
“‘ΜΤΧζΩσΈΣ‘≠Νœ÷ΤΝρΥα≤ζ…ζΒΡΝρΥα‘ϋ÷–Κ§Fe2O3ΓΔSiO2ΓΔAl2O3ΓΔMgOΒ»ΓΘ Β―ι “ΡΘΡβΙΛ“Β“‘ΝρΥα‘ϋ÷Τ±ΗΧζΚλ(Fe2O3)Θ§Ιΐ≥Χ»γœ¬ΘΚ
Θ®1Θ©ΝρΥα‘ϋΒΡ≥…Ζ÷÷– τ”ΎΝΫ–‘―θΜ·ΈοΒΡ « Θ§ –¥≥ωΥα»ήΙΐ≥ΧFe2O3”κœΓΝρΥαΖ¥”ΠΒΡάκΉ”Ζ¥”ΠΖΫ≥Χ ΫΘΚ ΘΜ
Θ®2Θ©…ζ≤ζΙΐ≥Χ÷–Θ§ΈΣΝΥ»Ζ±ΘΧζΚλΒΡ¥ΩΕ»Θ§―θΜ·Ιΐ≥Χ–η“ΣΒςΫΎ»ή“ΚΒΡpHΒΡΖΕΈß «_________ΘΜΘ®≤ΩΖ÷―τάκΉ”“‘«β―θΜ·Έο–Έ Ϋ≥ΝΒμ ±»ή“ΚΒΡpHΦϊœ¬±μΘ©
| ≥ΝΒμΈο | Fe(OH)3 | Al(OH)3 | Fe(OH)2 | Mg(OH)2 |
| ΩΣ Φ≥ΝΒμ | 2.7 | 3.8 | 7.5 | 9.4 |
| Άξ»Ϊ≥ΝΒμ | 3.2 | 5.2 | 9.7 | 12.4 |
Θ®16Ζ÷Θ©Θ®1Θ© Al2O3 2Ζ÷ Fe2O3 + 6H+ ==2Fe3+ + 3H2O 2Ζ÷
Θ®2Θ©3.2ΓΣ3.8 2Ζ÷ Θ®3Θ© SiO2 2Ζ÷ Na2SO4 Al2(SO4)3 MgSO4 3Ζ÷Θ§ΟΩΗω1Ζ÷
Θ®4Θ© ―Ί≤ΘΝßΑτœρ¬©ΕΖΘ®Ιΐ¬ΥΤςΘ©ΒΡ≥ΝΒμΦ”’τΝσΥ°÷Ν―ΆΟΜ≥ΝΒμΘ§Ψ≤÷Ο Ι“ΚΧεΉ‘»ΜΝς≥ωΘ§÷ΊΗ¥≤ΌΉς ΐ¥ΈΘ®3¥ΈΘ©÷Ν≥ΝΒμœ¥Β”Η…ΨΜΓΘ 2Ζ÷
Θ®5Θ©14m/15w 2Ζ÷
ΫβΈω ‘ΧβΖ÷ΈωΘΚΘ®1Θ©Al2O3ΈΣΝΫ–‘―θΜ·ΈοΘ§ΝρΥα‘ϋΒΡ≥…Ζ÷÷– τ”ΎΝΫ–‘―θΜ·ΈοΒΡ «Al2O3ΘΜFe2O3”κœΓΝρΥαΖ¥”Π…ζ≥…Fe2(SO4)3ΚΆH2OΘ§άκΉ”ΖΫ≥Χ ΫΈΣΘΚFe2O3 + 6H+ ==2Fe3+ + 3H2OΓΘ
Θ®2Θ©ΗυΨίΧβΡΩΥυΗχ±μ÷– ΐΨίΩ…“‘Ω¥≥ωΘ§Fe3+Άξ»Ϊ≥ΝΒμΕχΤδΥϋάκΉ”ΜΙΈ¥≥ΝΒμΒΡpHΖΕΈß «ΘΚ3.2ΓΣ3.8ΓΘ
Θ®3Θ©ΝρΥα‘ϋ÷–SiO2≤Μ”κH2SO4Ζ¥”ΠΘ§Υυ“‘¬Υ‘ϋAΒΡ÷ς“Σ≥…Ζ÷ΈΣSiO2ΘΜΦ”»κNaOHΒςΫΎpHΚσΘ§Fe3+ΉΣΜ·ΈΣFe(OH)3≥ΝΒμΘ§ΤδΥϋάκΉ”»‘ΈΣΝρΥα―ΈΘ§Υυ“‘¬Υ“ΚBΩ…“‘ΜΊ ’ΒΡΈο÷ ”–ΘΚ Na2SO4 Al2(SO4)3 MgSO4
Θ®4Θ©”Ο’τΝσΥ°œ¥Β”≥ΝΒμΒΡΖΫΖ®ΈΣΘΚ―Ί≤ΘΝßΑτœρ¬©ΕΖΘ®Ιΐ¬ΥΤςΘ©ΒΡ≥ΝΒμΦ”’τΝσΥ°÷Ν―ΆΟΜ≥ΝΒμΘ§Ψ≤÷Ο Ι“ΚΧεΉ‘»ΜΝς≥ωΘ§÷ΊΗ¥≤ΌΉς ΐ¥ΈΘ®3¥ΈΘ©÷Ν≥ΝΒμœ¥Β”Η…ΨΜΓΘ
Θ®5Θ©m kgΧζΚλΚ§Χζ‘ΣΥΊΘΚmΓΝ112/160, ‘≠ά¥ΝρΥα‘ϋ÷–Χζ‘ΣΥΊ÷ ΝΩΖ÷ ΐΈΣ: mΓΝ112/160Γ¬(1-25%)Γ¬w=14m/15wΓΘ
ΩΦΒψΘΚ±ΨΧβΩΦ≤ιΜ·―ßΙΛ“’Νς≥ΧΒΡΖ÷ΈωΓΔάκΉ”ΖΫ≥Χ ΫΒΡ ι–¥ΓΔ≥ΝΒμΒΡœ¥Β”ΓΔ÷ ΝΩΖ÷ ΐΒΡΦΤΥψΓΘ



| ΡξΦΕ | ΗΏ÷–ΩΈ≥Χ | ΡξΦΕ | ≥θ÷–ΩΈ≥Χ |
| ΗΏ“Μ | ΗΏ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ“Μ | ≥θ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏΕΰ | ΗΏΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θΕΰ | ≥θΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏ»ΐ | ΗΏ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ»ΐ | ≥θ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
ΚΘ―σ «“ΜΗωΖαΗΜΒΡΉ ‘¥±ΠΩβΘ§Ά®ΙΐΚΘΥ°ΒΡΉέΚœάϊ”ΟΩ…ΜώΒΟ–μΕύΈο÷ Ι©»Υάύ Ι”ΟΓΘ
Θ®1Θ© ΚΘΥ°÷–―ΈΒΡΩΣΖΔάϊ”ΟΘΚ
Δώ.ΚΘΥ°÷Τ―ΈΡΩ«Α“‘―ΈΧοΖ®ΈΣ÷ςΘ§Ϋ®―ΈΧο±Ί–κ―Γ‘Ύ‘ΕάκΫ≠Κ”»κΚΘΩΎΘ§ΕύΖγ…Ό”ξΘ§≥±œΪ¬δ≤ν¥σ«“”÷ΤΫΧΙΩ’ΩθΒΡΚΘΧ≤ΓΘΥυΫ®―ΈΧοΖ÷ΈΣ÷ϋΥ°≥ΊΓΔ’τΖΔ≥ΊΚΆ_______≥ΊΓΘ
II.ΡΩ«ΑΙΛ“Β…œ≤…”Ο±»Ϋœœ»ΫχΒΡάκΉ”ΫΜΜΜΡΛΒγΫβ≤έΖ®Ϋχ––¬»ΦνΙΛ“Β…ζ≤ζΘ§‘ΎΒγΫβ≤έ÷–―τάκΉ”ΫΜΜΜΡΛ÷Μ‘ –μ―τάκΉ”Ά®ΙΐΘ§Ήη÷Ι“θάκΉ”ΚΆΤχΧεΆ®ΙΐΘ§«κΥΒΟς¬»Φν…ζ≤ζ÷–―τάκΉ”ΫΜΜΜΡΛΒΡΉς”Ο____________________________________________ΓΘΘ®–¥“ΜΒψΦ¥Ω…Θ©
Θ®2Θ©Βγ…χΈωΖ® «ΫϋΡξά¥ΖΔ’ΙΤπά¥ΒΡ“Μ÷÷ΫœΚΟΒΡΚΘΥ°Β≠Μ·ΦΦ θΘ§Τδ‘≠άμ»γœ¬ΆΦΥυ ΨΓΘ«κΜΊ¥πΚσΟφΒΡΈ ΧβΘΚ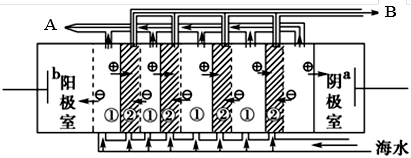
Δώ.ΚΘΥ°≤ΜΡή÷±Ϋ”Ά®»κΒΫΗΟΉΑ÷Ο÷–Θ§άμ”… «_____________________________________________ΓΘ
Δρ. BΩΎ≈≈≥ωΒΡ «________(ΧνΓΑΒ≠Υ°Γ±ΜρΓΑ≈®Υ°Γ±)ΓΘ
Θ®3Θ©”ΟΩύ¬±Θ®Κ§Na+ΓΔK+ΓΔMg2+ΓΔCl-ΓΔBr-Β»άκΉ”Θ©Ω…Χα»ΓδεΘ§Τδ…ζ≤ζΝς≥Χ»γœ¬ΘΚ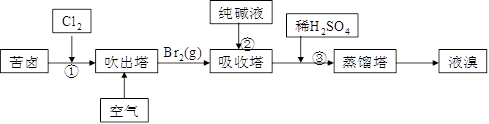
Δώ.»τΈϋ ’Υΰ÷–ΒΡ»ή“ΚΚ§BrO3Θ≠Θ§‘ρΈϋ ’Υΰ÷–Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣΘΏΘΏΘΏΘΏΘΏΘΏΘΏΘΏΘΏΘΏΘΏΘΏΘΏ
_________________________________________ΓΘ
Δρ.Ά®ΙΐΔΌ¬»Μ·“―ΜώΒΟΚ§Br2ΒΡ»ή“ΚΘ§ΈΣΚΈΜΙ–ηΨ≠Ιΐ¥Β≥ωΓΔΈϋ ’ΓΔΥαΜ·ά¥÷Ί–¬ΜώΒΟΚ§Br2ΒΡ»ή“ΚΘΩ_____________________________________________________________________ΓΘ
Δσ.œρ’τΝσΥΰ÷–Ά®»κΥ°’τΤχΦ”»»Θ§ΩΊ÷ΤΈ¬Ε»‘Ύ900CΉσ”“Ϋχ––’τΝσΒΡ‘≠“ρ «___________________________________________________________________________ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
ΖΑΦΑΜ·ΚœΈο”ΟΆΨΙψΖΚΓΘΙΛ“Β…œ≥Θ”ΟΚ§…ΌΝΩAl2O3ΒΡΖΑΧζΩσΘ®FeOΓΝV2O5Θ©Φν»έΖ®Χα»ΓV2O5ΓΘΦρ“ΣΝς≥Χ»γœ¬ΘΚ
“―÷ΣΘΚΔΌ±Κ…’ ±Ω…ΖΔ…ζΖ¥”ΠΘΚV2O5 + Al2O3+2Na2CO3 2NaVO3 +2NaAlO2 +2CO2
2NaVO3 +2NaAlO2 +2CO2
ΔΎ≥ΘΈ¬œ¬Έο÷ ΒΡ»ήΫβΕ»ΘΚNaVO3ΓΪ21Θ°2 g /100gΥ°ΘΜHVO3ΓΪ0Θ°008 g /100gΥ°
Θ®1Θ©ΓΑΫΰ≥ω‘ϋBΓ±ΒΡ÷ς“Σ≥…Ζ÷ « ΓΘΘ®–¥Μ·―ß ΫΘ©
Θ®2Θ©…ζ≤ζ÷–Θ§≤Μ÷±Ϋ””ΟH2SO4Ϋΰ≈ίΓΑ…’‘ϋAΓ±Μώ»ΓHVO3ΒΡ‘≠“ρ « ΓΘ
Θ®3Θ©ΓΑ≤ΌΉςΔΌΓ±Αϋά® ΓΔœ¥Β”ΓΘ»γΙϊ≤Μœ¥Β”Θ§‘ρ≤ζΤΖ÷–Ω…ΡήΚ§”–ΒΡΫπ τ―τάκΉ” « ΓΔ ΓΘœ¬Ν–ΉΑ÷ΟΘ®≤ΩΖ÷Φ–≥÷“«Τς Γ»ΞΘ©Ω…”Ο‘Ύ Β―ι “Ϋχ––ΓΑ≤ΌΉςΔΎΓ±ΒΡ « ΓΘΘ®Χν–ρΚ≈Θ©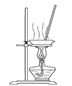

A B C D
Θ®4Θ©NaVO3”Ο”Ύ‘≠”ΆΒΡΆ―ΝρΦΦ θΘ§”…V2O5»ή”ΎNaOH»ή“Κ÷–÷Τ»ΓΘ§Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣ ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
Α¥ΆΦΥυ Ψ Β―ιΙΐ≥ΧΧνΩ’ΘΚ
Θ®1Θ©–¥≥ω…œ ωΖ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΘΚ
ΔΌ ΘΜ
ΔΎ ΘΜ
Δέ ΓΘ
Θ®2Θ©ΧνΩ’ΘΚa b c d ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
ΝυΥ°¬»Μ·ο»Θ®SrCl2ΓΛ6H2OΘ© « Β―ι “÷Ί“ΣΒΡΖ÷Έω ‘ΦΝΘ§ΙΛ“Β…œ≥Θ“‘Χλ«ύ ·Θ®÷ς≥…Ζ÷ΈΣSrSO4Θ©ΈΣ‘≠Νœ÷Τ±ΗΘ§…ζ≤ζΝς≥Χ»γœ¬ΘΚ
Θ®1Θ©ΒΎΔΌ≤ΫΖ¥”Π«ΑΧλ«ύ ·œ»―–ΡΞΖέΥιΘ§ΤδΡΩΒΡ «________________ΓΘ
ΒΎΔέ≤ΫΦ”»κ ΝΩœΓΝρΥαΒΡΡΩΒΡ «______ _ΓΘ
Θ®2Θ©ΒΎΔΌ≤ΫΖ¥”Π»τ0.5 mol SrSO4÷–÷Μ”–S±ΜΜΙ‘≠Θ§ΉΣ“ΤΝΥ4 molΒγΉ”ΓΘ–¥≥ωΗΟΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ________________________________________ΓΘ
Θ®3Θ©ΒΎΔή≤Ϋ≤ΌΉς“ά¥Έ «’τΖΔ≈®ΥθΓΔά以ΫαΨßΓΔΙΐ¬ΥΓΔ________ΓΔ_______ΓΘ
Θ®4Θ©≥Τ»Γ1.000 g≤ζΤΖ»ήΫβ”Ύ ΝΩΥ°÷–Θ§œρΤδ÷–Φ”»κΚ§AgNO3 1.100ΓΝ10Θ≠2 molΒΡAgNO3»ή“ΚΘ®≤ζΤΖ÷–≤ΜΚ§ΤδΥϋ”κAg+Ζ¥”ΠΒΡάκΉ”Θ©Θ§¥ΐCl®DΆξ»Ϊ≥ΝΒμΚσΘ§”ΟΚ§Fe3+ΒΡ»ή“ΚΉς÷Η ΨΦΝΘ§”Ο0.2000 mol/LΒΡKSCN±ξΉΦ»ή“ΚΒΈΕ® Θ”ύΒΡAgNO3Θ§ Ι Θ”ύΒΡAg+ “‘AgSCNΑΉ…Ϊ≥ΝΒμΒΡ–Έ ΫΈω≥ωΓΘ»τΒΈΕ®Ιΐ≥הϻ΅œ ω≈®Ε»ΒΡKSCN»ή“Κ20.00 mLΘ§‘ρ≤ζΤΖ÷–SrCl2ΓΛ6H2OΒΡ÷ ΝΩΑΌΖ÷Κ§ΝΩΈΣ________Θ®±ΘΝτ4ΈΜ”––ß ΐΉ÷Θ©ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
¥Φ”κ«β¬±ΥαΖ¥”Π «÷Τ±Η¬±¥ζΧΰΒΡ÷Ί“ΣΖΫΖ®ΓΘ Β―ι “÷Τ±Ηδε““ΆιΚΆ1-δεΕΓΆιΒΡΖ¥”Π»γœ¬ΘΚ ΔΌ NaBr+H2SO4 = HBr+NaHSO4ΘΜΔΎR-OH+HBr R-Br+H2O
R-Br+H2O
Ω…Ρή¥φ‘ΎΒΡΗ±Ζ¥”Π”–ΘΚ¥Φ‘Ύ≈®ΝρΥαΒΡ¥φ‘Ύœ¬Ά―Υ°…ζ≥…œ©ΚΆΟ―Θ§BrΘ≠±Μ≈®ΝρΥα―θΜ·ΈΣBr2Β»ΓΘ”–ΙΊ ΐΨίΝ–±μ»γœ¬
| | ““¥Φ | δε““Άι | ’ΐΕΓ¥Φ | 1-δεΕΓΆι |
| ΟήΕ»/gΓΛcm-3 | 0Θ°7893 | 1Θ°4604 | 0Θ°8098 | 1Θ°2758 |
| Ζ–Βψ/Γφ | 78Θ°5 | 38Θ°4 | 117Θ°2 | 101Θ°6 |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
Ρ≥ΙΛ≥ß≈≈≥ωΒΡΈέΥ°÷–Κ§”–¥σΝΩΒΡFe2ΘΪΓΔZn2ΘΪΓΔHg2ΘΪ»ΐ÷÷Ϋπ τάκΉ”ΓΘ“‘œ¬ «Ρ≥Μ·―ß―–ΨΩ–‘―ßœΑ–ΓΉιΒΡΆ§―ß…ηΦΤΒΡ≥ΐ»ΞΈέΥ°÷–ΒΡΫπ τάκΉ”Θ§ΜΊ ’¬ΧΖ·ΓΔπ©Ζ·(ZnSO4·7H2O)ΚΆΙ·ΒΡΖΫΑΗΓΘ
[“©ΤΖ]ΓΓNaOH»ή“ΚΓΔΝρΜ·ΡΤ»ή“ΚΓΔΝρΜ·―«ΧζΓΔœΓΝρΥαΓΔΧζΖέ
[ Β―ιΖΫΑΗ]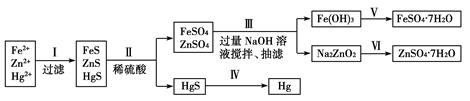
[Έ ΧβΧΫΨΩ]
Θ®1Θ©≤Ϋ÷ηΔρΥυΖΔ…ζΖ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣ_________________________________________________ΓΘ
Θ®2Θ©≤Ϋ÷ηΔσ÷–ΒΡ≥ι¬ΥΈΣΦθ―ΙΧθΦΰœ¬ΒΡΙΐ¬ΥΘ§Ω…“‘Φ”ΩλΙΐ¬ΥΥΌΕ»ΘΜΗΟ≤Ϋ÷η…φΦΑΖ¥”ΠΒΡάκΉ”ΖΫ≥Χ Ϋ”–Zn2ΘΪΘΪ4OHΘ≠=ZnO22-ΘΪ2H2OΚΆ________________ΓΘ
Θ®3Θ©≤Ϋ÷ηΔω÷–ΒΟΒΫΝρΥα–Ω»ή“ΚΒΡάκΉ”Ζ¥”ΠΖΫ≥Χ ΫΈΣ______________________________ΓΘ
Θ®4Θ©”ϊ Βœ÷≤Ϋ÷ηΔθΘ§Υυ–ηΦ”»κΒΡ ‘ΦΝ”–________ΓΔ________Θ§Υυ…φΦΑΒΡ÷ς“Σ≤ΌΉς“ά¥ΈΈΣ______________________ΓΘ
Θ®5Θ©≤Ϋ÷ηΔτ≥Θ”ΟΒΡΖΫΖ® «Φ”»»Θ§ΗΟ≤Ϋ÷η «ΖώΕ‘ΜΖΨ≥”–”ΑœλΘΩ__________(ΧνΓΑ «Γ±ΜρΓΑΖώΓ±)Θ§»γ”–”ΑœλΘ§«κΡψ…ηΦΤ“ΜΗωΜΖΨ≥±ΘΜΛΖΫΑΗά¥ Βœ÷≤Ϋ÷ηΔτΒΡΖ¥”Π________________________ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
Ή‘»ΜΫγ÷–ΒΡΈο÷ Εύ ΐ «“‘ΜλΚœΈοΒΡ–Έ Ϋ¥φ‘ΎΘ§œ¬Οφ «ΜλΚœΈοΒΡΖ÷άκΚΆΧα¥ΩΘ§«κΧνΩ’ΘΚ
Θ®1Θ©≥ΐ»ΞNaClΙΧΧε÷–Μλ”–ΒΡ…ΌΝΩCaCO3Θ§ΥυΫχ––ΒΡ Β―ι≤ΌΉςΈΣΘΚ ΓΔ ΓΔ’τΖΔΓΔΫαΨßΓΘ
Θ®2Θ©≥ΐ»ΞNaCl÷–ΒΡNa2SO4Θ§“ά¥ΈΦ”»κΒΡ»ή“ΚΈΣ(Χν»ή÷ Μ·―ß Ϋ)ΘΚ ΓΔ ΓΔ ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
Ρ≥ΩΤ―––ΓΉι“‘Ρ―»ή–‘ΦΊ≥Λ ·Θ®K2O?Al2O3?6SiO2)ΈΣ‘≠ΝœΘ§Χα»ΓA12O3ΓΔ K2CO3Β»Έο÷ Θ§ΙΛ“’Νς≥Χ»γœ¬ΘΚ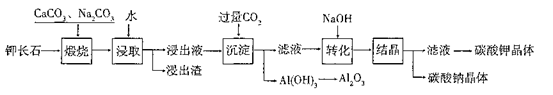
Θ®1Θ©ΓΑλ―…’Γ±Ιΐ≥Χ÷–”–»γœ¬Ζ¥”ΠΖΔ…ζΘΚ
ΔΌΦΊ≥Λ ·÷–ΒΡΙη‘ΣΥΊ‘ΎCaCO3Ής”Οœ¬ΉΣΜ·ΈΣCaSiO3Θ§–¥≥ωSiO2ΉΣΜ·ΈΣCaSiO3ΒΡΜ·―ßΖΫ≥Χ Ϋ_____________________ΓΘ
ΔΎΦΊ≥Λ ·÷–ΒΡΦΊ‘ΣΥΊΚΆ¬Ν‘ΣΥΊ‘ΎNa2CO3Ής”Οœ¬ΉΣΜ·ΈΣΩ…»ή–‘ΒΡNaAlO2ΚΆ KAlO2Θ§–¥≥ωAl2O3ΉΣΜ·ΈΣNaAlO2ΒΡΜ·―ßΖΫ≥Χ Ϋ____________________________ΓΘ
Θ®2Θ©“―÷ΣNaAlO2ΚΆKAlO2“ΉΖΔ…ζ»γœ¬Ζ¥”ΠΘΚAlO2Θ≠+2H2O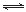 Al(OH)3+OHΘ≠ΓΘ
Al(OH)3+OHΘ≠ΓΘ
ΓΑΫΰ»ΓΓ± ±Θ§”Π±Θ≥÷»ή“Κ≥ ______–‘Θ®ΧνΓΑΥαΓ±ΜρΓΑΦνΓ±Θ©ΓΘΓΑΫΰ»ΓΓ± ±≤ΜΕœΫΝΑηΒΡΡΩΒΡ «______________ΓΘ
Θ®3Θ©ΓΑΉΣΜ·Γ± ±Φ”»κNaOHΒΡ÷ς“ΣΉς”Ο «_______ (”ΟάκΉ”ΖΫ≥Χ Ϋ±μ ΨΘ©ΓΘ
Θ®4Θ©…œ ωΙΛ“’÷–Ω…“‘―≠ΜΖάϊ”ΟΒΡ÷ς“ΣΈο÷ «_______ΓΔ_______ΚΆΥ°ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΙζΦ ―ß–Θ”≈―Γ - ΝΖœΑ≤αΝ–±μ - ‘ΧβΝ–±μ
Κΰ±± ΓΜΞΝΣΆχΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΤΫΧ® | Άχ…œ”–ΚΠ–≈œΔΨΌ±®Ή®«χ | Βγ–≈’©Τ≠ΨΌ±®Ή®«χ | …φάζ Ζ–ιΈό÷ς“ε”–ΚΠ–≈œΔΨΌ±®Ή®«χ | …φΤσ«÷»®ΨΌ±®Ή®«χ
ΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΒγΜΑΘΚ027-86699610 ΨΌ±®” œδΘΚ58377363@163.com