
ʯī�ڲ�����������ҪӦ�á�ij����ʯī�к�SiO2(7.8%)��Al2O3(5.1%)��Fe2O3(3.1%)��MgO(0.5%)�����ʡ���Ƶ��ᴿ���ۺ����ù������£�
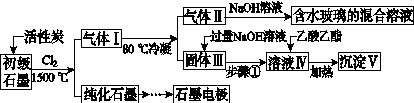
(ע��SiCl4�ķе�Ϊ57.6 �棬�����Ȼ���ķе������150 ��)
(1)��Ӧ����ͨ��Cl2ǰ����ͨһ��ʱ��N2����ҪĿ����____________________��
(2)���·�Ӧ��ʯī�����������ʾ�ת��Ϊ��Ӧ���Ȼ��������е�̼��������ҪΪ________�����������ij��õ�ˮ�����Ļ�ѧ��Ӧ����ʽΪ____________________________________________��
(3)�����Ϊ�����衢________��������Һ���е���������________��
(4)����Һ�����ɳ��������ܷ�Ӧ�����ӷ���ʽΪ______________________________________________��100 kg����ʯī�����ܻ�â�������Ϊ______kg��
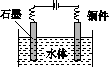
(5)ʯī��������Ȼˮ����ͭ���ĵ绯ѧ�����������ͼ����ʾ��ͼ��������Ӧ��ע��
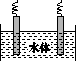
(1)�ų�װ���еĿ��� ��
(2)CO�� SiCl4��6NaOH===Na2SiO3��4NaCl��3H2O ��
(3)���ˡ�AlO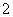 ��Cl�� ��
��Cl�� ��
(4)AlO ��CH3COOCH2CH3��2H2O
��CH3COOCH2CH3��2H2O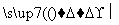 CH3COO����CH3CH2OH��Al(OH)3����7.8 ��
CH3COO����CH3CH2OH��Al(OH)3����7.8 ��
(5)
[����] (1)ͨ��N2��Ŀ����Ϊ���ų�װ���еĿ���(�ر�������)����ֹ�ڸ���ʱʯī��������Ӧ��(2)���·�Ӧ��SiO2��Al2O3��Fe2O3��MgO�ֱ�ת��ΪSiCl4��AlCl3��FeCl3��MgCl2����Ϊʯī�ǹ����ģ��ʸ��������£�C��SiO2��Fe2O3��Ӧ���ɵ���CO������SiCl4�ķе�Ϊ57.6 �棬����80 �棬�������ΪSiCl4����NaOH��Һ��ˮ������Na2SiO3��NaCl: SiCl4��6NaOH===Na2SiO3��4NaCl��3H2O��(3)AlCl3��FeCl3��MgCl2�ķе������150 �棬��80 ���±�Ϊ����� AlCl3��FeCl3��MgCl2����NaOH��Ӧ������NaAlO2��Fe(OH)3��Mg(OH)2��NaCl��ͨ�����˽�����Fe(OH)3��Mg(OH)2�˳����õ�����Һ����Ҫ��NaAlO2��NaCl��(4)NaAlO2����ˮ�����Һ�Լ��ԣ�NaAlO2��2H2O Al(OH)3��NaOH������������������ˮ�⣺CH3COOCH2CH3��NaOH
Al(OH)3��NaOH������������������ˮ�⣺CH3COOCH2CH3��NaOH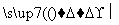 CH3COONa��CH3CH2OH����ʹNaAlO2����ˮ������Al(OH)3��������Һ��ת��Ϊ�������ķ�ӦΪNaAlO2��2H2O��CH3COOCH2CH3
CH3COONa��CH3CH2OH����ʹNaAlO2����ˮ������Al(OH)3��������Һ��ת��Ϊ�������ķ�ӦΪNaAlO2��2H2O��CH3COOCH2CH3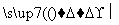 Al(OH)3����CH3COONa��CH3CH2OH������Alԭ���غ㣬��֪100 kg����ʯī�ɵ�m[Al(OH)3]��
Al(OH)3����CH3COONa��CH3CH2OH������Alԭ���غ㣬��֪100 kg����ʯī�ɵ�m[Al(OH)3]��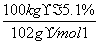 ��2��78 g��mol��1��7.8 kg��
��2��78 g��mol��1��7.8 kg��
(5)ˮ����ͭ���ĵ绯ѧ���������˵�Ᵽ�������÷�����ʯī��������ͭ��������������ӵ���������������������ʯī��ͭ��ֱ���������γ�ԭ��أ���ͭ�����������������Բ��ɲ��á�


 �¿α�����Ķ�ѵ��ϵ�д�
�¿α�����Ķ�ѵ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij�� ����ֻ����̼Ԫ�أ�����˵����ȷ����( )
����ֻ����̼Ԫ�أ�����˵����ȷ����( )
A. ������һ���ǵ��� B. ������һ���Ǻ�ɫ��
C. ��������O2��ȼ�ղ��������C02 D. �����ʿ����ǻ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ͼ��X��һ�־���ˮ����ζ�ĺϳ����ϣ�A��ֱ���л��E��FeCl3��Һ��������ɫ��
�����������Ϣ�ش�
��1��H�к��������ŵ�������____________��B��I�ķ�Ӧ����Ϊ____________��
��2��ֻ��һ���Լ�����D��E��H�����Լ���____________��
��3��H��J��Ϊͬ���칹�壬J������������ˮ�����������ɣ�J�Ľṹ��ʽΪ____________��
��4��D��F��Ӧ����X�Ļ�ѧ����ʽΪ____________________________________��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������������Ч���ٶ���������ŷš�ʵ�����÷�ú��(��Ҫ��Al2O3��SiO2��)�Ʊ���ʽ������[Al2(SO4)x(OH)6��2x]��Һ�����������������о���
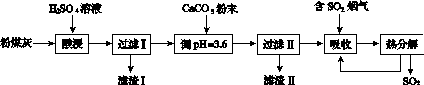
(1)���ʱ��Ӧ�Ļ�ѧ����ʽΪ____________________�����������Ҫ�ɷ�Ϊ________(�ѧʽ)��
(2)��CaCO3������Һ��pH��3.6����Ŀ�����к���Һ�е��ᣬ��ʹAl2(SO4)3ת��ΪAl2(SO4)x(OH)6��2x�����������Ҫ�ɷ�Ϊ________(�ѧʽ)������Һ��pHƫ�ߣ����ᵼ����Һ����Ԫ�صĺ������ͣ���ԭ����__________________________(�����ӷ���ʽ��ʾ)��
(3)���������о���ȫ�ȷֽ�ų���SO2������С�����յ�SO2��������Ҫԭ����________________________��������SO2ǰ����Һ��ȣ��ȷֽ��ѭ�����õ���Һ��pH��________(�������С�����䡱)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���з�Ӧ�У���Ӧ������������ص���(����)
A������ͨ�����ȵ�CuO��ĩ
B��������̼ͨ��Na2O2��ĩ
C������Fe2O3�������ȷ�Ӧ
D����п��Ͷ��Cu(NO3)2��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������л�ѧ������˵����ȷ����(����)
A�����·������������ۻ�Ϊͬ���칹��
B���Ʒ��ס����С���ѹ���ȵIJ�����ǺϽ�
C����ըʳ��Ļ����ͺ�ţ�Ͷ��ǿ������ı�������
D��ĥ�����Ĵ��������ʣ�������к��ʱ���˰�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
1.52g ͭþ�Ͻ���ȫ�ܽ���50mL �ܶ�Ϊ1.40 g/mL����������Ϊ63%��Ũ�����У��õ�NO2��N2O4�Ļ������1120 mL����״��������Ӧ�����Һ�м���1.0 mol/L NaOH��Һ������������ȫ������ʱ���õ�2.54 g����������˵������ȷ���ǣ�������
A���úϽ���ͭ��þ�����ʵ���֮����2:1
B. ��Ũ������HNO3�����ʵ���Ũ����14.0 mol/L
C��NO2��N2O4�Ļ�������У�NO2�����������80%
D���õ�2.54 g����ʱ������NaOH��Һ�������600 mL
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
һ���ѧ�о��ɹ�������ͭ��������(CuMn2O4)���ڳ����´����������е�һ����̼�ͼ�ȩ(HCHO)��
(1)��һ�����ʵ���Ũ�ȵ�Cu(NO3)2��Mn(NO3)2��Һ�м���Na2CO3��Һ�����ó������������գ����Ƶ�CuMn2O4��
��Mn2����̬�ĵ����Ų�ʽ�ɱ�ʾΪ ��
��NO3���Ŀռ乹�� (����������)��
(2)��ͭ��������Ĵ��£�CO��������CO2��HCHO��������CO2��H2O��
�ٸ��ݵȵ���ԭ����CO���ӵĽṹʽΪ ��
��H2O������Oԭ�ӹ�����ӻ�����Ϊ ��
��1molCO2�к��еĦҼ���ĿΪ ��
(3)��CuSO4��Һ�м������NaOH��Һ������[Cu(OH)4]2���������ǿռ乹�ͣ�[Cu(OH)4]2���Ľṹ����ʾ��ͼ��ʾΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��. A��B��C��D��E��F��G����Ԫ�أ����������������ƶϣ�
��A��B��C��ͬһ���ڵĽ���Ԫ�أ�ԭ�Ӻ������������Ӳ㣬A��ԭ�Ӱ뾶�����������������ԭ�Ӱ뾶A>B>C��
��D��E�Ƿǽ���Ԫ�أ����Ǹ��������Ͽ�������̬�⻯��HD��HE������ʱ��D�ĵ�����Һ�壬E�ĵ����ǹ���
��F�ڳ����������壬�����ȶ����dz����������������
��G�dz�����ԭ�Ӱ뾶��С��Ԫ��
��1��A �������� ��Bλ�����ڱ��е� ���ڵ� �塣
��2��C��ԭ�ӽṹʾ��ͼ�� ��
AԪ����DԪ���γɻ�����ĵ���ʽ�� ��
��3��F��Ԫ�ط����� ��
��4������������Ԫ���У�����������Ӧ��ˮ���������ǿ�Ļ�ѧʽ�� ��������ǿ�Ļ�ѧʽ�� ����̬�⻯�����ȶ��Ļ�ѧʽ�� ��
��5����C���������Ӧ��ˮ����Ͷ�뵽A���������Ӧ��ˮ�����У���Ӧ�Ļ�ѧ����ʽ��
��
��1���±��е�ʵ�߱�ʾԪ�����ڱ����ֱ߽磬����ʵ�������ڱ�δ��ɵı߽缰����Ԫ����ǽ���Ԫ�صķֽ��ߡ�
��2���ѷ�������Ҫ���Ԫ�ص�Ԫ�ط��ű��ڱ��ж�Ӧλ�ô���
�� ����������ˮ���������ǿ��������Ԫ�س��⣩
�� ��̬�⻯�����ȶ��ķǽ���Ԫ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com