
������������A��B��C��D��E�����ǵ������ӿ�����Na���� ��Cu2����Ba2����Al3����Ag����Fe3���������ӿ�����Cl����
��Cu2����Ba2����Al3����Ag����Fe3���������ӿ�����Cl���� ��
�� ��
�� ����֪��
����֪��
�������ξ�����ˮ��ˮ��Һ��Ϊ��ɫ��
��D����ɫ��Ӧ�ʻ�ɫ��
��A����Һ�����ԣ�B��C��E����Һ�����ԣ�D����Һ�ʼ��ԡ�
�������������ε���Һ�зֱ����Ba(NO3)2��Һ��ֻ��A��C����Һ������������
�������������ε���Һ�У��ֱ���백ˮ��E��C����Һ�����ɳ����������Ӱ�ˮ��C�г�����ʧ��
�ް�A����Һ�ֱ���뵽B��C��E����Һ�У��������ɲ�����ϡ����ij�����
��ش��������⣺
(1)�������У�һ��û�е��������� ��������������ͬ�������εĻ�ѧʽ�� ��
(2)D�Ļ�ѧʽΪ ��D��Һ�Լ��Ե�ԭ���� (�����ӷ���ʽ��ʾ)��
(3)A��C����Һ��Ӧ�����ӷ���ʽ�� ��
E�Ͱ�ˮ��Ӧ�����ӷ���ʽ�� ��
(4)��Ҫ����B�������������ӣ���ȷ��ʵ�鷽���� ��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�����£����и������ӡ�������ָ����Һ��һ���ܴ����������
| A��Na2S��Һ�� SO42-�� K����Br����Cu2������ |
| B����c(H+)=1.0��10-13mol��L-1����Һ�� Na+��S2����AlO2-��SO32- |
| C����ʹ�����Ժ�ɫ����Һ�� K+��MnO4-��H2C2O4��SO42- |
| D���� NH3��H2O ����Һ��Ba2+��NO3-��Cl-��Ag+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������I��II����ȷ���������ϵ����
| ѡ�� | ����I | ����II |
| A | KNO3���ܽ�ȴ� | ���ؽᾧ����ȥKNO3�л��е�NaCl |
| B | BaSO4�������� | �������BaCl2��Һ����SO42- |
| C | NH3��ʹ��̪��Һ��� | NH3�����������Ȫʵ�� |
| D | Ca(OH)2���Ƴɳ���ʯ��ˮ | ������2.0 mol?L-1��Ca(OH)2��Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Ͼ�����ӳص���������(��Ҫ����LiCoO2��������Al��Fe��)�����ڻ����ܡ�ﮣ������������£�
(1)�������ܽ�����У�S2O32-��������SO42-��LiCoO2���ܽ�����з�����Ӧ�Ļ�ѧ����ʽΪ ����ԭ������ ��
(2)���������ӹ����У�ͨ������������� ��������Ҫ�ɷ��� ��
(3)�������ܡ��͡�����ﮡ������ӷ���ʽ�ֱ�Ϊ ��
(4)���˷������Ի��������⣬�����ջ��ɻ��յĸ���Ʒ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧ����ѡ��2����ѧ�뼼������15�֣�
��ˮAlCl3���������������л��ϳɵĴ����ȡ���ҵ����������A12O3��Fe2O3��Ϊԭ���Ʊ���ˮAlCl3�Ĺ����������¡�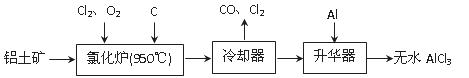
��1���Ȼ�¯��Al2O3��C12��C��Ӧ�Ļ�ѧ����ʽΪ ��
��2����Na2SO3��Һ�ɳ�ȥ��ȴ���ų�β���е�Cl2���˷�Ӧ�����ӷ���ʽΪ ��
��3������������Ҫ����AlCl3��FeCl3�����������Al���������� ��
��4��Ϊ�ⶨ�Ƶõ���ˮAlCl3��Ʒ��������FeCl3���Ĵ��ȣ���ȡ16.25 g��ˮAlCl3��Ʒ�����ڹ�����NaOH��Һ�����˳�����������ᆳϴ�ӡ����ա���ȴ�����أ�������������Ϊ0.32 g��
��д�����ӹ������漰�����ӷ���ʽ �� ��
��AlCl3��Ʒ�Ĵ���Ϊ ��
��5����ҵ����һ��������Ϊԭ���Ʊ���ˮAlCl3�����У����һ������AlCl3��6H2O��ˮ�Ʊ���ˮAlCl3��ʵ����һ���ķ����� ��
��6�������к���һ����̼�����Fe3C������ҵ��Ҫ�ⶨ̼Ԫ�ص�������������һ�����������Ŀ��������գ������д��ԵĹ��壬�÷�Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Ƴ���ͭ��ˮ�к���CN-��Cr2O72-����,��Ҫ������������ŷš��ó��ⶨ�������̽��з�ˮ������ �ش��������⣺
��1������������ˮ��������Ҫʹ�õķ����� ��
��2�����з�Ӧ��������ų����÷�Ӧ�����ӷ���ʽΪ ��
��3��������У�ÿ����0.4mol Cr2O72-ʱת�Ƶ���2.4mol���÷�Ӧ���ӷ���ʽΪ ��
��4��ȡ��������ˮ�����Թ��У�����NaOH��Һ���۲쵽����ɫ�������ɣ��ټ�Na2S��Һ�� ��ɫ����ת���ɺ�ɫ��������ʹ�û�ѧ��������ֽ��Ͳ����������ԭ�� ��
��5��Ŀǰ��������Cr2O72-��ˮ������������巨���÷������ˮ�м���FeSO4��7H2O��Cr2O72-��ԭ��Cr3+������pH��Fe��Crת�����൱�� (��������,�������ֱ�ʾԪ�ؼ�̬)�ij���������1mol Cr2O72-�������a mol FeSO4?7H2O�����н�����ȷ���� ��
(��������,�������ֱ�ʾԪ�ؼ�̬)�ij���������1mol Cr2O72-�������a mol FeSO4?7H2O�����н�����ȷ���� ��
| A��x ="0.5" ,a =8 | B��x ="0.5" ,a = 10 | C��x =" 1.5" ,a =8 | D��x =" 1.5" ,a = 10 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ɫϡ��ҺX�У����ܺ����±����������е�ij���֡�
| ������ | CO32����SiO32����AlO2����Cl�� |
| ������ | Al3����Fe3����Mg2����NH4����Na�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��һƿ�������Һ�����п�����NH4+��K����Na����Mg2����Ba2����Al3����Cl����I����NO3-��CO32-��S2����SO42-��AlO2-��MnO4-��ȡ����Һ��������ʵ�飺
(1)ȡpH��ֽ���飬��Һ��ǿ���ԣ������ų�________���ӵĴ��ڣ�
(2)ȡ��������Һ����������CCl4������������ˮ������CCl4�����ɫ�������ų�________���ӵĴ��ڣ�
(3)��ȡ��������Һ��μ���NaOH��Һ��ʹ��Һ��������Ϊ���ԣ��ڵμӹ����к͵μ���Ϻ���Һ���������������ֿ����ų�________���ӵĴ��ڣ�
(4)ȡ����������������Һ�μ�Na2CO3��Һ���а�ɫ�������ɣ�֤��________���Ӵ��ڣ��ֿ��ų�________���ӵĴ��ڣ�
(5)��(3)�õ��ļ�����Һ���ȣ�������ų�����������ʹʪ��ĺ�ɫʯ����ֽ��������������ʵ����ʵȷ��������Һ�п϶����ڵ�������________��������ȷ���Ƿ���ڵ�������________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������A��B����ѧ���������ʣ�����������ֻ�ܴ��±���ѡ��
| ������ | K����Na����Fe2����Ba2����NH4+��Ca2�� |
| ������ | OH����NO3����I����HCO3����AlO2����HSO4�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com