
Na��Cu��O��Si��S��Cl�dz���������Ԫ�ء�
(1)Naλ��Ԫ�����ڱ���________���ڵ�________�壻S�Ļ�̬ԭ�Ӻ�����________��δ�ɶԵ��ӣ�Si�Ļ�̬ԭ�Ӻ�������Ų�ʽΪ________________________��
(2)�á�>����<����գ�
| ��һ������ | ���Ӱ뾶 | �۵� | ���� |
| Si____S | O2��____Na�� | NaCl____Si | H2SO4____HClO4 |
(3)CuCl(s)��O2��Ӧ����CuCl2(s)��һ�ֺ�ɫ���塣��25 �桢101 kPa�£���֪�÷�Ӧÿ����1 mol CuCl(s)������44.4 kJ���÷�Ӧ���Ȼ�ѧ����ʽ��________________________________________________________________________
________________________________________________________________________��
(4)ClO2������ˮ�ľ�������ҵ�Ͽ���Cl2����NaClO2��Һ��ȡClO2��д���÷�Ӧ�����ӷ���ʽ�����������ת�Ƶķ������Ŀ��________________________________________________________________________��
 ͬ����ϰ���ϴ�ѧ������ϵ�д�
ͬ����ϰ���ϴ�ѧ������ϵ�д� ͬ����ϰ���ϴ�ѧ������ϵ�д�
ͬ����ϰ���ϴ�ѧ������ϵ�д� ����ϰ�⽭��ϵ�д�
����ϰ�⽭��ϵ�д� ѧ���쳵��������������������ϵ�д�
ѧ���쳵��������������������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ҵ�ϴӷ�Ǧ���ص�Ǧ�����Ǧ�Ĺ����У�����̼������Һ�봦�����Ǧ��(��Ҫ�ɷ�ΪPbSO4)������Ӧ��PbSO4(s)��CO32��(aq) PbCO3(s)��SO42��(aq)��ij��������PbSO4Ϊԭ��ģ��ù��̣�̽��������Ӧ��ʵ���������������ijɷ֡�
PbCO3(s)��SO42��(aq)��ij��������PbSO4Ϊԭ��ģ��ù��̣�̽��������Ӧ��ʵ���������������ijɷ֡�
��1��������Ӧ��ƽ�ⳣ������ʽ��K=
��2������ʱ����������ͬ��PbSO4��Ʒ�зֱ����ͬ�����ͬŨ�ȵ�Na2CO3��NaHCO3��Һ����ʵ������ת������ ��Һ��PbSO4ת���ʽϴ�������
��3���������ף�������Ӧ���������ɼ�ʽ̼��Ǧ[2PbCO3��Pb(OH)2]������PbCO3���ȶ��ֽ�����PbO���ÿ�����Թ�����������PbSO4���ijɷ�������¼��衣������ɼ�����ͼ�������
����һ��ȫ��ΪPbCO3��
������� ��
�������� ��
��4��Ϊ��֤����һ�Ƿ��������������������о���
�ٶ����о�����������±�������
| ʵ�鲽��(��Ҫ��д������������̣� | Ԥ�ڵ�ʵ������ͽ��� |
| ȡһ������Ʒ��ָ�� |
�ڶ����о���ȡ26.7 mg�ĸ�����Ʒ�����ȣ���
�ù����������¶ȵı仯��ϵ����ͼ��ijͬѧ
��ͼ����Ϣ�ó����ۣ�����һ�����������Ƿ�
ͬ���ͬѧ�Ľ��ۣ����������ɣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Na2CO3������������ǶԴ�����ò�ͬ���෨�ķ��࣬����ȷ���� �� ��
A��Na2CO3�Ǽ� B��Na2CO3����
C��Na2CO3������ D��Na2CO3��̼����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ͬ��ͬѹ�µ�N2��H2���������ͬʱ�����������������Ϊ___________�����ܶȱ�Ϊ__________�����������ʱ����������������___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Na��Cu��O��Si��S��Cl�dz���������Ԫ�ء�
(1)Naλ��Ԫ�����ڱ���________���ڵ�________�壻S�Ļ�̬ԭ�Ӻ�����________��δ�ɶԵ��ӣ�Si�Ļ�̬ԭ�Ӻ�������Ų�ʽΪ________________________��
(2)�á�>����<����գ�
| ��һ������ | ���Ӱ뾶 | �۵� | ���� |
| Si____S | O2��____Na�� | NaCl____Si | H2SO4____HClO4 |
(3)CuCl(s)��O2��Ӧ����CuCl2(s)��һ�ֺ�ɫ���塣��25 �桢101 kPa�£���֪�÷�Ӧÿ����1 mol CuCl(s)������44.4 kJ���÷�Ӧ���Ȼ�ѧ����ʽ��________________________________________________________________________
________________________________________________________________________��
(4)ClO2������ˮ�ľ�������ҵ�Ͽ���Cl2����NaClO2��Һ��ȡClO2��д���÷�Ӧ�����ӷ���ʽ�����������ת�Ƶķ������Ŀ��________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
X��Y��Z��RΪǰ������Ԫ�أ���ԭ��������������XY2�Ǻ���ɫ���壻X����Ԫ�ؿ��γ�XH3��Z��̬ԭ�ӵ�M����K���������ȣ�R2����3d�������9�����ӡ�
��ش��������⣺
(1)Y��̬ԭ�ӵĵ����Ų�ʽ��________��Z���������е�һ��������������Ԫ����________��
(2)XY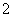 �����幹����________��R2����ˮ�������У��ṩ�µ��ӶԵ�ԭ����________��
�����幹����________��R2����ˮ�������У��ṩ�µ��ӶԵ�ԭ����________��
(3)Z��ijԪ���γɵĻ�����ľ�����ͼ��ʾ���������������������ӵĸ�������________��
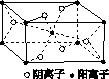
(4)��R���ʵķ�ĩ����XH3��Ũ��Һ�У�ͨ��Y2����ַ�Ӧ����Һ������ɫ���÷�Ӧ�����ӷ���ʽ��____________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���л������У����зǼ��Թ��ۼ������ӻ�������
A��CaC2 B��N2H4
C��Na2S2 D��NH4NO3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�������ڵ�ء��Ͻ𡢴����ȷ���Ӧ�ù㷺��
(1)���й��ڽ�������������˵����ȷ����________��
a�����������з������뱥����
b���������ǽ��������������ɵ��Ӽ�������
c��������������Ϊ����ӵ糡�����²������ɵ���
d���������й�������Ϊ�������������ղ��ų��ɼ���
(2)Ni��Ԫ�����ڱ��е�28��Ԫ�أ��ڶ����ڻ�̬ԭ��δ�ɶԵ�������Ni��ͬ�ҵ縺����С��Ԫ����_______________________ _________________________________________________��
(3)���˽��������Ni(CO)n������ԭ�Ӽ۵������������ṩ��������֮��Ϊ18����n��________��CO��N2�ṹ���ƣ�CO�����ڦҼ���м�����֮��Ϊ________��
(4)��ȩ(H2C===O)��Ni�������¼���ɵü״�(CH3OH)���״�������Cԭ�ӵ��ӻ���ʽΪ________���״������ڵ�O��C��H����________(����ڡ������ڡ���С�ڡ�)��ȩ�����ڵ�O��C��H���ǡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����������ȷ����
A.����������Һ�ķ�Ӧ�����û���Ӧ
B.�����Ӷ�ֻ�л�ԭ��
C.��ǿ�ᡢǿ���Ӧ������ֻ�������������������������
D.���Ӿ����ж����ڷ����߶�˹�������ܲ����ڹ��ۼ�
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com