



 ����ѧУ�ֲ����ܲ�ϵ�д�
����ѧУ�ֲ����ܲ�ϵ�д� �ƸԺ���ȫ�����Ų��Ծ�ϵ�д�
�ƸԺ���ȫ�����Ų��Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��C | B��N | C��O | D��Fe |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����������[Be��OH��2]�ļ��Ա�������þ�� |
| B������At��Ϊ��ɫ���壬HAt���ȶ� |
| C�������ȣ�SrSO4����������ˮ�İ�ɫ���� |
| D�������⣨H2Se������ɫ���ж�����H2S�ȶ������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| AԪ��ԭ�ӵĺ���p����������s����������1 |
| BԪ��ԭ�Ӻ���s����������p����������ȣ��Ҳ���AԪ����ͬһ���� |
| Cԭ�Ӻ�������p���ȫ������� |
| DԪ�ص������������������IJ�Ϊ4 |
| E��ǰ�������е縺����С��Ԫ�� |
| F�����ڱ��ĵ����� |
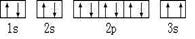 ��Υ���� ԭ����
��Υ���� ԭ����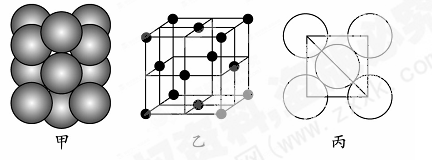
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 �������й�˵����ȷ����________����ѡ����ĸ����
�������й�˵����ȷ����________����ѡ����ĸ����| A��NaN3��KN3�ṹ���ƣ�ǰ�߾����ܽ�С |
B������صľ����ṹ����ͼ��ʾ��ÿ�������з�̯2����ԭ�� |
| C�����ĵ�һ�����ܴ����� |
| D�����������º��ȶ�������Ϊ���ĵ縺��С |
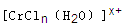 ��n��x��Ϊ���������������ӣ�����ͨ�������ӽ�����֬(R-H)���ɷ������ӽ�����Ӧ��
��n��x��Ϊ���������������ӣ�����ͨ�������ӽ�����֬(R-H)���ɷ������ӽ�����Ӧ��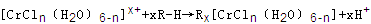
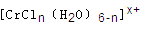 ����Һ����R-H��ȫ�������к����ɵ���Ũ��Ϊ
����Һ����R-H��ȫ�������к����ɵ���Ũ��Ϊ�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

 4FeCl2+N2O��+6HCl+H2O
4FeCl2+N2O��+6HCl+H2O ����������
����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
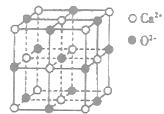
| ��A��B��C��D��E��FΪ����������Ԫ�أ�ԭ�Ӱ뾶��С��ϵΪA��D��C��B��F��E�� |
| ��A��D�γɵĻ����ﳣ����ΪҺ̬�� |
| ��BԪ��ԭ�Ӽ۵��ӣ���Χ���ӣ��Ų�ΪnSnnPn |
| ��FԪ��ԭ�ӵĺ���p����������s����������1�� |
| �ݵ�һ�����ܣ�F��E�� |
| ��G�Ļ�̬ԭ�Ӻ�����6��δ�ɶԵ��ӣ� |
��H���γɺ�ɫ����ש��ɫ���� �ͺ�ɫ��HD���ֻ���� �ͺ�ɫ��HD���ֻ���� |
 ��Bԭ�Ӳ�ȡ���ӻ��������Ϊ ��
��Bԭ�Ӳ�ȡ���ӻ��������Ϊ �� ���ӵĿռ乹��Ϊ ��
���ӵĿռ乹��Ϊ �� ��������
�������� ,ԭ���� ��
,ԭ���� �� ��ʾ����٤�������������ı߳�Ϊa�������ĸ�Ϊh�������ܶȿɱ�ʾΪ g/cm3������ֻ��r��
��ʾ����٤�������������ı߳�Ϊa�������ĸ�Ϊh�������ܶȿɱ�ʾΪ g/cm3������ֻ��r��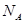 ����ʽ��ʾ��
����ʽ��ʾ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| A��SO2��O3��PO43-��ClO4-��Ϊ�ȵ����� |
| B���������ʧȥһ��H�����γ�CH3-����̼ԭ�ӵ��ӻ����ͷ����˸ı� |
| C��Tiԭ�ӵĺ�������Ų�ʽΪ[Ar]3d34s2 |
| D��CS2��H2O��C2H2����ֱ���η��� |

�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com