��Ӧ�Ļ�ѧ����ʽ��_________________________��
���¶���T2��T3֮�䣬�����ƽ����Է��������ǣ�����1λС����______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
20���Ϳ�ѧ���ٷ�չ������������������δ������д���ѧ��ѧ�����������( )
A.������ӽṹ��ȷ�� B.��¯����
C.���³�ѹ�ϳɰ� D.�ɳ��ɵ�ص�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
A���ʺ�ʹ�ö��ڿθ��½̲ĵĿ������B���ʺ�ʹ��һ�ڿθĽ̲ĵĿ������
26.��A��ʵ����ģ��ϳɰ��Ͱ����������������£�

��֪ʵ���ҿ��ñ����������ƣ�NaNO2����Һ�뱥���Ȼ����Һ�����Ⱥ�Ӧ��ȡ������
��1������ͼ��ѡ����ȡ����ĺ���װ�ã�

���� ������
��2������������ͨ����װ�ã���װ�õ����ó��˽��������⣬���� �� 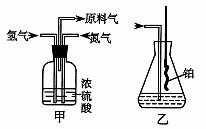
��3�����ϳ�����������ȴ����������ͨ����װ�õ�ˮ�����հ��� �����ᡱ���ᡱ������������ԭ���ǣ� ��
��4������װ������һ��ʱ�䰱����ͨ�������ͬʱ�������ȵIJ�˿������װ�õ���ƿ�ڣ���ʹ��˿���ֺ��ȵ�ԭ���ǣ� ����ƿ�л��ɹ۲쵽�������ǣ� ��
��5��д����װ���а������Ļ�ѧ����ʽ��
��6����Ӧ��������ƿ�ڵ���Һ�к���H+��OH���� �� ���ӡ�
��B��ij�����о�С�飬�ú��н϶����ʵ�ͭ�ۣ�ͨ����ͬ�Ļ�ѧ��Ӧ��ȡ����������Ƶ�ʵ�����Ϊ��
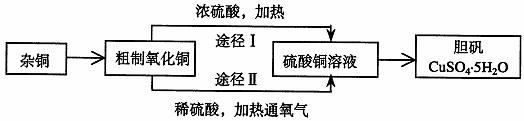
��1��ͭ�к��д������л���ɲ������յķ�����ȥ�л������ʱ������������
�ϣ����������������ı�����룬��ͬ����ȡ������Ӧʹ�� �����պ������Ӧ���� �ϣ�����ֱ�ӷ��������ϡ�
ʵ������������a ������ b ʯ���� c ������ d ������ e ����ǯ f �Թܼ�
��2����ͭ�����պ�õ��IJ���������ͭ������ͭ�Ļ���������ȡ���������պ�������ͭ�Ŀ���ԭ����
a ���չ����в�������ͭ����ԭ b ���ղ����ͭδ����ȫ����
c ����ͭ�ڼ��ȹ����зֽ�����ͭ d ��������ͭ������������
��3��ͨ��;����ʵ���ô�������ͭ��ȡ������������е�ʵ��������裺���ܡ�����ͨ���������ˡ� ����ȴ�ᾧ�� ����Ȼ����
��4���ɴ�������ͭͨ������;����ȡ��������;������ȣ�;���������Ե������ŵ��ǣ� ��
��5���ڲⶨ���õ�����CuSO4��xH2O���нᾧˮxֵ��ʵ������У������������ٽ��� ��
��6�����ⶨ���xֵƫ�ߣ����ܵ�ԭ����
a �����¶ȹ��� b ��������Ŀ����ϴ�
c ���Ⱥ���ڿ�������ȴ d �������岿�ַ绯
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ʯ��ʯ�����Ҫ�ɷ���̼��ƣ����γɹ�������ͼ��ʾ�������������������ӣ���ʪ�ܶ��е�����ʯ��ʯ���ܵ�������̼��ʴ���仯ѧ����ʽΪ��_______________________________��
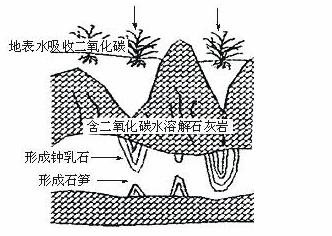
51.ijѧ�����һ�����ܶ��ײ�����ʯ����о��������������岽��ɣ���ȷ��ʵ�鲽��������_______��
�ٽ�ʢ�м���Һ����������ʯ����ϲ�
������һ��Ũ�ȵļ���Һ
��ѡ����ʵļ�
�ܵ��ڼ���Һ�µε��ٶ�
�ݹ۲�ʯ����渴ԭ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ҵ����������ʱ�����ô�������Ӧ��SO2 ת��ΪSO3��һ���ؼ����衣ѹǿ���¶ȶ�SO2ת���ʵ�Ӱ�����±���ԭ�������ɷֵ��������Ϊ�� SO2 7% ��O2 11%��N2 82%����
SO2 7% ��O2 11%��N2 82%����
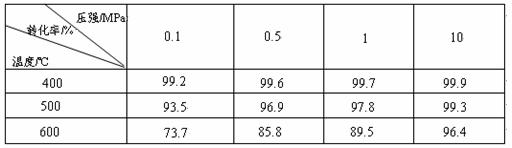
��1����֪SO2�������Ƿ��ȷ�Ӧ��������ñ��������ƶϴ˽��ۣ�
��
��2���ڴ�400��500��ʱ��SO2�Ĵ��������ó�ѹ�����Ǹ�ѹ����Ҫԭ���ǣ�
��
��3��ѡ�����˵Ĵ������Ƿ�������SO2��ת���ʣ� ����ǡ������Ƿ��������÷�Ӧ���ų��������� ����ǡ�����
��4��Ϊ���SO3�����ʣ�ʵ���������� ����SO3��
��5����֪��2SO2(g)+O2(g)��2SO3(g)����H����196.6kJ��mol��1������ÿ����1���98%��������Ҫ��SO3��������SO2������ЩSO3���ų���������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪X��Y��Z���Ƕ����ڵ�Ԫ�أ����ǵ�ԭ���������ε�����Xԭ�ӵĵ��Ӳ��������ĺ������������ͬ����Zԭ�ӵ������������Ǵ�����������Y��Z�����γ�����������̬�������
��1��X�� ��Y�� ��Z�� ��
��2����Y��Z��ɣ���Y��Z��������Ϊ7��20�Ļ�����Ļ�ѧʽ������ʽ���� ��
��3����X��Y��Z�е�����Ԫ����ɣ�����X2Z���Ӿ�����ͬ������������������ �� ��
��4��X��Y��Z�����γ�һ���Σ�������X��Y��ZԪ�ص�ԭ�ӵĸ�����Ϊ4��2��3�����εĻ�ѧʽ������ʽ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪��λ�����ϡ��Һ�У��ǻӷ������ʵķ��ӻ�������Խ�࣬����Һ�ķе��Խ�ߣ���������Һ�е���ߵ���(����)
A��0.01 mol/L��������Һ B��0.01 mol/L��CaCl2��Һ
C��0.02 mol/L��NaCl��Һ D��0.02 mol/L��CH3COOH��Һ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com