
| ���� | CH3COOH | H2SO3 | HCN |
| ���ʵ���Ũ��mol/L | 0.01 | 0.01 | 0.01 |
| pH | 3.4 | 2.3 | 5.6 |
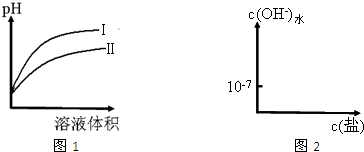
| c(H+) |
| c(CH3COOH) |
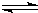



| c(H+) |
| c(CH3COOH) |
| c(H+) |
| c(CH3COOH) |
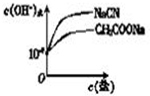
| 1 |
| Ka |
| 0.01 |
| 2.5��10-6��2.5��10-6 |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| ������ | Ksp |
| Ca3��AsO4��2 | 6.8��10-19 |
| CaSO4 | 9.1��10-6 |
| FeAsO4 | 5.7��10-21 |
| ��Ⱦ�� | H2SO4 | As |
| ��ˮŨ�� | 29.4g/L | 1.6g?L-1 |
| �ŷű� | pH 6��9 | 0.5mg?L-1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��㶫ʡ�����и����ڶ��θ߿�ģ�⿼�����ۻ�ѧ�Ծ��������棩 ���ͣ������
ij���Ṥ�������Է�ˮ���飨As��Ԫ�غ������ߣ�Ϊ��������ŷţ����û�ѧ���������������ˮ�������������£�

����������±�����ش��������⣺
��1�������ε�Ksp
|
������ |
Ksp |
|
Ca3(AsO4)2 |
6.8��10��19 |
|
CaSO4 |
9.1��10��6 |
|
FeAsO4 |
5.7��10��21 |
��2��������Ⱦ���ŷ�Ũ�ȼ������ŷű�
|
��Ⱦ�� |
H2SO4 |
As |
|
��ˮŨ�� |
29.4g/L |
1.6g��L��1 |
|
�ŷű� |
pH 6��9 |
0.5mg��L��1 |
��1�������Ṥ���ŷŵķ�ˮ����������ʵ���Ũ��c(H2SO4)= mol��L��1��
��2�������Է�ˮ��Fe3+��Ũ��Ϊ1.0��10��4mol��L��1��c(AsO43��)= mol��L��1��
��3�������ŷų������Է�ˮ�е������飨H3AsO3���ᣩ���׳�������Ͷ��MnO2�Ƚ�������������飨H3AsO4���ᣩ��MnO2����ԭΪMn2������Ӧ�����ӷ���ʽΪ ��
��4���ڴ��������ˮʱ���÷ֶ�ʽ�������ˮ��Ͷ����ʯ�ҵ���pH��2����Ͷ����ʯ�ҽ�pH���ڵ�8����ʹ�������Ca3(AsO4)2��ʽ������
�ٽ�pH���ڵ�2ʱ��ˮ���д�������������������Ҫ�ɷֵĻ�ѧʽΪ ��
��Ca3(AsO4)2��pH���ڵ�8���Ҳſ�ʼ������ԭ��Ϊ
��
�����ᣨH3AsO4���ֲ������ƽ�ⳣ��(25��)Ϊ��K1=5.6��10��3 K2=1.7��10��7 K3=4.0��10��12�������������ƽ�ⳣ���ı���ʽΪK3=
��Na3AsO4�ĵ�һ��ˮ������ӷ���ʽΪ��AsO43��+H2O HAsO42��+OH�����ò�ˮ���ƽ�ⳣ����25�棩Ϊ��
��������λ��Ч���֣���
HAsO42��+OH�����ò�ˮ���ƽ�ⳣ����25�棩Ϊ��
��������λ��Ч���֣���
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com