
ijУѧ��������ͼ��ʾװ�ý���ʵ�飬��̽�������巢����Ӧ��ԭ���������ᴿ��Ӧ�IJ��
��ش��������⣺
��1��д��II�з�Ӧ�Ļ�ѧ����ʽ ��
��2���۲쵽II�е������� ��
��3��ʵ�鿪ʼʱ���ر�K2������K1�ͷ�Һ©���������μӱ���Һ��Ļ��Һ����Ӧ��ʼ��III��С�Թ��ڱ��������� ��
��4����˵������Һ�巢����ȡ����Ӧ�������� ��
��5����������ƿ�ڷ�Ӧ���Һ�����ν�������ʵ������Ϳɵõ��ϴ������屽��
��������ˮϴ�ӣ�����Һ������5%��NaOH��Һϴ�ӣ�����Һ��
��������ˮϴ�ӣ�����Һ���ܼ�����ˮCaCl2��ĩ���
�� ����������ƣ���
 ��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
(15 ��)
������ͭ�Ǻϳ��������������в��ϡ�����������ͭ����Ҫǰ����֮һ������������һ��ʵ���Һϳ�·�ߣ�
�Ʊ��������װ��ʾ��ͼ����(���Ⱥͼг�װ�õ���)��
��֪����������۵�Ϊ76.5 �棬������ˮ�������Ҵ���
�ش��������⣺
��1����250 mL����ƿa�м���70 mL70%���ᡣ���ƴ�����ʱ����������ˮ��Ũ������Ⱥ�˳����
��
��2����a�е���Һ������100 �棬�����μ�40 g�����浽������Һ�У�Ȼ��������130 �������Ӧ����װ���У�����b�������� ������c�������� ���������� ��
��Ӧ�������������ˮ���ٷ�����������Ʒ��������ˮ��Ŀ���� �����������п����ڷ��뱽�����Ʒ���� (����)��
| A����Һ©�� | B��©�� | C���ձ� | D��ֱ��������E�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧ��ȤС����ѧϰ�������ε�ijЩ���ʺ���;���У�����������ʵ��̽����
��ʵ��һ������(NH4)2Fe(SO4)2��6H2O��Һ�к��е������ӡ�
��1�����±���д�����������ʵ������
��ѡ�Լ���������ˮ��1mol/LH2SO4��Һ��0.1mol/L KSCN��Һ��NaOHŨ��Һ��Ũ���ᡢ1mol/LNa2CO3��Һ
| �� �� �� �� | ʵ������ | �� �� |
| ����I��ȡ��������Һ���Թ��У� �� | | ��Һ�к���Fe2+ |
| ����II��ȡ��������Һ���Թ��У� �� | | ��Һ�к���NH4�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ˮ����ͭ��ǿ���»ᷢ���ֽⷴӦ��
CuSO4  CuO + SO3�� 2SO3
CuO + SO3�� 2SO3  2SO2��+ O2��
2SO2��+ O2��
����ͼ��ʾװ�ã��г���������ȥ��������D���ڷ�Ӧǰ��������������ֽ��˵���ˮ����ͭ��������
ʵ�鲽�裺
�ٳ�����ӦǰD�ܵ�������
�����Ӻ�װ�ã��ر�K������Ӳ�ʲ�����Aһ��ʱ���ֹͣ���ȡ�
�۴�Ӳ�ʲ�����A��ȴ��K��ͨ��һ��ʱ����ѳ�ȥ������̼����������Ŀ�����
���ٳ���D�ܣ����䷴Ӧǰ���������Ϊm��
�ش��������⣺
��1����Ӧ2SO3(g)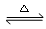 2SO2(g) + O2(g)��ƽ�ⳣ������ʽΪK= ��
2SO2(g) + O2(g)��ƽ�ⳣ������ʽΪK= ��
��2��B���г��¶����������⣬���ɿ����������� �����¶��������ߵ���Ҫԭ���� ��B���з�����Ӧ���й����ӷ���ʽ�� ��
��3������E�������� ��
��4������������ʵ�飬����B��C��D����������վ���ȫ�������Կ�����CO2��Ӱ�죬�ܷ����m������ֽ��˵���ˮCuSO4��������(��ѡ��һ�ش�)
������ܣ���ֽ����ˮCuSO4������Ϊ ����m��ʾ����
��������ܣ���ԭ���� ��Ϊ���ܲ�÷ֽ��˵���ˮ����ͭ����������ļ�ʵ�鷽���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
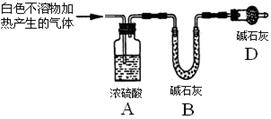
������ʾ��þ�뱥��̼��������Һ��Ӧ������������Ͱ�ɫ�����ijͬѧ���������ʵ�鷽������֤���̽����Ӧԭ����
��1���������
ʵ��I����ɰֽ��ȥþ����������Ĥ���������ʢ���������з�̪�ı���̼��������Һ���Թ��У�Ѹ�ٷ�Ӧ�������������ݺͰ�ɫ�������Һ��dz���졣
��ͬѧ�Է�Ӧ�в����İ�ɫ�������������²²⣺
�²�1����ɫ���������Ϊ ��
�²�2����ɫ���������ΪMgCO3��
�²�3����ɫ����������Ǽ�ʽ̼��þ[xMgCO3��yMg(OH)2]��
��2����ƶ���ʵ��ȷ�����ﲢ��֤�²⣺
| ʵ����� | ʵ�� | ʵ������ | ���� |
| ʵ��� | ��ʵ��I���ռ����������ȼ | �ܰ���ȼ�ա���������ɫ���� | ����ɷ�Ϊ _? _ |
| ʵ��� | ȡʵ��I�еİ�ɫ�����ϴ�ӣ� ��������? | ? | ��ɫ��������ܺ���MgCO3 |
| ʵ��� | ȡʵ��I�еij���Һ�������м�������CaCl2ϡ��Һ | ������ɫ���� | ��Һ�д��� �� ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��15�֣�ij��ȤС���ͬѧ���֣���CuSO4��Һ��Na2CO3��Һ��ϻ��������ɫ���������ǶԳ�������ɺܸ���Ȥ����������ʵ��ķ�������̽����
��������롿
����1�����ó���Ϊ ��
����2�����ó���Ϊ ��
����3�����ó���Ϊ��ʽ̼��ͭ[��ѧʽ�ɱ�ʾΪmCu(OH)2��nCuCO3]��
���������ϡ���������һ�ֳ������Ⱦ��ֽ⣨����������ᾧˮ����
��ʵ��̽����
����1������������Һ���ˣ���������ˮϴ�ӣ�������ˮ�Ҵ�ϴ�ӣ���ɣ�
����2��ȡһ�������ù��壬������װ�ã��г�����δ�������ж���ʵ�飻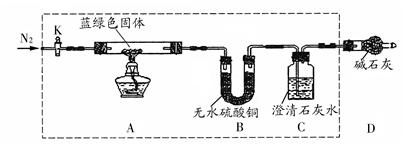
�������������ۡ�
��֤������1������ʵ�������� ��
��1������������߿���װ�������Ե�ʵ������ǣ��ر�K�� ��
��2����������װ��B��ʢ����ˮCaCl2��C��ʢ��Ba(OH)2��Һ�������Բⶨ���ó�������ɡ�
��C��ʢ��Ba(OH)2��Һ������ʹ�ó���ʯ��ˮ��ԭ���� ��
������ȡ����ɫ��������Ϊ27.1 g��ʵ�������װ��B����������2.7 g��C�в�������������Ϊ19.7 g ���������ɫ����Ļ�ѧʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
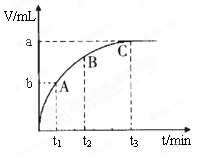
Ϊ���о���������Թ�������ֽ����ʵ�Ӱ�죬ijͬѧ��������ʵ�飬��ش��������⡣
| ��� | ���� | ʵ������ |
| �� | �ֱ����Թ�A��B�м���5 mL 5% H2O2��Һ��������2��1mol/L FeCl3��Һ�����Թ��о����������ݳ���ʱ�����Թ�A����ʢ��5��������ˮ���ձ��н��ݣ����Թ�B����ʢ��40��������ˮ���ձ��н��ݡ� | �Թ�A�в��ٲ������ݣ� �Թ�B�в��������������� |
| �� | ��ȡ��֧�Թֱܷ����5 mL 5% H2O2��Һ��5 mL 10% H2O2��Һ | �Թ�A��B�о�δ���Լ��������ݲ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���й��ڽ̲��еĸ���˵�������һ���ǣ� ��
| A���ڻ�ѧ��Ӧ�У���Ӧ��ת��Ϊ�������ͬʱ����Ȼ���������ı仯 |
| B�����ʰ����̬���ʡ���̬���ʺ����� |
| C������д�Ȼ�ѧ����ʽʱӦ������Ӧ�P�������״̬�����������Ӧ�¶Ⱥ�ѹǿ |
| D��ҽ�����õ�ʯ�������������ʯ����ˮ��ϳɺ�״��ܿ����̵����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����˵���У���ȷ����
| A�����ࡢ��֬�͵����ʶ�����������Ҫ��Ӫ���� |
| B��������������ͷ���ijЩ�����������ʵ����� |
| C����ѧ��Ӧ�ܹ�������µ����ʣ�ͬʱҲ��������µ�Ԫ�� |
| D��Ϊ����ʳ���ɫ���㡢ζ����ֹ���ʣ��������м������ʳƷ���Ӽ� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com