
����Ŀ��������þ������һ������ϡHCl�У������������ݺ���ֻҰ�ɫ���塣ijͬѧ�Ը�����������̽����(��֪��MgCl2����������ˮ)
(һ)�������ݷ�Ӧ�Ļ�ѧ����ʽ____��
(��)���ˡ�ϴ�ӡ��������ɵûҰ�ɫ���塣�ԻҰ�ɫ���������²²Ⲣ����ʵ�飺
���²⣩�Ұ�ɫ��������ǣ���ʣ���Mg�� ��____�� ��Mg��MgCl2���塣
��ʵ��1��(1)ȡ�Ұ�ɫ������Ʒ������ϡHCl�������ݲ�������²� __(ѡ�����)��������(2)��ȡ�������ĻҰ�ɫ�����MgCl2����ֱ����ͬ�¡�����ˮ�У��Ұ�ɫ���岻�ܽ⣬��MgCl2����ȫ��____����²�ڲ�������
���������ϣ����Ұ�ɫ����ijɷ�Ϊ��ʽ�Ȼ�þ����ѧʽ�ɱ�ʾΪMgx(OH)yClznH2O[��ÿ��Mgx(OH)yClz�����n��H2O]�������ȷֽ�ɲ�������þ���Ȼ����ˮ��
��ʵ��2����ȡ�ûҰ�ɫ����47.5g��������ȷ�IJ�������������ȷֽ�ʵ�飬����Ӧֹͣ�������þ��������Ϊ24.0g��ˮ������16.2g��
���������ۣ�
(1)X��Z=____��
(2)Mgx(OH)yCl�Ļ�ѧʽ������ ______��
(3)������Mgx(OH)yClznH2O�У�n��5����Y����ֵΪ____
���𰸡�Mg+2HCl=MgCl2+H2�� MgCl2���� �١��� �ܽ� X��Z=3:1 Mg3(OH)5Cl 10
��������
(һ)þ�����������Ȼ�þ����������Ӧ�Ļ�ѧ����ʽMg+2HCl=MgCl2+H2����
(��) [�²�]��Ӧ���������һ�����������������ʣ��ķ�Ӧ��Ұ�ɫ��������ǣ���ʣ���Mg�� ��MgCl2���壻 ��Mg��MgCl2���壻
[ʵ��1](1)þ�����ᷴӦ�����������������ݣ��Ȼ�þ�������Ӧ��ȡ�Ұ�ɫ������Ʒ������ϡHCl�������ݲ�������û��þ���²�٢۲�������
(2)þ��������ˮ����ȡ�������ĻҰ�ɫ�����MgCl2����ֱ����ͬ�¡�����ˮ�У��Ұ�ɫ���岻�ܽ⣬��MgCl2����ȫ���ܽ⣬��²�ڲ�������
[ʵ��2] ��ʽ�Ȼ�þ����ѧʽ�ɱ�ʾΪMgx(OH)yClznH2O[��ÿ��Mgx(OH)yClz�����n��H2O]�������ȷֽ�ɲ�������þ���Ȼ���͡���ȡ�ûҰ�ɫ����47.5g��������ȷ�IJ�������������ȷֽ�ʵ�飬����Ӧֹͣ�������þ��������Ϊ24.0g��þԪ�ص�����Ϊ��24.0g��![]() =14.4g��ˮ������16.2g������Ԫ�ص�����Ϊ��16.2g��
=14.4g��ˮ������16.2g������Ԫ�ص�����Ϊ��16.2g��![]() =1.8g �������Ȼ������������Ϊ��47.5g-24.0g-16.2g=7.3g����Ԫ�ص�����Ϊ7.3g��
=1.8g �������Ȼ������������Ϊ��47.5g-24.0g-16.2g=7.3g����Ԫ�ص�����Ϊ7.3g��![]() =7.1g����Ԫ�ص�����=7.3g-7.1g=0.2g��
=7.1g����Ԫ�ص�����=7.3g-7.1g=0.2g��
[��������]
(1)X��Z=![]() ��
��![]() =3:1��
=3:1��
(2) X��Z =3:1����X=3��Z=1ʱ���ɵ�Mg3(OH)yCl����������Ԫ�صĻ��ϼ۴�����Ϊ�㣬��������-1�ۣ�þ��+2�ۣ�����-1�ۣ�����y=5����ѧʽ��Mg3(OH)5Cl��
(3) X��Z =3:1����Z=m����x=3m��������Mgx(OH)yClznH2O�У�n��5��Mgx(OH)yClznH2O�ɱ�ʾΪMg3m(OH)yClm5H2O�������������֪����������Ԫ�ع�2g����Ԫ�ع�7.1g
�ɵã�![]() ��
��
![]()
�ⷽ�����Y����ֵΪ10��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ������ʵ������ȡ���ռ������װ�ã���ش��������⣺

��1��д����ͼ�д�������������ƣ���________����________��
��2���ø��������ȡ��������Ӧ�Ļ�ѧ����ʽ��________���ռ�������ѡ��___������ĸ��ţ���ͬ����
��3����ʵ������ȡ������̼����Ӧ�Ļ�ѧ����ʽ��______��Ӧѡ��ķ���װ����_______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ʯ��ʯ��Դ�ḻ��ij����С��ͬѧΪ�˲ⶨʯ��ʯ��̼��Ƶ�����������ȡij��ʯ��ʯ��Ʒ9.0g���ձ��У���50gϡ�����5�μ����ձ��У���ַ�Ӧ�����ʲ���ϡ���ᷴӦ�������ʣ������������¼������
���� | 1 | 2 | 3 | 4 | 5 |
����ϡ��������/g | 10 | 10 | 10 | 10 | 10 |
ʣ���������/g | 7.0 | 5.0 | 3.0 | 1.5 | X |
����㣺
��1��X��ֵΪ ��
��2����ʯ��ʯ��Ʒ��̼��Ƶ���������Ϊ ��
��3������ϡ�������ʵ�������������д��������̣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ȡ10gʯ��ʯ��Ʒ���ձ��У��������м�������ϡ���ᣨ���ʲ���Ӧ�����й�ʵ�����ݼ��±���
��Ӧǰ | ��Ӧ�� | |
�ձ���ϡ��������� | ʯ��ʯ��Ʒ������ | �ձ������л��������� |
160g | 10g | 166.7g |
��1��CaCO3��CaԪ�ص���������Ϊ____________��
��2������CO2������____________��
��3����ʯ��ʯ��Ʒ��̼��Ƶ���������____________��д��������̣���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ��ʾ��������ƿ�е�ˮʹ֮���ڡ�ˮ������ͭ��������ѻ���ܿ�B�������ȼ�գ�Ѹ���ƿ�����ʼȼ�ա�
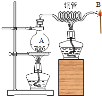
(1)B��ˮ������������ʹ���____(��ȼ������֮һ����ͬ)��
(2)�ѻ���ܿ�B�������ȼ�գ�Ѹ���ƿ�����ʼȼ��˵��____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����۵ĽǶ��˽����ʼ���仯�������ڸ��õ���ʶ������ɺͱ仯�ı��ʡ�
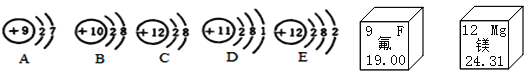
��1����֪A�����ԭ������Ϊ19������A����������Ϊ___________��A��E�γɻ�����Ļ�ѧʽΪ_____________��
��2������A��B��C��D��E�У�����ͬһ��Ԫ�ص���___________������ţ������ڵڶ����ڵ�Ԫ�ع�___�֡�
��3��D��E��ѧ���ʲ�ͬ��ԭ����_____________��
��4����ͼΪ���ֻ�������һ�������·�����Ӧ����ʾ��ͼ��
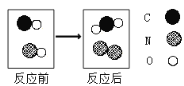
�ٸ÷�Ӧ�Ļ�ѧ����ʽΪ__________________��
��������Ӧ�У���Ϊ��ԭ����������________________�������ʻ�ѧʽ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼΪ���ˮ��ʵ��װ��ͼ����ͨ��Դ�۲쵽���缫�������ݲ�����

��1��ͨ��һ��ʱ�������b�ռ���������ļ��鷽����_____��a��b���������ռ��������������ԼΪ_____��
��2����ͼΪˮ�ֽ���۹���:

��ˮ�ķֽ�����У��������仯������_____��
�ڽ��ʵ������д��ˮͨ��ֽ�����ֱ���ʽ:_____��
��3����֪ͬ��ͬѹ�£�����������ȡ��������ķ��Ӹ����ȣ���ʵ�����ܹ�˵��ˮ�Ļ�ѧ����Ϊ��H2O����ʵ��������_____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��̼Ԫ����ɵĵ����ж��֡���ش��������⣺
��1�����нṹͼ�У����α�ʾʯī�����ʯ��C60 ���ǣ�����ţ�_____��
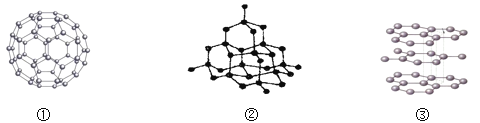
��2�����ʯ��ʯī��C60������_____��ɵĵ��ʣ������ߵ����������кܴ�IJ��죬��Ҫ����Ϊ_____��
��3�����������У���Ҫ��ʯī��ȡ���ǣ�����ţ�_____��
��Ǧ��о �ڲ����� ��譲Ӷ�Ŀ����Ȼ��ʯ �ܸɵ�ص缫 ����̽������ͷ ������ ����糵�ġ���ˢ��
��4����Ϊ��˾���������Ƴ����³�����ʯīϩ������ӵ�أ���֪ʯīϩ��̼�ĵ��ʣ�����ʯī�ֱ�����������ȫȼ�յIJ���_____�����ǡ�����ͬ��
��5��ʯī��һ�������¿�ת��Ϊ���ʯ���ñ仯����_____�仯��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���������ƾ��壨CaO28H2O�����ȶ����ʰ�ɫ������ˮ���㷺Ӧ���ڻ���ɱ�����������Ա���Ϊԭ���Ʊ�CaO2�������£�
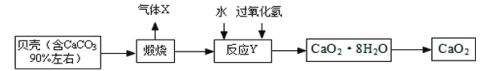
��1������X��______�����������ƾ�������Һ����IJ���������______��
��2����ӦY�ǻ��Ϸ�Ӧ����Ӧ������CaO28H2O����д����ѧ����ʽ______�������ѣ�CaO28H2O�ɿ�����1��CaO2���Ӻ�8��ˮ���ӹ��ɵ�ˮ���
��3���������ƾ��壨CaO28H2O����H��OԪ�ص�������Ϊ_____��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com