
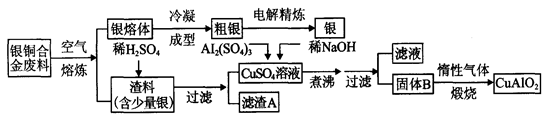
 4NO+6H2O��1�֣� NH4NO3��1�֣� (4)30 ��1�֣���15 ��1�֣�
4NO+6H2O��1�֣� NH4NO3��1�֣� (4)30 ��1�֣���15 ��1�֣� 4NO+6H2O�����ɵ�NO��������ˮ���������������ᣬ�����백����Ӧ��������臨�ð���̡�
4NO+6H2O�����ɵ�NO��������ˮ���������������ᣬ�����백����Ӧ��������臨�ð���̡�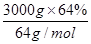 ��30mol������Ԫ���غ�ɵ����ɵ�CuAlO2Ҳ��30.0mol�����ݻ�ѧʽCuAlO2�е�Cu��Al������ϵ��Alԭ�Ӹ����غ�ɵ�,n[Al2(SO4)3]�� 30.0mol��2��15.0mol��������Ҫ��������Һ�������15.0mol��1.0mol/L��15.0L��
��30mol������Ԫ���غ�ɵ����ɵ�CuAlO2Ҳ��30.0mol�����ݻ�ѧʽCuAlO2�е�Cu��Al������ϵ��Alԭ�Ӹ����غ�ɵ�,n[Al2(SO4)3]�� 30.0mol��2��15.0mol��������Ҫ��������Һ�������15.0mol��1.0mol/L��15.0L��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 |  |  |  |
| A���������ճɻ� B�����˵ú�I����Һ C���ų���ı���Һ D������Ⲣ���ձ� | |||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
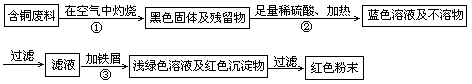
 2CuSO4+2H2O ) ���������������һ������
2CuSO4+2H2O ) ���������������һ������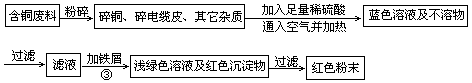
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ȥCO�л��е�CO2��������������Һϴ�� |
| B����AlCl3��Һ������ˮ�Ʊ�Al(OH)3 |
| C��������ͭ�۳�ȥFeCl2��Һ�е�FeCl3���� |
| D������ˮ���ռ�ϡHNO3��Cu��Ӧ������NO���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
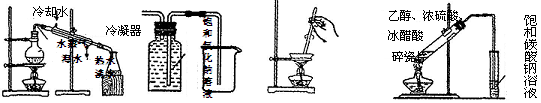
| A����ȡ����ˮ | B���ռ��Ȼ��� | C�������ᾧ | D��ʵ������ȡ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
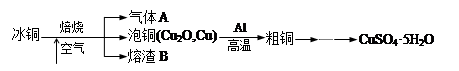
|

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
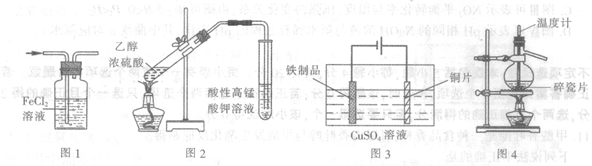
| A����ͼ1��ʾװ�ó�ȥHCl�����е�����C12 |
| B����ͼ2��ʾװ�ü������ʱ��������ϩ |
| C����ͼ3��ʾװ��������Ʒ�����ͭ |
| D����ͼ4��ʾװ�÷���������Ȼ�̼��Һ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com