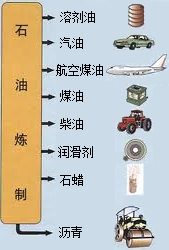
ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã�����ͼ������ѧ�Ƽ������߰�ȼ�����нϴ�ķ����ѽ�ɺ��������������ĸ�̼ԭ�ӵȵ�С���ӣ�Ȼ������Ǽӹ��Ƴɸ��ֲ�Ʒ�������ϡ��ϳ���ά���ϳ���ҩ�ũҩ��ըҩ�����ʵȵȣ�
��1��ʯ������
������������ʯ�ͷ�������
���������ѧ���仯��
��2��������ʯ�����ƵIJ�Ʒ֮һ��ֱ����Ϊȼ��ʹ������ɻ�����Ⱦ���������м��������Ҵ����ɽ�ʡʯ����Դ����������β����Ⱦ��д���Ҵ��ڿ�����ȼ�յĻ�ѧ����ʽ
��
��3��ֻҪ���˶������̼Ҿ��ṩ��ѵ����ϴ��������ǵ������ṩ�˺ܶ�������������ڹ���ʹ�����Ϸ������ɵġ���ɫ��Ⱦ�����ѳ�Ϊһ�����ص�������⣬Ϊ�ˣ�����Ժ�칫������֪ͨ���ӽ����6��1�������еij��С��̳������м�ó�г��������ṩ��ѵ����ϴ���ij��ѧ�о�С���ͬѧ����ij�����Ϸ������������ʾ������ֻ��C��H����Ԫ�أ�����ɽ��вⶨ�����������ʵ�飬��ͼ��ʾ��
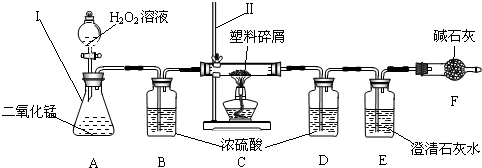
���������������
���������������
��
��װ��A���������ķ�Ӧ��1ѧ����ʽΪ
��
װ��E���������ķ�Ӧ��ѧ����ʽΪ
��
��װ��B��������
��
���о�С���ͬѧ��ʵ��ǰ����γ���װ��D����Ŀ����
��
��������C�IJ������з����������������ΪWg�������������ȼ�պ�������D����a g����Wg�����������к���Ԫ�ص�����Ϊ
g����������Ϊ������ʽ��
����ʵ����û������װ��B���ⶨ��������������Ԫ�����������Ľ����
���ƫС������ƫ������Ӱ�족����

 ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã�����ͼ������ѧ�Ƽ������߰�ȼ�����нϴ�ķ����ѽ�ɺ��������������ĸ�̼ԭ�ӵȵ�С���ӣ�Ȼ������Ǽӹ��Ƴɸ��ֲ�Ʒ�������ϡ��ϳ���ά���ϳ���ҩ�ũҩ��ըҩ�����ʵȵȣ�
ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã�����ͼ������ѧ�Ƽ������߰�ȼ�����нϴ�ķ����ѽ�ɺ��������������ĸ�̼ԭ�ӵȵ�С���ӣ�Ȼ������Ǽӹ��Ƴɸ��ֲ�Ʒ�������ϡ��ϳ���ά���ϳ���ҩ�ũҩ��ըҩ�����ʵȵȣ�


 �߽�������ϵ�д�
�߽�������ϵ�д�
 �й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����
�й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����
 �й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����
�й�ʯ����Ȼ�����Ź�˾�����������ڲ�����̲���������ִ�����ģ��ʮ�ڶֵĴ�����--�����ϱ���������ǿ�ҹ���Դ��ȫ��Ӧ�ı�������������Ҫ���壮ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã���ͼ����
 ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã�����ͼ������ѧ�Ƽ������߰�ȼ�����нϴ�ķ����ѽ�ɺ��������������ĸ�̼ԭ�ӵȵ�С���ӣ�Ȼ������Ǽӹ��Ƴɸ��ֲ�Ʒ�������ϡ��ϳ���ά���ϳ���ҩ�ũҩ��ըҩ�����ʵȵȣ�
ʯ������Ҫ����̼��������Ԫ�أ�����ʯ���и��ɷֵķе㲻ͬ�������Ƿ��루��Ϊ�����ɵõ���ͬ�IJ�Ʒ��ʹʯ�͵õ��ۺ����ã�����ͼ������ѧ�Ƽ������߰�ȼ�����нϴ�ķ����ѽ�ɺ��������������ĸ�̼ԭ�ӵȵ�С���ӣ�Ȼ������Ǽӹ��Ƴɸ��ֲ�Ʒ�������ϡ��ϳ���ά���ϳ���ҩ�ũҩ��ըҩ�����ʵȵȣ�
