
ijѧ����������ʵ��װ��̽������ʽԭ��صĹ���ԭ��(CuԪ�ص����ԭ������Ϊ64)��
����ʵ�鲽�����λش��������⣺
(1)�����е�������Ϊ__________________(��a��b��ʾ)��
(2)д��װ����п�缫�ϵĵ缫��Ӧʽ��____________________________________��
(3)��װ����ͭ�缫����������0.64 g��������ת�Ƶĵ�����ĿΪ________��(�����á�NA����ʾ)
(4)װ�õ������г�������֬�⣬��Ҫ����KCl�ı�����Һ����ع���ʱ���������е�K����Cl�����ƶ�����ı�����ȷ����________��
| A�������е�K��������ձ��ƶ���Cl�����Ҳ��ձ��ƶ� |
| B�������е�K�����Ҳ��ձ��ƶ���Cl��������ձ��ƶ� |
| C�������е�K����Cl����������ձ��ƶ� |
| D�������е�K����Cl�����������ƶ� |
 �����Ļ���������人������ϵ�д�
�����Ļ���������人������ϵ�д� ���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д�
���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д� ��ٽ������½������������ϵ�д�
��ٽ������½������������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
1991���ҹ��״���������������ˮΪ��Դ�����͡���ˮ��ء����õ���Ժ�ˮΪ�������Һ���������е���ʹ�������������������������ȸɵ��������20��50�������ܷ�Ӧʽ��ʾΪ��4Al��3O2��6H2O===4Al(OH)3��
��1���õ�Դ�ĸ�������Ϊ ����2��д��������ӦʽΪ �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijʵ��С�������¼���ʵ��װ��̽���绯ѧ���̶Խ�����ϡ���ᷴӦ���ʵ�Ӱ��, �ձ��ж�ʢ��ϡH2SO4��



�Իش�
��1��Bװ����Cu�缫�ϵĵ缫��ӦʽΪ ��Dװ����Fe�缫�ϵĵ缫��ӦʽΪ ��
��2��Dװ���е�ʵ�������� �����õ�����������Ӧʱͨ�����ߵĵ�������Ϊ0.2mol����Fe�缫�������仯Ϊ ��
��3��B��C����װ����FeƬ����ʴ�����ʸ������ ����B��C����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼΪֱ����Դ���ϡNa2SO4ˮ��Һ��װ�á�ͨ�����ʯī�缫a��b�����ֱ�μ�һ��ʯ����Һ������ʵ����������ȷ���ǣ� ��
| A���ݳ���������a�缫��С��b�缫�� |
| B��һ�缫�ݳ���ζ���壬��һ�缫�ݳ��̼�����ζ���� |
| C��a�缫�����ʺ�ɫ��b�缫����������ɫ |
| D��a�缫��������ɫ��b�缫�������ֺ�ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪�׳ص��ܷ�ӦʽΪ�� 2CH3OH+3O2+4KOH 2K2CO3+6H2O
��1����ش�ͼ�мס������ص����ơ�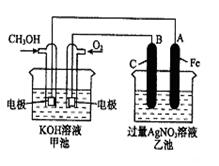
�׳��� װ�ã��ҳ��� װ�á�
��2����ش����е缫�����ƣ�ͨ��CH3OH�ĵ缫������ ��B��ʯī���缫�������� ��
��3��д���缫��Ӧʽ��ͨ��O2�ĵ缫�ĵ缫��Ӧʽ�� ��
��4���ҳ��з�Ӧ�Ļ�ѧ����ʽΪ ��
��5�����ҳ���A��Fe��������������5��40gʱ���׳�������������O2 mL����״���£�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij����С��ֱ�����ͼ��ʾװ�ö�ԭ��غ͵���ԭ������ʵ��̽����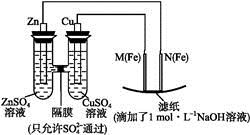

ͼ1 ͼ2
��ش�:
��.��ͼ1��ʾװ�ý��е�һ��ʵ�顣
(1)�ڱ�֤�缫��Ӧ����������,�������Cu���缫����������(�����)��
| A���� | B��ʯī | C���� | D���� |
 ������������(��������ҡ�������������)�ƶ�;��ֽ���ܹ۲쵽����������������������������������������������
������������(��������ҡ�������������)�ƶ�;��ֽ���ܹ۲쵽����������������������������������������������  )����Һ�г��Ϻ�ɫ��
)����Һ�г��Ϻ�ɫ��
 +4H2O���������� ������
+4H2O���������� ������  Fe2O3+ZnO+2K2ZnO2
Fe2O3+ZnO+2K2ZnO2�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ǧ�����ǵ��͵Ŀɳ��͵��,����ܷ�ӦΪ:Pb+PbO2+4H++2S 2PbSO4+2H2O��
2PbSO4+2H2O��
��ش���������(�������⡢����������ԭ):
�ŵ�ʱ,�����ĵ缫��Ӧ��������������������;���Һ��H2SO4��Ũ�Ƚ���������;�����·ͨ��1 mol����ʱ,�����ϸ��������������������g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ�ס����ǵ绯ѧʵ��װ�á�
(1)���ס������ձ��о�ʢ�б���NaCl��Һ��
�ټ���ʯī���ϵĵ缫��ӦʽΪ__________________________________��
�������ܷ�Ӧ�����ӷ���ʽΪ____________________________________��
�۽�ʪ��ĵ��ۣ�KI��ֽ�������ձ��Ϸ���������ֽ�ȱ�������ɫ��������Ϊ������Cl2���������ɵ�I2��
����Ӧ��Cl2��I2�����ʵ���֮��Ϊ5��1�������������ᣬ�÷�Ӧ�Ļ�ѧ����ʽΪ________________________________��
(2)���ס������ձ��о�ʢ��CuSO4��Һ��
�ټ��������ϵĵ缫��ӦʽΪ____________________________________��
�������ʼʱ����ʢ��200 mL pH��5��CuSO4��Һ(25��)��һ��ʱ�����Һ��pH��Ϊ1����Ҫʹ��Һ�ָ������ǰ��״̬��������Һ�м���___________(��д���ʵĻ�ѧʽ)________g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ��A��B��C�����ձ��зֱ�ʢ����ͬ���ʵ���Ũ�ȵ�ϡ���ᡣ
��1��A�з�Ӧ�����ӷ���ʽΪ_____________________________��
��2��B��Sn���ĵ缫��ӦʽΪ____________________________��
��3��C�б���ʴ�Ľ�����________���ܷ�Ӧ�Ļ�ѧ����ʽΪ_____________________���Ƚ�A��B��C��������ʴ�����ʣ��ɿ쵽����˳��Ϊ__________��
������ţ���
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com