
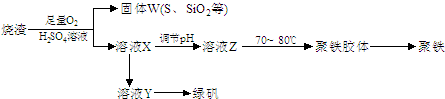
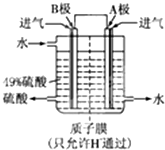
ЪЕбщЪвРћгУСђЫсГЇЩедќ(жївЊГЩЗжЮЊЬњЕФбѕЛЏЮяМАЩйСПFeSЁЂSiO2ЕШ)жЦБИОлЬњ(МюЪНСђЫсЬњЕФОлКЯЮя)КЭТЬЗЏ(FeSO4ЁЄ7H2O)ЃЌЙ§ГЬШчЯТЃК
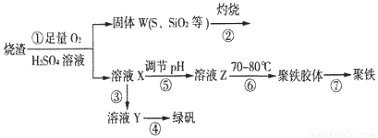
ЃЈ1ЃЉНЋЙ§ГЬЂкжаЕФВњЩњЕФЦјЬхЭЈШыЯТСаШмвКжаЃЌШмвКЛсЭЪЩЋЕФЪЧ__________ЃЛ
AЃЎЦЗКьШмвК??????? BЃЎзЯЩЋЪЏШяШмвК????? CЃЎЫсадKMnO4ШмвК???? DЃЎфхЫЎ
ЃЈ2ЃЉЙ§ГЬЂйжаЃЌFeSКЭO2ЁЂH2SO4ЗДгІЕФЛЏбЇЗНГЬЪНЮЊЃК___________________________________ЁЃ
ЃЈ3ЃЉЙ§ГЬЂлжаЃЌашМгШыЕФЮяжЪЪЧ___________________________ЁЃ
ЃЈ4ЃЉЙ§ГЬЂмжаЃЌеєЗЂНсОЇашвЊЪЙгУОЦОЋЕЦЁЂШ§НЧМмЁЂФрШ§НЧЃЌЛЙашвЊЕФвЧЦїга_______________ЁЃ
ЃЈ5ЃЉЙ§ГЬЂнЕїНкpHПЩбЁгУЯТСаЪдМСжаЕФ___________ (ЬюбЁЯюађКХ)ЃЛ
AЃЎЯЁСђЫс??????? BЃЎCaCO3?????? CЃЎNaOHШмвК
ЃЈ6ЃЉЙ§ГЬЂожаЃЌНЋШмвКZМгШШЕН70вЛ80ЁцЃЌФПЕФЪЧ_____________________ЁЃ
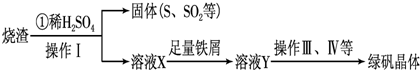
ЃЈ7ЃЉЪЕбщЪвЮЊВтСПЫљЕУЕНЕФОлЬњбљЦЗжаЬњдЊЫиЕФжЪСПЗжЪ§ЃЌНјааЯТСаЪЕбщЁЃЂйгУЗжЮіЬьЦНГЦШЁ2.70gбљЦЗЃЛЂкНЋбљЦЗШмгкзуСПЕФбЮЫсКѓЃЌМгШыЙ§СПЕФТШЛЏБЕШмвКЃЛЂлЙ§ТЫЁЂЯДЕгЁЂИЩдяЃЌГЦСПЃЌЕУЙЬЬхжЪСПЮЊ3.495gЁЃШєИУОлЬњжївЊГЩЗжЮЊ[(Fe(OH)(SO4)]nЃЌдђИУОлЬњбљЦЗжаЬњдЊЫиЕФжЪСПЗжЪ§ЮЊ___________ЁЃ(МйЩшдгжЪжаВЛКЌЬњдЊЫиКЭСђдЊЫи)ЁЃ
ЃЈ1ЃЉACD ЃЈ2ЃЉ4FeS + 3O2 + 6H2SO4 = 2Fe2(SO4)3 + 6H2O+4S ЃЈ3ЃЉFe(ЛђЬњ)
ЃЈ4ЃЉеєЗЂУѓЁЂВЃСЇАє ЃЈ5ЃЉC? ЃЈ6ЃЉДйНјFe3+ЕФЫЎНт ЃЈ7ЃЉ31.11%
ЁОНтЮіЁП
ЪдЬтЗжЮіЃКЃЈ1ЃЉЙЬЬхWжаКЌгаSдкзЦЩеЪБЛсВњЩњSO2ЦјЬхЁЃAЃЎАбSO2ЭЈШыЦЗКьШмвК SO2гаЦЏАзадЃЌФмЪЙЦЗЛЈШмвКЭЪЩЋЁЃе§ШЗЁЃBЃЎSO2ЕФЫЎШмвКЯдЫсадЃЌзЯЩЋЪЏШяШмвКБфЮЊКьЩЋЁЃДэЮѓЁЃCЃЎSO2гаЛЙдадЃЌФмБЛЫсадKMnO4ШмвКбѕЛЏЮЊСђЫсЃЌKMnO4дђБЛЛЙдЮЊЮоЩЋЕФMnSO4 .е§ШЗЁЃ DЃЎSO2гаЛЙдадЃЌФмБЛфхЫЎбѕЛЏЮЊСђЫсЃЌфхЫЎБЛЛЙдЮЊЮоЩЋЕФHBrЁЃе§ШЗЁЃвђДЫбЁЯюЮЊACDЁЃЃЈ2ЃЉИљОнЕчзгЪиКуКЭжЪСПЪиКуЖЈТЩПЩЕУГіЙ§ГЬЂйжаЃЌFeSКЭO2ЁЂH2SO4ЗДгІЕФЛЏбЇЗНГЬЪНЮЊЃК4FeS+3O2+ 6H2SO4 = 2Fe2(SO4)3 +6H2O + 4SЁЃЃЈ3ЃЉдкШмвКXжаКЌгаFe2(SO4)3 КЭH2SO4ЃЌЖјТЬЗЏЪЧСђЫсбЧЬњОЇЬхЁЃЫљвдЙ§ГЬЂлжаЃЌашМгШыЕФЮяжЪЪЧЛЙдМСFeЗлЁЃЃЈ4ЃЉ ОЙ§ЛЙдFeЗлЛЙдЕФFe2(SO4)3 КЭH2SO4ШмвКЮЊFeSO4.вЊДгИУШмвКжаЕУЕНТЬЗЏЃЌвђЮЊСђЫсбЧЬњЕФШмНтЖШЪмЮТЖШЕФгАЯьБфЛЏНЯДѓЃЌЫљвдВЩгУРфШДШШБЅКЭШмвКЕФЗНЗЈжЦШЁЁЃЪЕбщВйзїгаеєЗЂЁЂХЈЫѕЁЂРфШДЁЂНсОЇЁЃдкЙ§ГЬЂмжаЃЌеєЗЂНсОЇашвЊЪЙгУОЦОЋЕЦЁЂШ§НЧМмЁЂФрШ§НЧЃЌЛЙашвЊеєЗЂУѓЁЂВЃСЇАє ЁЃЃЈ5ЃЉЙ§ГЬЂнЕїНкpHПЩбЁгУЯТСаЪдМСжаЕФгІИУгУМюЃЌЯрБШЖјбдNaOHЕФШмНтЖШДѓЃЌдгжЪРызгNa+БШCa2+ШнвзГ§ШЅЁЃбЁЯюЮЊCЁЃЃЈ6ЃЉ Fe2(SO4)3ЪЧЧПЫсШѕМюбЮЃЌЫЎНтВњЩњFe(OH)3НКЬхЁЃбЮЕФЫЎНтЗДгІЪЧЮќШШЗДгІЃЌдкЙ§ГЬЂожаЃЌНЋШмвКZМгШШЕН70вЛ80ЁцЃЌФПЕФЪЧЮЊСЫДйНјFe3+ЕФЫЎНт ЁЃЃЈ7ЃЉИУОлЬњдкОЙ§ЫсШмНтКѓЃЌМгШыЙ§СПЕФТШЛЏБЕШмвКЁЃЕУЕНЕФГСЕэЮЊBa SO4ЁЃn(Ba SO4)= 3.495gЁТ233g/mol=0.015mol.Ыљвдn(Fe)=0.015mol.m(Fe)=0.015molЁС56g/mol=0.84gЁЃвђДЫдкдкОлЬњжаFeдЊЫиЕФКЌСПЮЊ(0.84gЁТ2.70g) ЁС100% =33.11%ЁЃ
ПМЕуЃКПМВщSO2ЕФЛЏбЇаджЪЁЂЗНГЬЪНЕФЪщаДЁЂбЮЕФЫЎНтЁЂЛьКЯЮяЕФЗжРыЗНЗЈЁЂдЊЫиЕФКЌСПЕФМЦЫуЕФжЊЪЖЁЃ


 ПЊаФЭмПкЫуЬтПЈЯЕСаД№АИ
ПЊаФЭмПкЫуЬтПЈЯЕСаД№АИ
| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК

ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃКдФЖСРэНт
| ||
| ||
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃКдФЖСРэНт
| ДпЛЏМС |
| Ёї |
| c2(SO3) |
| c(O2)?c2(SO2) |
| c2(SO3) |
| c(O2)?c2(SO2) |
| O | - 4 |
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК

ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
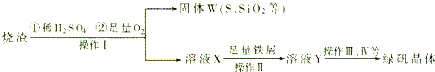

- 4 |
ВщПДД№АИКЭНтЮі>>
ЙњМЪбЇаЃгХбЁ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com