
����Ŀ������N��P��Fe��Ti��Ԫ�ص����Ͳ������Ź㷺����;��
(1)��̬Feԭ��δ�ɶԵ�����Ϊ______������̬Tiԭ�ӵļ۵����Ų�ͼ��_____________��
(2)�����������ѧ�ģ�FuNvio Cacace���˻���˼��������о������N4���ӣ����е�ԭ�ӵĹ���ӻ���ʽΪ__________________________��
(3)�Ƚ���̬�⻯���(PH3)�Ͱ�(NH3)�ļ��ǣ�PH3________NH3(����ڡ�����С�ڡ����ڡ�)����Ҫԭ��Ϊ____________________________________________________________��
(4)����Ľṹ����M�ܴ���ϩ����ϩ������ϩ�ľۺϣ���ṹ��ͼ��ʾ��
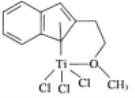
�����M��Ԫ���У��縺��������___________(������)��
��M���_________(����)��
A ���� B ����
C ���Ӽ� D ��λ��
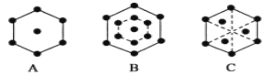
(5)��֪���ʯ�ľ���������Խ��ߴ�ֱ��ֽƽ���ϵ�ͶӰͼ����ͼB��ʾ�������������������Խ��ߴ�ֱ��ֽƽ���ϵ�ͶӰͼӦ����ͼ__________(����)��
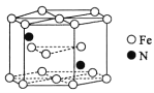
(6)ij�ִ��Ե������ľ����ṹ��ͼ��ʾ�����о�����ԭ���������ԭ�ӵĸ���Ϊ____________�������������ױ߳�Ϊa cm����Ϊc cm�������ִ��Ե������ľ����ܶ�Ϊ__________g��cm-3(�ú�a��c��NA�ļ���ʽ��ʾ)��
���𰸡�4 ![]() sp3 С�� �縺��Nǿ��P������ԭ�ӵĵ縺��Խ�ɼ����Ӷ�������ԭ��Խ�����ɼ����Ӷ�֮�����ԽС���ɼ����Ӷ�֮����ų��������DZ�� �� ABD A 12
sp3 С�� �縺��Nǿ��P������ԭ�ӵĵ縺��Խ�ɼ����Ӷ�������ԭ��Խ�����ɼ����Ӷ�֮�����ԽС���ɼ����Ӷ�֮����ų��������DZ�� �� ABD A 12 ![]()
��������
(1) FeΪ26��Ԫ�أ���̬Fe�ĺ�������Ų�ʽΪ1s22s22p63s23p63d64s2����̬ԭ��δ�ɶԵ�����Ϊ4����TiΪ22��Ԫ�أ���̬Ti�ĺ�������Ų�ʽΪ1s22s22p63s23p63d24s2�����Ի�̬Tiԭ�ӵļ۵����Ų�ͼ��![]() ���𰸣�4��
���𰸣�4��![]() ��
��
��2��N4���ӵĿռ乹����P4���ƣ�4��Nԭ���γ��������幹�ͣ�ÿ��Nԭ���γ�3��N-N����������1�Թµ��Ӷԣ��ӻ������ĿΪ4����Nԭ�Ӳ�ȡsp3�ӻ����𰸣�sp3��
(3) ��Ϊ�縺��Nǿ��P������ԭ�ӵĵ縺��Խ�ɼ����Ӷ�������ԭ��Խ�����ɼ����Ӷ�֮�����ԽС���ɼ����Ӷ�֮����ų��������DZ������PH3С��NH3���𰸣�С�ڣ��縺��Nǿ��P������ԭ�ӵĵ縺��Խ�ɼ����Ӷ�������ԭ��Խ�����ɼ����Ӷ�֮�����ԽС���ɼ����Ӷ�֮����ų��������DZ��
(4) �����M��Ԫ����Ti��C��H��O��Cl ������O�ķǽ�������ǿ���ǽ�����Խǿ�縺��Խ�����Ե縺�������������𰸣�����
��M����̼̼˫����̼̼������C��H����C��O���ȣ�����Ϊ![]() ���� ˫���к�1��
���� ˫���к�1��![]() ����1������������M�Ľṹ֪Ti��O֮������λ����û�����Ӽ�����ѡABD��
����1������������M�Ľṹ֪Ti��O֮������λ����û�����Ӽ�����ѡABD��
(5) )�ɽ��ʯ�ľ����ṹ��֪�����ʯ�ľ����൱��һ��������������ѻ�����һ��С�����������ѻ����������ľ���Ϊ���������ѻ������ݽ��ʯ�ľ���������Խ��ߴ�ֱ��ֽƽ���ϵ�ͶӰͼ֪���������ľ���������Խ��ߴ�ֱ��ֽ���ϵ�ͶӰͼΪA���𰸣�A��
(6) ���ݾ�̯�����ڵ����������У�����Nԭ����Ϊ2��Feԭ����Ϊ2��1/2+12��1/6+3=6�����Ե������Ļ�ѧʽFe3N��������ԭ���������ԭ�ӵĸ���Ϊ12���������ױ߳�Ϊacm����Ϊccm�����������![]() a2ccm3���������ִ��Ե������ľ����ܶ�Ϊ
a2ccm3���������ִ��Ե������ľ����ܶ�Ϊ![]() g��
g��![]() a2ccm3=
a2ccm3=![]() g/cm3���𰸣�12��
g/cm3���𰸣�12��![]() ��
��


 ��ѧ��������������Ͼ���ѧ������ϵ�д�
��ѧ��������������Ͼ���ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ɫ��ѧ����������������һ���¸����Ҫ����֮һ��ָ�Ӽ�������������ƿ��еĻ�ѧ��Ӧ�������ܼ��ٶԻ����ĸ����á����л�ѧ��Ӧ������ɫ��ѧ�������
A. ��CuSO4��Cu��2H2SO4(Ũ)![]() CuSO4��SO2����2H2O
CuSO4��SO2����2H2O
B. ��CuSO4��2Cu��O2![]() 2CuO��CuO��H2SO4(ϡ)��CuSO4��H2O
2CuO��CuO��H2SO4(ϡ)��CuSO4��H2O
C. ��Cu(NO3)2��3Cu��8HNO3(ϡ)��3Cu(NO3)2��2NO����4H2O
D. ��Cu(NO3)2��Cu��4HNO3(Ũ)��Cu(NO3)2��2NO2����2H2O
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��һ������̼����ȼ�ϵ��ԭ��ʾ����ͼ�������йظõ�ص�˵����ȷ���ǣ� ��

A. ��ӦCH4��H2O![]() 3H2��CO,ÿ����1molCH4ת��12mol ����
3H2��CO,ÿ����1molCH4ת��12mol ����
B. �缫A��H2����ĵ缫��ӦΪ��H2��2OH����2e��=2H2O
C. ��ع���ʱ��CO32����缫B�ƶ�
D. �缫B�Ϸ����ĵ缫��ӦΪ��O2��2CO2��4e��=2CO32��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ���Ҵ����ϴ���FeBr3��Һ���õ���ı���Һ����ˮFeCl3����������ܴﵽ��Ӧʵ��Ŀ�ĵ���
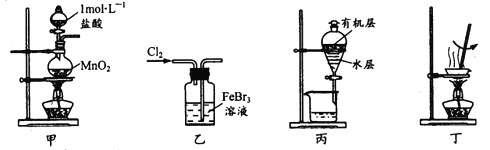
A.��װ�ü���ȡ����
B.��װ����ʹBr��ȫ��ת��Ϊ�嵥��
C.��װ�ñ���Һʱ�ȴ��¿ڷų�ˮ�㣬�ٴ��Ͽڵ����л���
D.��װ�ö�����Һ���ˮ���������ɣ��������Ƶ���ˮFeCl3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͷ���ؾ��������Ŀ������ã���ϳ�·����ͼ��ʾ��
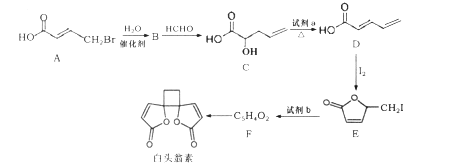
��֪��
��RCH2Br![]() RCH=CHR��
RCH=CHR��
��2RCH=CHR��![]()
������R��R�������⡢�����
(1)��ͷ���صķ���ʽΪ____��
(2)�Լ�aΪ______��E��F�ķ�Ӧ����Ϊ________��
(3)F�Ľṹ��ʽΪ_________��
(4)C�к��еĹ���������Ϊ________��
(5) A��B��Ӧ�Ļ�ѧ����ʽΪ_________��
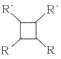
(6)F�����������ӳɵõ�G��G�ж���ͬ���칹�壬����������״��������____�֡�
(7)����ϩΪ��ʼԭ�ϣ�ѡ�ñ�Ҫ�����Լ��ϳ� ��·��Ϊ____���ýṹ��ʽ��ʾ�л���ü�ͷ��ʾת����ϵ����ͷ��ע���Լ��ͷ�Ӧ��������
��·��Ϊ____���ýṹ��ʽ��ʾ�л���ü�ͷ��ʾת����ϵ����ͷ��ע���Լ��ͷ�Ӧ��������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������(NaNO2)��һ�ֳ��õ�ʳƷ���Ӽ���ʹ��ʱ���ϸ����������ʵ������2NO+Na2O2=2NaNO2Ϊԭ������������װ����ȡNaNO2(�гֺͼ���������)��
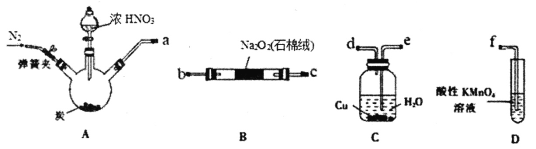
��֪��������KMnO4��Һ�ɽ�NO��NO2-����ΪNO3-��MnO4-����ԭΪMn2+��
��HNO2���в��ȶ��ԣ�2HNO2=NO2��+NO��+H2O��
�ش��������⣺
(1)�������������������ӿ�______________(��ӿ���ĸ)��
(2)ʵ�������Cװ���ڹ۲쵽��������___________________________��
(3)Na2O2��ַ�Ӧ�ⶨNaNO2��������ȡ��Ӧ��B�й�����Ʒ3.45g��������ˮ����ȴ����0.50mol�� L-1����KMnO4��Һ�ζ����ظ����Σ���Һƽ������Ϊ20.00mL��
�ٸòⶨʵ����Ҫ�õ����������е�___________(�����)��
a����ƿ b������ƿ c����ͷ�ι� d����ʽ�ζ��� e����ʽ�ζ��� f��������
�ڼٶ��������ʲ���KMnO4��Ӧ���������Ʒ��NaNO2�Ĵ���Ϊ____����
��ʵ��õ�NaNO2�ĺ�������ƫ�ͣ�������Ʒ�к��е���Ҫ����Ϊ_____(�ѧʽ)��Ϊ��߲�Ʒ��������ʵ��װ�õĸĽ�����Bװ��֮ǰ��װʢ��_____(��ҩƷ����)��______(����������)��
(4)���һ��ʵ�鷽��֤������������NaNO2����������________________________��
(�ṩ���Լ���0.10mol��L-1NaNO2��Һ��KMnO4��Һ��0.10mol��L-1KI��Һ��������Һ��ϡ���ᡢϡ����)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������ͼ��ʾװ�õ�����,�������( )

A. п�Ǹ�������������СB. ��������ͭ���汻��ԭ����������
C. ���Ӵ�пƬ��������Һ����ͭƬD. ���Ӵ�пƬ����������ͭƬ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ������Ũ��������1.0mol/L������Һ480mL���ش��������⣺
��1����ͼ��ʾ��������������Һ�϶�����Ҫ���� ______������ĸ��������������Һ�����õ��IJ���������_________�����������ƣ���
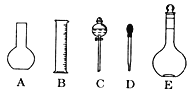
��2������ƿ�ϱ�������5���е�_____������ţ�
��ѹǿ ���¶� ������ ��Ũ�� �ݿ̶���
��3�������ƹ����У����в���ʹ������ҺŨ��ƫ�����__________ .
��δ��ϡ�ͺ��H2SO4��Һ��ȴ�����¾�ת�Ƶ�����ƿ��
�ڶ���ҡ�Ⱥ���Һ����ڿ̶��ߣ����ý�ͷ�ιܼ�����ˮ���̶���
�۶���ʱ�����ӿ̶���
��ʹ������ƿǰ������ˮϴ����û����
��4������ʵ��������������Һ������Ͳ��ȡ��������Ϊ98%���ܶ�Ϊ1.84g/mL��Ũ��������Ϊ��_____mL��������С�����һλ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��I�����Ǽ���ʵ���г��õ�������
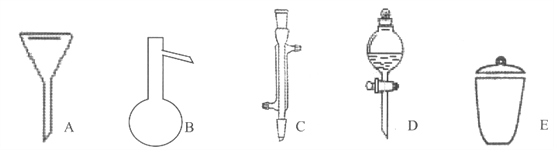
д����������������������ƣ�
A__________��B__________��C__________��D__________��E__________
IIʵ����Ҫ����100 mL 2 mol��L NaCl��Һ����ش��������⣺
��1�����ƹ�������Ҫʹ�õ���Ҫ�������������ձ�������������ͷ�ιܡ���ƽ����Ͳ��__________________��
��2����������ƽ��ȡ�Ȼ��ƹ��壬������Ϊ__________g��
��3��������Ҫ�����������ȷ˳����____________________������ţ���
�ٳ�ȡһ���������Ȼ��ƣ������ձ��У�����������ˮ�ܽ⣻
�ڼ�ˮ��Һ��������ƿ���̶�����1��2����ʱ�����ý�ͷ�ιܵμ�����ˮ����Һ����̶������У�
�۽���Һת�Ƶ�����ƿ�У�
�ܸǺ�ƿ�����������µߵ���ҡ�ȣ�
������������ˮϴ���ձ��ڱںͲ�����2��3�Σ�ϴ��Һת�Ƶ�����ƿ�С�
��4�����ʵ�������ȱ�ٲ���ݣ������������Һ�����ʵ���Ũ��__________ ���ƫ�ߡ���ƫ�͡�����Ӱ�족����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com