
��������һ����Ҫ�����ȼ�ϣ�Ҳ�������������������ȣ��Գ��������ƻ����á���ҵ�Ͽ�����ú���������ˮú�����ϳɶ����ѡ���ش��������⣺
�� ú����������Ҫ��ѧ��Ӧ����ʽΪ��___________________________��
�� ����ˮú���ϳɶ����ѵ�������Ӧ���£�
�� 2H2(g) + CO(g)![]() CH3OH(g) ��H����90.8 kJ��mol��1 K1
CH3OH(g) ��H����90.8 kJ��mol��1 K1
�� 2CH3OH(g)![]() CH3OCH3(g) + H2O(g)����H����23.5 kJ��mol��1 K2
CH3OCH3(g) + H2O(g)����H����23.5 kJ��mol��1 K2
�� CO(g) + H2O(g)![]() CO2(g) + H2(g)����H����41.3 kJ��mol��1 K3
CO2(g) + H2(g)����H����41.3 kJ��mol��1 K3
�ܷ�Ӧ��3H2(g) + 3CO(g)![]() CH3OCH3(g) +CO2 (g)����H�� _____ , K= ���ð���K1�� K2�� K3��ʽ�ӱ�ʾ��.һ�������µĵ����ܱ������У����ܷ�Ӧ�ﵽƽ�⣬Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ�� ������ĸ���ţ���
CH3OCH3(g) +CO2 (g)����H�� _____ , K= ���ð���K1�� K2�� K3��ʽ�ӱ�ʾ��.һ�������µĵ����ܱ������У����ܷ�Ӧ�ﵽƽ�⣬Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ�� ������ĸ���ţ���
a�����¸�ѹ b��������� c����CO2��Ũ��
d������CO��Ũ�� e������H2��Ũ�� f������He
��3����֪��Ӧ��2CH3OH(g) ![]() CH3OCH3(g) + H2O(g)ij�¶��µ�ƽ�ⳣ��Ϊ400 �����¶��£����ܱ������м���CH3OH ����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
CH3OCH3(g) + H2O(g)ij�¶��µ�ƽ�ⳣ��Ϊ400 �����¶��£����ܱ������м���CH3OH ����Ӧ��ijʱ�̲�ø���ֵ�Ũ�����£�
| ���� | CH3OH | CH3OCH3 | H2O |
| Ũ��/��mol��L��1�� | 0.44 | 0.6 | 0.6 |
�� �Ƚϴ�ʱ�����淴Ӧ���ʵĴ�С��v������ ______ v���棩 ���>������<������)��
�� ������CH3OH��10 min��Ӧ�ﵽƽ�⣬��ʱc(CH3OH) �� _________����ʱ���ڷ�Ӧ����v(CH3OH) �� __________��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
| ||
| ���� | CH3OH | CH3OCH3 | H2O |
| Ũ��/��mol?L-1�� | 0.44 | 0.6 | 0.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2012?�������ģ����������һ����Ҫ�����ȼ�ϣ�Ҳ�������������������ȣ��Գ��������ƻ����ã���ҵ������H2��CO2�ϳɶ����ѵķ�Ӧ���£�6H2��g��+2CO2��g��?CH3OCH3��g��+3H2O��g��
��2012?�������ģ����������һ����Ҫ�����ȼ�ϣ�Ҳ�������������������ȣ��Գ��������ƻ����ã���ҵ������H2��CO2�ϳɶ����ѵķ�Ӧ���£�6H2��g��+2CO2��g��?CH3OCH3��g��+3H2O��g��| 0.03 |
| t |
| 0.03 |
| t |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ||
| ||
| ���� | CH3OH | CH3OCH3 | H2O |
| Ũ��/��mol?L-1�� | 0.44 | 0.6 | 0.6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
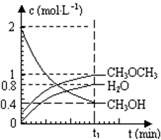 ��֪��������һ����Ҫ�����ȼ�ϣ�����ͨ��CH3OH���Ӽ���ˮ�Ƶã�2CH3OH��g��?CH3OCH3��g��+H2O��g����H=23.5kJ?mol-1����T1�棬�����ܱ������н�������ƽ�⣬��ϵ�и����Ũ����ʱ��仯��ͼ��ʾ����ش��������⣺
��֪��������һ����Ҫ�����ȼ�ϣ�����ͨ��CH3OH���Ӽ���ˮ�Ƶã�2CH3OH��g��?CH3OCH3��g��+H2O��g����H=23.5kJ?mol-1����T1�棬�����ܱ������н�������ƽ�⣬��ϵ�и����Ũ����ʱ��仯��ͼ��ʾ����ش��������⣺| c(CH3OCH3)?c(H2O) |
| c2(CH3OH) |
| c(CH3OCH3)?c(H2O) |
| c2(CH3OH) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ��ѧ�� | C-H | O-H | H-H | C-O | C��O |
| ����/kJ��mol-1 | 413 | 463 | 436 | 358 | 1072 |
| ���� | CH3OH ��g�� | CH3OCH3 ��g�� | H2O ��g�� |
| ��ʼŨ��/mol��l-1 | 2 | 0.2 | 0 |
| ƽ��Ũ��/mol��l-1 | 0.4 | 1 | 0.8 |
| ���� | CH3OH ��g�� | CH3OCH3 ��g�� | H2O ��g�� |
| ��ʼŨ��/mol��l-1 | 0.4 | 1.2 | 0.6 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com