
��ͼ��Na��Cu��Si��H��C��N��Ԫ�ص��ʵ��۵�ߵ͵�˳������c��d�����Ⱥ͵����
���塣�����жϲ���ȷ����
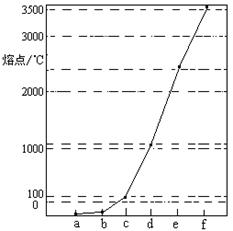
A��e��f���ʾ����ۻ�ʱ�˷����ǹ��ۼ�
B��d���ʶ�ӦԪ��ԭ�ӵĵ����Ų�ʽ��1s22s22p63s23p2
C��bԪ���γɵ���ۺ���������ˮ����֮���γ����
D������a��b��f��Ӧ��Ԫ����ԭ�Ӹ�����1��1��1�γɵķ����к�2���Ҽ���2���м�

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�о����ʵ��۽ṹ�������������������ʱ仯�ı��ʡ���ش��������⣺
��O��Si��NԪ�صĵ縺���ɴ�С��˳����____________________��C60�ͽ��ʯ����̼��ͬ�������壬�������۵�ϸߵ���____________��
��AΪ�����ڽ���Ԫ�أ������±����ݣ�A�Ļ�̬ԭ�ӵĹ����ʾʽΪ
________________________________��
| ������/kJ��mol��1 | I1 | I2 | I3 | I4 |
| A | 932 | 1821 | 15390 | 21771 |
�ǹ��ɽ���������ˮ�����γɵ�������Ƿ�����ɫ������d����ĵ����Ų��йء�һ��أ���Ϊd0��d10�Ų�ʱ������ɫ����Ϊd1��d9�Ų�ʱ������ɫ����[Cu(H2O)4]2������ɫ���ݴ��ж�25��Ԫ��Mn�γɵ��������[Mn(H2O)6]2��_____(��С����ޡ�)��ɫ��
��H��C��C��COOH�����ں��еĦҼ����м��ĸ�������Ϊ_______________������̼ԭ�ӵ��ӻ���ʽΪ___________________��
��CO������������γ���������Fe(CO)5��Fe(CO)5��һ�������·����ֽⷴӦ��Fe(CO)5(s)��Fe(s)��5CO(g)����Ӧ�����У����ѵĻ�ѧ��ֻ����λ�������γɵĻ�ѧ����������______________��
��WԪ�ص�ԭ�ӵ�M�ܲ�Ϊȫ����״̬���Һ����δ�ɶԵ���ֻ��һ����W���������Ķѻ���ʽ����ͼ�� (ѡ��ס������ҡ�����)����W������һ�������ı߳�Ϊa cm����W������ܶ�Ϊ ��д����a�ı���ʽ����NA��ʾ�����ӵ���������
�� �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�콭��ʡ�����ѧ������ѧ��������⻯ѧ�Ծ� ���ͣ������
�о����ʵ��۽ṹ�������������������ʱ仯�ı��ʡ���ش��������⣺
��O��Si��NԪ�صĵ縺���ɴ�С��˳����____________________��C60�ͽ��ʯ����̼��ͬ�������壬�������۵�ϸߵ���____________��
��AΪ�����ڽ���Ԫ�أ������±����ݣ�A�Ļ�̬ԭ�ӵĹ����ʾʽΪ
________________________________��
| ������/kJ��mol��1 | I1 | I2 | I3 | I4 |
| A | 932 | 1821 | 15390 | 21771 |



�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ�����ʡ�����и���Ԫ�µ������ۻ�ѧ�Ծ��������棩 ���ͣ������
�����غ��������ŷḻ�ĺ�ˮ��Դ����ˮ����Ҫ����Na����K����Ca2����Mg2����Cl����SO42����Br����CO32����HCO3�������ӡ�����������Դ�ͱ��������ǿɳ�����չ����Ҫ��֤��
 ��1����ˮ������������Եõ���ˮ�Ȼ�þ����ˮ�Ȼ�þ�ǹ�ҵ��ȡþ��ԭ�ϡ���д����������Ȼ�þ��ȡ����þ�Ļ�ѧ��Ӧ����ʽ???????????????????????????????? ��
��1����ˮ������������Եõ���ˮ�Ȼ�þ����ˮ�Ȼ�þ�ǹ�ҵ��ȡþ��ԭ�ϡ���д����������Ȼ�þ��ȡ����þ�Ļ�ѧ��Ӧ����ʽ???????????????????????????????? ��
��2��ij���������������л��������Cu2����Pb2������ˮ���ŷ�ǰ���ó�������ȥ���������ӣ������������ݣ�����ΪͶ��?????????? ��ѡ����Na2S������NaOH����Ч�����á�
���ܵ���� | Cu(OH)2 | CuS | Pb(OH)2 | PbS |
Ksp | 4��8��10��20 | 6��3��10��36 | 1��2��10��15 | 1��0��10��28 |
��3�������������ҹ�����Դ������ռ�ϴ���أ������ŷų���SO2�����һϵ�л�������̬���⡣���ú�ˮ������һ����Ч�ķ������乤����������ͼ��ʾ��

����Ȼ��ˮ��pH��8���������ӷ���ʽ������Ȼ��ˮ�������Ե�ԭ��????????? ����дһ������
��ij�о�С��Ϊ̽����ߺ���������SO2����Ч�ʵĴ�ʩ����������Ȼ��ˮ���պ���������ģ��ʵ�飬ʵ������ͼ��ʾ��

�������ͼʾʵ��������������һ��Ũ�Ⱥ���������SO2������Ч�ʣ����һ�����������飺??? ��
����Ȼ��ˮ�����˺��������������H2SO3��HSO3���ȷ��ӻ����ӣ�ʹ���������������Ļ�ѧԭ����?????????????????? ����дһ����ѧ����ʽ�����ӷ���ʽ���������������ˮ����Ҫ�����������Ȼ��ˮ��֮��Ϻ�����ŷţ��ò�������ҪĿ����???????????????????????? ��
������Դ����������ͷ�չ����Ҫ֧�����о���ѧ��Ӧ�����е������仯����Դ��ȱ�Ľ��������Ҫ���������塣��֪�����Ȼ�ѧ����ʽ
��2H2(g)+O2(g)��2H2O(l)????  H����570kJ/mol��
H����570kJ/mol��
��H2(g)+1/2O2(g)��H2O(g)???  H����242kJ/mol��
H����242kJ/mol��
��C(s)+1/2O2(g)��CO(g)????  H����110��5kJ/moL��
H����110��5kJ/moL��
��C(s)+O2(g)��CO2(g)???????  H����393��5kJ/moL��
H����393��5kJ/moL��
��CO2(g)+2H2O(g)��2CH4(g)+2 O2(g)?  H��+890kJ/moL
H��+890kJ/moL
�ش���������
��1��������Ӧ���������ȷ�Ӧ����??????????????? ��
��2��H2��ȼ����Ϊ��H��??????????????? ��
��3����˹�����������Ϳ�ѧ�о����к���Ҫ�����塣��Щ��Ӧ�ķ�Ӧ����Ȼ��ֱ�Ӳⶨ������ͨ����ӵķ�����á���֪C(s) + H2O(g)��H2(g)+ CO(g)????  H��akJ/moL����a��???????? ���÷�Ӧ����
H��akJ/moL����a��???????? ���÷�Ӧ���� S???????? 0(ѡ������������������������)����֪������
S???????? 0(ѡ������������������������)����֪������ G��
G�� H��T
H��T S����
S���� G��0ʱ���Է����С���÷�Ӧ��ʲô�����¿��Է�����__________________��
G��0ʱ���Է����С���÷�Ӧ��ʲô�����¿��Է�����__________________��
��4��CO��������ȼ�ϵ��Ϊ����ԭ������װ����ͼ��ʾ���õ���е����Ϊ�����ƣ������ƣ�����O2-�����ڹ������NASICON�������ƶ�������˵���������????? ��

A�������ĵ缫��ӦʽΪ��CO+O2���D2e-��CO2
B������ʱ�缫b��������O2�����缫a����缫b
C������ʱ�����ɵ缫aͨ������������缫b
D����������ͨ��������Խ����β����CO�ĺ���Խ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��ɽ��ʡ�����и���5�¸߿�ģ�����ۻ�ѧ�Ծ��������棩 ���ͣ������
����ͭ�㷺��Ӧ���ڵ�������е���졢����������
��1��Cu��Ԫ�����ڱ��е�29��Ԫ�أ�д���������ڻ�̬ԭ��δ�ɶԵ�������Cu��ͬ�ҵ縺������Ԫ���� (��Ԫ������)��
��2��CuO�����ֽ�ΪCu2O��O2�����ͭ��ԭ�ӽṹ��˵��CuO�����ֽ��ԭ��
��
��3����ͼ��ͭ��ij��������ľ���ʾ��ͼ����֪�þ����ı߳�Ϊa cm�������ӵ�����ΪNA���þ�����ܶ�Ϊ ��

��4��������ͭ��Һ�еμӰ�ˮ��������ɫ�������ڵμӰ�ˮ�������պ�ȫ���ܽ�ɵõ�����ɫ��Һ�����������м��뼫�Խ�С���Ҵ�������������ɫ��[Cu(NH3)4]SO4��H2O������
��SO42����Sԭ�ӵ��ӻ���ʽΪ ��
��NH3�����ڵ�H��N��H������������ ������ڡ������ڡ���С�ڡ���H2O�����ڵ�H��O��H���ǡ�
��S��N��O����Ԫ�ص�һ�������ɴ�С��˳��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ�꽭��ʡ������ѧ��������⻯ѧ�Ծ� ���ͣ������
�о����ʵ��۽ṹ�������������������ʱ仯�ı��ʡ���ش��������⣺
��O��Si��NԪ�صĵ縺���ɴ�С��˳����____________________��C60�ͽ��ʯ����̼��ͬ�������壬�������۵�ϸߵ���____________��
��AΪ�����ڽ���Ԫ�أ������±����ݣ�A�Ļ�̬ԭ�ӵĹ����ʾʽΪ
________________________________��
|
������/kJ��mol��1 |
I1 |
I2 |
I3 |
I4 |
|
A |
932 |
1821 |
15390 |
21771 |
�ǹ��ɽ���������ˮ�����γɵ�������Ƿ�����ɫ������d����ĵ����Ų��йء�һ��أ���Ϊd0��d10�Ų�ʱ������ɫ����Ϊd1��d9�Ų�ʱ������ɫ����[Cu(H2O)4]2������ɫ���ݴ��ж�25��Ԫ��Mn�γɵ��������[Mn(H2O)6]2��_____(��С����ޡ�)��ɫ��
��H��C��C��COOH�����ں��еĦҼ����м��ĸ�������Ϊ_______________������̼ԭ�ӵ��ӻ���ʽΪ___________________��
��CO������������γ���������Fe(CO)5��Fe(CO)5��һ�������·����ֽⷴӦ��Fe(CO)5(s)��Fe(s)��5CO(g)����Ӧ�����У����ѵĻ�ѧ��ֻ����λ�������γɵĻ�ѧ����������______________��
��WԪ�ص�ԭ�ӵ�M�ܲ�Ϊȫ����״̬���Һ����δ�ɶԵ���ֻ��һ����W���������Ķѻ���ʽ����ͼ�� (ѡ��ס������ҡ�����)����W������һ�������ı߳�Ϊa cm����W������ܶ�Ϊ ��д����a�ı���ʽ����NA��ʾ�����ӵ���������



�� �� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com