
����Ŀ����ѧ�һ���̽���¼�����CO2�����ۺ����á�
��CO2����FeO����ͬʱ���H2��
i. 6FeO(s)+CO2(g)=2Fe3O4(s)+C(s) ��Hl=��76.0 kJ��mol��1
��. C(s)+2H2O(g)=CO2(g)+2H2(g) ��H2= +113.4 kJ��mol��1
��1����3FeO(s)+H2O(g)=Fe3O4(s)+H2(g) ��H3=_________��
��CO2����������ȼ�ϼ״���
CO2(g)+3H2(g)![]() CH3OH(g)+H2O(g)����H����49.0 kJ��mol��1�������Ϊ1L�ĺ����ܱ������У�����1 mol CO2��3 mol H2��һ�������·�����Ӧ�����CO2��CH3OH(g)��Ũ����ʱ��仯��ͼ��ʾ��
CH3OH(g)+H2O(g)����H����49.0 kJ��mol��1�������Ϊ1L�ĺ����ܱ������У�����1 mol CO2��3 mol H2��һ�������·�����Ӧ�����CO2��CH3OH(g)��Ũ����ʱ��仯��ͼ��ʾ��

��2���ӷ�Ӧ��ʼ��ƽ�⣬������ƽ����Ӧ����v(H2)��_________ mol��(L��min) ��1��
��3��������ת���ʣ�________________________��
��4���÷�Ӧ��ƽ�ⳣ��Ϊ______________________(����С�����2λ)��
��5�����д�ʩ����ʹƽ����ϵ��n(CH3OH)/n(CO2)�������_________________��
A�������¶� B������He(g)��ʹ��ϵѹǿ����
C���ٳ���1 mol H2 D����H2O(g)����ϵ�з����ȥ
��6������Ӧ�ﵽƽ��ʱ��CH3OH���������Ϊm1��Ȼ�����������ټ���һ����H2������Ӧ��һ�δﵽƽ���CH3OH���������Ϊm2����m1________m2�Ĺ�ϵ(�����������������)��
���𰸡�+18.7 kJ��mol��1 0.22575%5.33CD��
��������
��1���ɸ�˹���ɿɵ���H3=����H1+��H2����2=����76.0 kJ��mol��1+113.4 kJ��mol��1��=+18.7 kJ��mol��1���ʴ�Ϊ��+18.7 kJ��mol��1����2����ͼ��֪����Ӧ��10min�ﵽƽ�⣬��ʱCO2��Ũ�ȱ仯1mol/L-0.25mol/L=0.75mol/L����ӦH2��Ũ�ȱ仯Ϊ0.75 mol/L��3=2.25 mol/L����v(H2)��2.25mol/L��10min=0.225 mol��(L��min) ��1���ʴ�Ϊ��0.225 mol��(L��min) ��1����3��������ת���ʣ���2.25mol��3mol����100%=75%���ʴ�Ϊ��75%����4��������ƽ��״̬�¸����ʵ�Ũ�ȷֱ�Ϊ��c(CO2)=0.25 mol/L��c(H2)=0.75 mol/L��c(CH3OH)=0.75 mol/L��c(H2O)=0.75 mol/L����ƽ�ⳣ��K=![]() =5.33���ʴ�Ϊ��5.33�� ��5��A�������¶ȣ��÷�Ӧƽ�������ƶ���n(CH3OH)/n(CO2)��С��B������He(g)��ʹ��ϵѹǿ����ƽ�ⲻ�ƶ���n(CH3OH)/n(CO2)���䣬C���ٳ���1 mol H2��ƽ�������ƶ���n(CH3OH)/n(CO2)����D����H2O(g)����ϵ�з����ȥ��ƽ�������ƶ���n(CH3OH)/n(CO2)���ʴ�Ϊ��CD����6�����������ټ���һ����H2����Ȼƽ��������Ӧ�����ƶ�������Ӧ������ܵķ�����Ŀ���ӣ�����CH3OH����������½�����m1>m2��
=5.33���ʴ�Ϊ��5.33�� ��5��A�������¶ȣ��÷�Ӧƽ�������ƶ���n(CH3OH)/n(CO2)��С��B������He(g)��ʹ��ϵѹǿ����ƽ�ⲻ�ƶ���n(CH3OH)/n(CO2)���䣬C���ٳ���1 mol H2��ƽ�������ƶ���n(CH3OH)/n(CO2)����D����H2O(g)����ϵ�з����ȥ��ƽ�������ƶ���n(CH3OH)/n(CO2)���ʴ�Ϊ��CD����6�����������ټ���һ����H2����Ȼƽ��������Ӧ�����ƶ�������Ӧ������ܵķ�����Ŀ���ӣ�����CH3OH����������½�����m1>m2��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����NA��ʾ�����ӵ�������ֵ�������ж���ȷ����
A. �����£�pH��1��������Һ�У���ˮ�����H������Ϊ10��13NA
B. ��״���£���ԭ������Ϊ0.2NA��CO2��NO2�Ļ�����壬�����Ϊ2.24L
C. �����£�5.6g���ֱ�������������Ũ���ᷴӦ��ת�Ƶĵ�����Ŀ��Ϊ0.3NA
D. 100g46%�������������Ҵ���Һ������O��H��ĿΪ7NA
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����������У�ʹ��ˮ�����Ը��������Һ������ɫ����(����)
A. ��ϩ B. �� C. ���� D. ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪A��BΪ���ʣ�CΪ�������ʵ������ת����ϵ���ǣ� ��
![]()
����C����ˮ��õ�ǿ����Һ����A������Na
����C��Һ��Na2CO3�ų�CO2���壬��A������H2
����C��Һ�еμ�KSCN��Һ�Ժ�ɫ����B����ΪFe
����C��Һ�еμ�NaOH��Һ����ɫ�������ɣ���B����ΪCu
A. �٢� B. �٢� C. �ڢ� D. �ۢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����з�Ӧ�������ȷ�Ӧ����( )
A. C6H12O6(������aq)��6O2![]() 6CO2��6H2O
6CO2��6H2O
B. ����������Һ��������кͷ�Ӧ
C. ��Ӧ��������������������������
D. �ƻ���Ӧ��ȫ����ѧ���������������ƻ�������ȫ����ѧ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����и���ʵ��������������ó��Ľ�����ȷ����
ѡ�� | ʵ����� | ʵ������ | �ᡡ�� |
A | �ýྻ�IJ�˿պȡijʳ�������ھƾ��ƻ��������� | �����Ի�ɫ | ��ʳ���в�����KIO3 |
B | ��SO2����ͨ�뵽Ba(NO3)2��Һ�� | ���ɰ�ɫ���� | �˳�����BaSO3 |
C | ȡ���õ�Na2O2��ĩ�������еμӹ��������� | ������ɫ���� | Na2O2û�б��� |
D | ��CuSO4��Һ�м���KI��Һ���ټ��뱽�������� | �а�ɫ�������ɣ��������ɫ | ��ɫ��������ΪCuI |
A. A B. B C. C D. D
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪����A�IJ���ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ����2CH3CHO��O2![]() 2CH3COOH������AΪ��Ҫԭ�Ϻϳɻ�����E����ϳ�·����ͼ1��ʾ���ش��������⣺
2CH3COOH������AΪ��Ҫԭ�Ϻϳɻ�����E����ϳ�·����ͼ1��ʾ���ش��������⣺
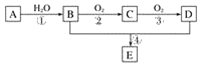
ͼ1
(1)д���������ʵĹ��������ƣ�
B��____________________��D��____________________��
(2)��Ӧ�ܵĻ�ѧ����ʽΪ________________________________________________����Ӧ���ͣ�________��
(3)ijѧϰС���������B��������ʵ��װ�����£�����ͼ2װ�ûش����⡣
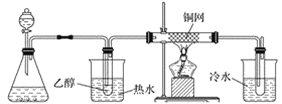
�� �� �� ��
ͼ2
��װ�ü���ƿ��ʢ�ŵĹ���ҩƷ����Ϊ________(����ĸ)��
A Na2O2 B KCl C Na2CO3 D MnO2
��ʵ������У���װ��Ӳ�ʲ������з�����Ӧ�Ļ�ѧ����ʽΪ_________________________��
������B�Ĵ����������������Ǿ�����ͬ��������Ӧ�������õ���������μӵ�����������ͭ����Һ�м��ȣ�����Ϊ______________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ijʳƷ���������Ҫ�ɷ��������ƣ������ƣ�CaO��Ӧ���ڣ�������
A. �� B. �� C. �� D. ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��(1)�������л��������CH4����CH2=CH2����CH3CH2OH��CH3COOH��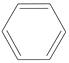 �У�������ҽ������������_________(����ţ���ͬ)������Ϊˮ�����������_________����ú��������пɻ�õ�Һ̬����ԭ����_________�������ˮ����ˮ������_________���������ȼ�ϵ���_________��
�У�������ҽ������������_________(����ţ���ͬ)������Ϊˮ�����������_________����ú��������пɻ�õ�Һ̬����ԭ����_________�������ˮ����ˮ������_________���������ȼ�ϵ���_________��
(2)��ʯȼ�ϰ���________��__________��_________������ϳɲ��ϰ���_____��_______��______��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com