
�ϳɰ���Ӧ��N2+3H2 2NH3+92.4KJ���ڻ�ѧ��ҵ������ҵ������Ҫ���塣��ҵ�ϳɰ�����ʾ��ͼ��ͼ��ʾ��
2NH3+92.4KJ���ڻ�ѧ��ҵ������ҵ������Ҫ���塣��ҵ�ϳɰ�����ʾ��ͼ��ͼ��ʾ��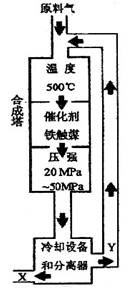
30. X�Ļ�ѧʽΪ ��X��״̬Ϊ_______̬��
31. ����ͼ����������˵����ȷ���� ����д��ţ���
a���ڴ��¶��£�������ƽ�������ƶ�������߰��IJ���
b������ý��ʹ��������ƽ�������ƶ�
c����ҵ�����ܶ��������ϡ��豸�����������ƣ�ѡ���ѹǿ
d��Ϊ���ԭ�ϵ�ת���ʣ�����ѭ������
32. �����°�����������ˮ����ˮ��Һ���Ե��硣
�� ��ˮ��ˮ�����ӻ���������ֵ��_______________________��
�� ����ͬ�������ͬ���ʵ���Ũ�ȵİ�ˮ�������Ϻ�
��Һ������Ũ���ɴ�С����Ϊ ��
30. NH3��Һ ��2�֣�
31. c��d��2�֡�ѡ��1����1�֣���ѡ����ѡ���÷֣�
32. �� 10-14 �� c(Cl��)>c(NH4+)>c(H+)>c(OH��) ��2�֣�
�������������30.��ΪNH3����ʱ��ΪҺ�壬�����²���������X��NH3ΪҺ̬��
31.a�����ڷ�Ӧ������ȣ���500���£����²�����ƽ�������ƶ�������ȷ��b������ýֻ�ӿ췴Ӧ���ʣ����ı�ƽ�⣬˵������ȷ��c����ҵ�����ܶ��������ϡ��豸�����������ƣ�ѡ���ѹǿ����ȷ��d��Ϊ���ԭ�ϵ�ת���ʣ�����ѭ����������ȷ��
32.�ڽ���ͬ�������ͬ���ʵ���Ũ�ȵİ�ˮ�������Ϻ�����NH4Cl��NHˮ�������ԣ�����Һ������Ũ���ɴ�С����Ϊc(Cl��)>c(NH4+)>c(H+)>c(OH��)��
���㣺�ϳɰ�ԭ����NH4Cl��Һ�е�����Ũ�ȴ�С�Ƚϡ�


 Ӧ�����������Ĵ���ѧ������ϵ�д�
Ӧ�����������Ĵ���ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
19����ĩ������Ϊ��ϡ���Ĺ���������۸����½���������Ϊ������ ( )
| A������������ | B�������������ȡ���ķ��� |
| C���ý����ƻ�ԭ�������ķ��� | D����ˮ�Ȼ��������ȡ���ķ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
ұ������һ�����������ַ������ٽ�̿������ˮú������H2��CO�������ۻ��ý����û������ܵ�ⷨ�����ַ����ڹ�ҵ�Ͼ���Ӧ�á��Ŵ��У����տ�ȸʯ����ͭ������ʪ����ͭ���ִ��У������ȷ��������������ӹ�±ʯ��KCl?MgCl2?6H2O������þ�������ǵ�ұ��������������ȷ����
| A�������� �� | B�������� �� |
| C�������� �ۡ� | D���������� �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧ��ѡ��2����ѧ�뼼������15�֣�
��ѧ����������Ĺؼ�����ѧΪ����������������ṩ�����ʱ�֤��
���Ļ���������Ҫ�Ļ�����Ʒ������������Ҳ���Ľ��У�������ѧ�Ҿ���Ϊ���������������ܺ������������̽����
��1��25��ʱ�ϳɰ���Ӧ�Ȼ�ѧ����ʽΪ��N2(g)+3H2(g)==2NH3(g)����H="-92.4kJ/mol" ���ڸ��¶�ʱ��ȡ1molN2��3molH2�����ܱ������У��ڴ��������½��з�Ӧ����÷�Ӧ�ų�����������С��92.4kJ����ԭ����_________________________________��
��2���������˽���ų�ֱ�Ӽ��ڵ�����������Ӧ�������ڣ��ڽϵ͵��¶Ⱥ�ѹǿ�����ºϳɰ�������˽ϺõIJ��ʡ��ӻ�ѧ��Ӧ���ʽǶȷ�������ų��Ժϳɰ���Ӧ�������� ���봫ͳ�ĺϳɰ��ķ����Ƚϣ��÷������ŵ��� ��
��(3)±ˮ���̺��ŷḻ��þ��Դ����ת����ɻ��MgCl2�ֲ�Ʒ����±ˮ����ȡþ�IJ���Ϊ��
a�������ߴ������ڵı������ճ�ʯ�ң�����ʯ���Ƴ�ʯ���飻
b����ʯ������뵽��ˮ�������о����˵õ�Mg(OH)2������
c.��Mg(OH)2�����м�������õ�MgCl2��Һ���پ������ᾧ�õ�MgCl2��6H2O��
d����MgCl2��6H2O��һ�������¼��ȵõ���ˮMgCl2��
e��������ڵ��Ȼ�þ�ɵõ�Mg��
�ٲ���d�еġ�һ��������ָ���� ��
����ͬѧ��Ϊ������b��ɼ���Mg(OH)2�õ�MgO���ٵ�����ڵ�MgO�ƽ���þ�������ɼ�ʵ�鲽�裬��ͬ���ͬѧ���뷨��?Ϊʲô?
(4) ���Ǻ˷�Ӧ����Ҫ��ȼ�ϣ��Ѿ����Ƴɹ�һ���������ӽ�����֬����ר��������ˮ�� ��U4+��������������Ԫ�ء��䷴Ӧԭ��Ϊ (��֬��HR����)���������ӽ���������ӽ���Ĥ���ᴦ�������������õ����˵���Һ���䷴Ӧԭ��Ϊ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������һ����Ļ�ѧ��Դ���⣬��ͼ�Ǻ�ˮ�ӹ���ʾ��ͼ��
��1����ˮ��������ͨ�����õ��Ʊ���ˮ�ķ����� (д������)��
��2����ͼ�Ǵ�Ũ����ˮ����ȡ�������ͼ��д����ͼ���Լ�A�Ļ�ѧʽ �� ��Һ�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��Һ�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��3���Ʊ�����þ��ͨ��������ڵ� ��������MgO����ԭ����
��������MgO����ԭ����
��
��4��ʳ��Ҳ��һ����Ҫ�Ļ���ԭ�ϣ��ȼҵ����ͨ����ⱥ��ʳ��ˮ���Ʊ�NaOH��H2��Cl2����ˮ�еõ��Ĵ�����������һЩ���ʣ��������һЩ��ѧ�Լ���ʹ���ʳ��������������ˮ������������ӽ���������ԭ���� �����ʳ��ˮ�����ӽ���Ĥ�����н��У������ӽ���Ĥ�������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)������һ����Ҫ�Ļ���ԭ�ϡ�Ŀǰ�Ƽҵ��Ҫ�С�������͡������Ƽ��(�����Ƽ)���ֹ��ա�
�١��������������CaCl2�����д���ù����в���CaCl2�Ļ�ѧ����ʽ ��
��д���������Ƽ���йط�Ӧ�Ļ�ѧ����ʽ________________��
��CO2���Ƽҵ����Ҫԭ�ϣ��������Ƽ���롰�������CO2����Դ�кβ�ͬ______________________________________��
(2)������ҵ�Դٽ����ú���ᷢչ������Ҫ���á�
�� ����ʱ������衢�̺�����Ŀ����_______________________��
�� ����ֺ��е�CrԪ���������ֹ��̵�����____(�ǰ����)���롣
�� ���������������У�β�������е���Ҫ��Ⱦ����________���ӻ����;��ýǶȿ��ǣ�����β��������������_________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ˮ��һ����Ҫ����Ȼ��Դ���������������治��ȱ�ٵ����ʡ�ˮ������ֱ��Ӱ�����彡��
��1�����н϶�______________��ˮ��ΪӲˮ��Ӳˮ���Ⱥ�������������ӷ���ʽΪ__________________________( д������һ�ֳ�����ļ���) ��
��2����ͼΪij����ˮ������Ȼˮ�Ʊ�����ˮ(ȥ����ˮ)�Ĺ�������ʾ��ͼ��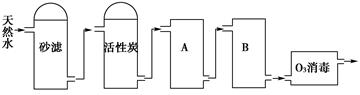
�ٻ���̿��������__________________��O3�������ŵ���________________��
��A��B�з��õ��������Ʒֱ��ǣ�A__________________��B_______________��A��B�з��õ������Ƿ���Ի���?��˵��ԭ��________________________________________��
��3��ͨ��ʩ��һ��ѹ��ʹˮ����ͨ����Ĥ��������ӻ����ӽ���, �Ӷ���ô���ˮ�ķ�����Ϊ ��������������ˮʱ, ʹ����ͨ����Ĥ���ƶ����� ��
��4����������ˮ�Ĵ���ʱ, ������еķ����Dzⶨˮ�� ��
��5��ij �� �� �� ˮ ��c(Ca2+)Ϊ1.0��10-3mol/L, c(Mg2+)Ϊ5.0��10-4mol/L,c(HCO3_)Ϊ8.0��10-4mol/L������ҩ��������1000L ,Ӧ����Ca(OH) 2 g , Na2CO 3 __________ g ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(14��)
LiPF6������ӵ���й㷺Ӧ�õĵ���ʡ�ij������LiF��PCl5Ϊԭ�ϣ����·�Ӧ�Ʊ�LiPF6�����������£�
��֪��HCl�ķе��ǣ�85.0 �棬HF�ķе���19.5 �档
��1���ڢٲ���Ӧ����ˮHF�������� �� ����Ӧ�豸�����ò������ʵ�ԭ���� (�û�ѧ����ʽ��ʾ)����ˮHF�и�ʴ�ԺͶ��ԣ�������ȫ�ֲ���ʾ�������С�Ľ�HFմ��Ƥ���ϣ���������2%�� ��Һ��ϴ��
��2��������������ˮ�����½��У��ڢ۲���Ӧ��PCl5����ˮ�⣬�����Ϊ�����ᣬд��PCl5ˮ��Ļ�ѧ����ʽ�� ��
��3���ڢܲ�������õķ����� ���ڢݲ�����β����HF��HCl���õķ����� ��
��4��LiPF6��Ʒ��ͨ����������LiF��ȡ��Ʒwg�����Li�����ʵ���Ϊnmol�������Ʒ��LiPF6�����ʵ���Ϊ mol(�ú���w��n�Ĵ���ʽ��ʾ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����Ԫ�آ�Cl����Na����Br����I����Mg����U�����ں�ˮ�е���Ԫ�ص��ǣ� ��
| A���٢ڢ� | B���ܢ� |
| C���٢ڢۢ� | D���ۢܢݢ� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com