
�� ���� |
��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| �� | �� | �� | ||||||
| �� | �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� |


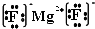
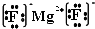
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
�� 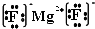 ���ʴ�Ϊ��
���ʴ�Ϊ��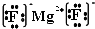 ��
��

 ������ϵ�д�
������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �� ���� |
��A | ��A | ��A | ��A | ��A | ��A | ����A | 0 |
| �� | �� | �� | �� | |||||
| �� | �� | �� | �� | �� | �� |




| ʵ�鲽�� | ʵ������ |
| 1����һС��ܵĵ��ʷ�����з�̪��Һ����ˮ�� | �Ƹ���ˮ���ϣ��۳�С���Ĵ��ζ� �Ƹ���ˮ���ϣ��۳�С���Ĵ��ζ� ������˻˻����������ʧ����Һ��ɺ�ɫ |
| 2����������ˮע��ʢ�д�ĥ���Ģݵĵ��ʵ��Թ��У��ٵμӷ�̪��һ��ʱ������������ | ���Ⱥ� þ�����������ɫ���ݣ���Һ�ʺ�ɫ þ�����������ɫ���ݣ���Һ�ʺ�ɫ |
| 3����2mL 1mol/L�������ʢ�д�ĥ���Ģݵĵ��ʵ��Թ��� | ���ҷ�Ӧ��Ѹ�ٲ���������ɫ���� |
| 4����2mL 1mol/L�������ʢ�д�ĥ���� Al Al �ĵ��ʵ��Թ��� |
��Ӧ������һ��ʱ�������ɫ���� |
| ���ۣ� Na��Mg��Al����Ԫ�صĽ��������μ��� Na��Mg��Al����Ԫ�صĽ��������μ��� ����Ԫ�ط��Ž������˵���� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A | |||||||||||||||||
| B | C | D | |||||||||||||||
| E | F | G | |||||||||||||||
| H | |||||||||||||||||


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�





�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�� ���� |
IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 |
| �� | �� | �� | ||||||
| �� | �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�


�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com