
ij��ѧʵ���Ҳ����ķ�Һ�к���Fe3����Cu2����Ba2����Cl���������ӣ���������з����Է�Һ���д������Ի��ս������Ʊ��Ȼ������Ȼ������塣
��1������1�к��еĽ��������� ��
��2������ʱ����H2O2��Һ������Ӧ�����ӷ���ʽΪ ��
��3�����������У�������Ϊ�Լ�X���� ������ĸ����
| A��BaCl2 | B��BaCO3 |
| C��NaOH | D��Ba(OH)2 |
��1������ͭ��2�֣���1�֣�
��2��2Fe2����2H����H2O2��2Fe3����2H2O��2�֣�
��3��BD��2�֣���1�֣�
��4��ȡ���һ��ϴ��Һ����������1��2����������Һ���������ְ�ɫ���ǣ�������ϴ����ȫ������ȡ���һ��ϴ��Һ����������1��2����������Һ���������ְ�ɫ���ǣ�������ϴ����ȫ������2�֣�
��5������Fe3��ˮ�⣨�����𰸾��ɣ���2�֣�
��6������Ũ�� ���ˣ�2�֣���1�֣�
���������������1��������м�ܰ�ͭ�û�������ͬʱ����ʣ�࣬����Ϊ����ͭ����2�����������������������Ϊ�����Ӻ��ٳ�����ȥ��˫��ˮ����Fe2������3������X�Լ���Ϊ�˵���pHʹ������������������������Ϊ�˲����������ʿ��Լ���BaCO3��Ba(OH)2�����ʣ���4���������ϴ���Ƿ�ɾ�һ��Ӧȡ���ε���Һ������ܺ��е��������ӣ�һ�������������ӡ������ӵȣ���5���Ȼ�����ˮ��Һ��ˮ�⣬�����������������Fe3��ˮ�⣻��6�����˺�õ�����ҺΪϡ��Һ������Һ�еõ�������Բ�������Ũ�����½ᾧ�ķ�����
���㣺���鹤ҵ�����в���ԭ�������й����⡣


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ú��ʯ��Ԥ������SiO2��63%����Al2O3��25%����Fe2O3��5%����������þ�Ļ�����ȣ�һ���ۺ����ù���������£�
��1�����������������Ҫ��Ӧ�����ӷ���ʽΪ__________ ��____________��
��2���������ʱ�������ʵ�Ӱ�����ؿ�����_____ ________��_____ ______����д��������
��3������X�Ļ�ѧʽΪ___________�������ܡ�ʱ��Ӧ����Ҫ���ӷ���ʽΪ��Fe3++3OH- = Fe(OH)3���� ��
��4����֪Fe3+��ʼ�����ͳ�����ȫ��pH�ֱ�Ϊ2.1��3.2��Al3+��ʼ�����ͳ�����ȫ��pH�ֱ�Ϊ4.1��5.4Ϊ�˻�ò�ƷAl(OH��3����ú��ʯ�������ȡҺ��ʼ����ֻ��CaCO3һ���Լ����������������� ��
��5����ú��ʯΪԭ�ϻ����Կ���������Ʒ��������ú��ʯ�������ȡҺ������������AlCl3��Һ�в���ͨ��HCl���壬����������AlCl3��6H2O���壬��ϻ�ѧƽ���ƶ�ԭ���������������ԭ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��(Sr)������������Ԫ�أ��䵥�ʺͻ�����Ļ�ѧ������ơ��������ơ�ʵ�����ú�̼���ȵķ���(��SrCO3 38.40%��SrO12.62%��CaCO3 38.27%��BaCO3 2.54%���������������������8.17%)�Ʊ������ȴ�Ʒ�IJ���ʵ��������£�
��1������Ũ�������������Ϊ65%���ܶ�Ϊ1.4g/cm3��Ҫ����30%ϡ����500mL������Ҫ���ĵ������� �������ƹ����в�ʹ����ƽ�������Ҫ����������� ������Ҫʹ�õ������� ��
��֪�����ε��ܽ��(g/100 gˮ)���±�
| �¶�/������ | 0 | 20 | 30 | 45 | 60 | 80 | 100 |
| Sr(NO3)2 | 28.2 | 40.7 | 47 | 47.2 | 48.3 | 49.2 | 50.7 |
| Ca(NO3)2��4H2O | 102 | 129 | 152 | 230 | 300 | 358 | 408 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����̿��Ʊ�������ص���Ҫ��Ӧ���£�
�������� 3MnO2+KClO3+6KOH 3K2MnO4+KCl+3H2O
3K2MnO4+KCl+3H2O
�����绯 3K2MnO4+2CO2=2KMnO4+MnO2��+2K2CO3
������ʵ��ܽ�ȣ�293K�����±���
| | K2CO3 | KHCO3 | K2SO4 | KMnO4 |
| �ܽ��/g | 111 | 33.7 | 11.1 | 6.34 |
 2KMnO4+2KOH+H2������ԭ������ȣ���ⷨ������Ϊ ��
2KMnO4+2KOH+H2������ԭ������ȣ���ⷨ������Ϊ ��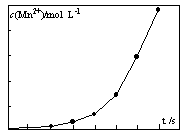
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����ӵĹ�ҵ��ˮ����������ͼ��ͼ��ʾ��
��1������������豸���н��е���_________��������д�������ƣ���ʵ��������һ��
����������____________����д�������ƣ����С�
��2�����豸������豸�������A��_____ ____�����豸������豸��������B��______________��������д���ʵĻ�ѧʽ��
��3�����豸���з�����Ӧ�Ļ�ѧ����ʽΪ��_______ ________��
��4�����豸���У�����B��ˮ��Һ��CaO��Ӧ������NaOH��H2O��________��ͨ��________��������д�������ƣ�������ʹ��������롣
��5��ͼ�У���ѭ��ʹ�õ�������C6H6��CaO��________��__________��������д���ʵĻ�ѧʽ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ˮ�Ǿ����Դ���⣬��ˮ���������ۺ����þ�����Ҫ���塣
���������գ�
��1���ȼҵ��Ҫ��ʳ��Ϊԭ�ϡ�Ϊ�˳�ȥ�����е�Ca2+��Mg2+��SO42������ɳ���ɽ���������ˮ��Ȼ��������в�������ȷ�IJ���˳���� ��
�ٹ��ˣ��ڼӹ�����NaOH��Һ���ۼ�����������ܼӹ�����Na2CO3��Һ���ݼӹ�����BaCl2��Һ
a���ڢݢܢ٢� b���٢ܢڢݢ� c d���ݢڢܢ٢�
��2����ʵ�����п�������ȡ�ķ�����ȡ�壬��ѡ�õ��Լ���________________��������Ҫ������������____________________��
��3����������������ữ�����Cl2�����ʵ�ԭ���� ��
��4������II��Ӧ�����ӷ���ʽ__________________________________________��
��5����ˮ������������У��¶�Ӧ������80~90�棬�¶ȹ�����Ͷ����������� �������ԭ�� ��
��6��Mg(OH)2�����л���Ca(OH)2����ѡ��__________��Һ����ϴ�ӳ�ȥ����ֱ�Ӽ���Mg(OH)2�õ�MgO���ٵ������MgO�ƽ���þ�������ɼ�ʵ�鲽�裬��_______��ѡ�ͬ�⡱������ͬ�⡱����˵���������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��һ�������������NaI��KCl��Na2CO3��Na2SO4��CaCl2��Cu(NO3)2�е�һ�ֻ�����ɣ�Ϊ�˼������������ʣ���������ʵ�飺
��ȡ������������ˮ���õ���ɫ����Һ��
��������Һ�еμ��Ȼ�����Һ���а�ɫ�������ɣ�
�۹��ˣ��������м���������ϡ���ᣬ���ֳ���û��ȫ���ܽ�������ɫ��ζ���������ɡ�
������Һ�м������������Ƶ���ˮ���ټ����������ͣ������ã��ϲ�Һ����Ϻ�ɫ��
��1�����жϣ����������п϶����� ��
һ��û�� ��
���ܺ��� ��
��2���Կ��ܺ��е����ʣ���ν���ʵ���Խ�һ�����顣
��
��3��ʵ����з����Ļ�ѧ��Ӧ���� ��Ӧ���Ӧ���ͣ�����Ҫʵ��������ƽ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�±����������ڲ�ͬ�¶��µ��ܽ�ȣ�g/100gˮ����
| | NaNO3 | KNO3 | NaCl | KCl |
| 10�� | 80.5 | 21.2 | 35.7 | 31.0 |
| 100�� | 175 | 246 | 39.1 | 56.6 |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����þ[Mg(ClO3)2]����������������ݼ��ȣ�ʵ�����Ʊ�����Mg(ClO3)2��6H2O���������£�
��֪����±����Ҫ�ɷ�ΪMgCl2��6H2O������MgSO4��FeCl2�����ʡ�
�����ֻ�������ܽ��(S)���¶�(T)�仯������ͼ��ʾ��
��1����������Ҫ����Ҫ���������� ��
��2������BaCl2��Ŀ���� ����MgO�����������������Ҫ�ɷ�Ϊ ��
��3������NaClO3������Һ������Ӧ�Ļ�ѧ����ʽΪ ���ٽ�һ����ȡMg(ClO3)2��6H2O��ʵ�鲽������Ϊ���������ᾧ���� ���� ���ܹ��ˡ�ϴ�ӡ�
��4����Ʒ��Mg(ClO3)2��6H2O�����IJⶨ��
����1��ȷ����3.50 g��Ʒ���100 mL��Һ��
����2��ȡ10.00 mL����ƿ�У�����10.00 mLϡ�����20 .00mL 1.000 mol��L��1��FeSO4��Һ���ȡ�
����3����ȴ�����£���0.100 mol��L��1 K2Cr2O7 ��Һ�ζ�ʣ���Fe2�����յ㣬�˹����з�Ӧ�����ӷ���ʽΪ��Cr2O72����6Fe2����14H����2Cr3����6Fe3����7H2O��
����4��������2��3�ظ����Σ�ƽ������K2Cr2O7 ��Һ15.00 mL��
��д������2�з�����Ӧ�����ӷ���ʽ�� ��
�ڲ�Ʒ��Mg(ClO3)2��6H2O����������Ϊ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com