
����Ŀ����֪����ʵ����ʵ��
��Cr2O3�����������KOH��Һ�õ�KCrO2��Һ��������������õ�Cr2(SO4)3��Һ��
����KCrO2��Һ�еμ�H2O2��Һ�����ữ���ɵ�K2Cr2O7��Һ��
����K2Cr2O7��Һ�μӵ����ۺ�KI�Ļ����Һ������Һ������
�����жϲ���ȷ����( )
A��������KCrO2��CrԪ��Ϊ��3��
B��ʵ����֤��Cr2O3������������
C��ʵ����֤��H2O2�������������л�ԭ��
D��ʵ����֤����������Cr2O![]() >I2
>I2
���𰸡�C
��������
���������A��������KCrO2�У�KΪ+1�ۣ�OΪ-2�ۣ��ɻ��������������ϼ۵ĵ�����Ϊ0����CrԪ��Ϊ+3�ۣ���ȷ��B���ɷ�Ӧ����֪�����������ᡢ�Ӧ�����κ�ˮ��Ϊ�����������ȷ��C��ʵ�����з���������ԭ��Ӧ��CrԪ�صĻ��ϼ���+3������Ϊ+6�ۣ������������OԪ�صĻ��ϼ���-1�۽���Ϊ-2�ۣ�����֤��H2O2�������ԣ�����D����������Һ���������ɵⵥ�ʿ�֪������������ԭ��Ӧ��CrԪ�صĻ��ϼ۽��ͣ�IԪ�صĻ��ϼ����ߣ����������������Դ�����������������Կ�֪��֤����������Cr2O![]() >I2����ȷ��
>I2����ȷ��


 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������β����������Ⱦ���̼�⻯�������������һ����̼�⣬����Ҫ����(����)
A. ���� B. ���� C. ���� D. ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���Ͻ�A�����ֵ�����ɣ���֪���ٺϽ�A�в���K��Ca��Na��������B��ȼ��������GΪ�����³´�л����Ҫ����֮һ�����ͼ�ش��������⣺

��1���Ͻ�A����Щ�������(�û�ѧʽ��ʾ)��_____��_______��________��________��
��2��д����Ӧ�ٵĻ�ѧ����ʽ��______________��
��3��д����Ӧ�ڵĻ�ѧ����ʽ��__________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ���ҴӺ����Һ(��H2O�⣬������CCl4��I2��I���ȣ��л��յ��CCl4����ʵ��������£�

(1)���Һ�м����Թ�����Na2SO3��Һ������Һ�е�I2��ԭΪI���������ӷ���ʽΪ
______________���ò�����I2��ԭΪI������ҪĿ���� ____________ ��
(2)����X������Ϊ ___��
(3)����ʱ����������ƿ�н���I����ˮ��Һ���������pHԼΪ2������ͨ��Cl2����40�����ҷ�Ӧ��ʵ��װ����ͼ��ʾ����ʵ������ڽϵ��¶��½��е�ԭ����_____________����ƿ��ʢ�ŵ���ҺΪ_______________
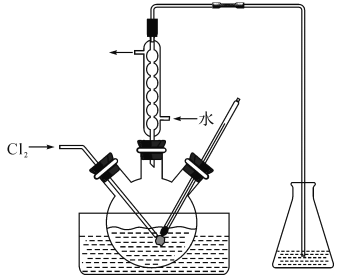
(4)�ڽ̲ĵ�ˮ����ȡʵ���У����Ҫ�ӵ��CCl4��Һ�л�������CCl4�����Բ������з�������ˮԡ����������CCl4��Һ���ռ��õ�dz��ɫ��Һ�塣�����Һ���м���������״�Ļ���̿�����á�����Һ��dz��ɫ��ʧ������Y�����ɵ���ɫ����CCl4Һ�塣
���ռ��õ�dz��ɫ��Һ��˵�����ռ��õ���Һ���к��� ___����ѧʽ�����ڼ�����������̿��Ŀ���� ______ ���۲���Y�������� ___��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��25 ��ʱ���������ʵĵ���ƽ�ⳣ�������ʾ��
��ѧʽ | CH3COOH | H2CO3 | HClO |
����ƽ�ⳣ�� | 1��7��10��5 | K1��4��3��10��7 K2��5��6��10��11 | 3��0��10��8 |
��ش��������⣺
��1��CH3COOH��H2CO3��HClO��������ǿ������˳��Ϊ__________________��
��2��ͬŨ�ȵ�CH3COO����HCO3-��CO32-��ClO�����H����������ǿ������˳��Ϊ____________________��
��3��������0��1 mol��L��1��CH3COOH��Һ�ڼ�ˮϡ�����У����б���ʽ������һ����С����___________��
A��c��H���� B��c��H����/c��CH3COOH��
C��c��H������c��OH���� D��c��OH����/c��H����
E��![]()
������Һ�����¶ȣ�����5�ֱ���ʽ�������������___________________��
��4�����Ϊ10 mL pH��2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1 000 mL��ϡ������pH�仯��ͼ��ʾ��

����ͬ������HX�ĵ���ƽ�ⳣ��__________________��������������������������С����������ĵ���ƽ�ⳣ����������____________________��ϡ�ͺ�HX��Һ����ˮ���������c��H����______________��������������������������С������������Һ����ˮ���������c��H������������___________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������������Ԫ�أ�a��b��c��d��ԭ������������������Ԫ���γɵĵ�������Ϊm��n��p��qʱ��x��y��z����ЩԪ����ɵĶ�Ԫ���������zΪ�γ��������Ҫ����֮һ��25��,0.01mol��L��1w��Һ�У�  ���������ʵ�ת����ϵ��ͼ��ʾ������˵����ȷ���ǣ�������
���������ʵ�ת����ϵ��ͼ��ʾ������˵����ȷ���ǣ�������

A. ԭ�Ӱ뾶�Ĵ�С��a<b<c<d B. �⻯��ķе㣺b>d
C. x�ĵ���ʽΪ��![]() D. y��w���еĻ�ѧ��������ȫ��ͬ
D. y��w���еĻ�ѧ��������ȫ��ͬ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����Mg��P��S��Cl��Ni��Ԫ����ɵ����Ͳ������Ź㷺����;����ش��������⡣
��1����̬Mgԭ��L������Ų�ʽΪ_____��P��S��Cl�ĵ�һ�������ɴ�С˳��Ϊ_______��
��2��PH4Cl�ĵ���ʽΪ_________��Ni��CO���γ������Ni(CO)4���÷����ЦҼ���м�������Ϊ__________��
��3��SCl2�����е�����ԭ���ӻ����������_______���÷��ӹ���Ϊ_________��
��4����֪MgO��NiO�ľ���ṹ����ͼ1����ͬ������Mg2+��Ni2+�����Ӱ뾶�ֱ�Ϊ66pm��69 pm�����۵㣺MgO_____NiO���>������<����=������������_____________��

��5����ͼ����ʾ��NiO�����������������AΪ��0��0��0����BΪ��1��0��1������C�����������Ϊ___________��
(6)һ���¶��£�NiO��������Է��ط�ɢ���γɡ������Ӳ㡱��������ΪO2�������õ������У�Ni2+������У���ͼ2������֪O2���İ뾶Ϊa m��ÿƽ��������Ϸ�ɢ�ĸþ��������Ϊ______g������a��NA��ʾ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��2002��ŵ������ѧ������ߵĹ���֮һ�Ƿ����˶��л����ӵĽṹ���з������������䷽�����ü�������10��9g���ң��Ļ�����ͨ�������ǵ����ӻ���ʹ��Ʒ���Ӵ������ӻ��������������ѳɸ�С�����ӡ���C2H6���ӻ���ɵõ�C2H6����C2H5����C2H4��������Ȼ��ⶨ���ʺɱ�������H�����ʺɱ�Ϊ1��ij�л�����Ʒ���ʺɱ�����ͼ���������Ӿ���һ����λ����ɣ��ź�ǿ��������Ӷ����йأ�������л������

A. CH3OH B. C3H8 C. C2H4 D. CH4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����������������һЩͻ���¼�������Ҫ��������ѧ����֪ʶ����ȡ��ѧ����Ч�ķ��������Լ����������������й©�������Ծȵõ�����
A. ��ʪë����պ�з���ˮ��ë����ס�ڱdz���
B. ����Ƶ͵ĵط�����
C. �۲����˳�糷��
D. �����ڷ�һ��ˮ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com