
��H2O2��H2SO4�Ļ����Һ���ܳ��Ͼ�ӡˢ��·���ϵ�ͭ����֪��
Cu(s)��2H��(aq)===Cu2��(aq)��H2(g)
��H����64.39 kJ��mol��1
2H2O2(l)===2H2O(l)��O2(g)
��H����196.46 kJ��mol��1
H2(g)�� O2(g)===H2O(l)
O2(g)===H2O(l)
��H����285.84 kJ��mol��1
��H2SO4��Һ�У�Cu��H2O2��Ӧ����Cu2��(aq)��H2O(l)�ķ�Ӧ�Ȧ�H����
A����417.91 kJ��mol��1���� B����319.68 kJ��mol��1
C����546.69 kJ��mol��1  D����448.46 kJ��mol��1
D����448.46 kJ��mol��1
 ������ϵ�д�
������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���б仯��˷���ͬ��������������
A.��ɱ������� B.���C60���ۻ�
C.�Ȼ�����Ȼ��ص��ܽ� D.���������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ס��ҡ����������������У��ס��ҡ�����������ͬ��ij��Ԫ�أ�����֮���������ͼת����ϵ�������й����ʵ��ƶ���ȷ����(����)
A������Ϊ��̿��������O2 B������ΪFe��������Cl2
C������ΪAlCl3��������NH3 D������ΪNaHCO3��������HCl

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
A��B��D��E��G��M�������ֳ���Ԫ�أ����ǵĺ˵���������������У�Ԫ��M�Ļ�̬3d�������2�����ӣ�A�Ļ�̬ԭ��L���������K���������2����E�ļ�������ͬ����Ԫ�صļ������а뾶��С��D��Gͬ���壻B��D�γɵĻ������ж��֣�����һ���Ǻ���ɫ���塣
(1)M�Ļ�̬ԭ�Ӽ۲�����Ų�ʽΪ___________________________________________��Ԫ��B��D��G�ĵ�һ�������ɴ�С��˳����________________________(��Ԫ�ط��ű�ʾ)��
(2)�ü۲���ӶԻ�������Ԥ�⣬GD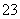 �����幹����____________(�����ֱ���)��
�����幹����____________(�����ֱ���)��
(3)M��D�γɵ�һ�ֳȺ�ɫ���徧���ṹ����ͼ��ʾ���仯ѧʽΪ________(��Ԫ�ط��ű�ʾ)��
(4)��֪������EB�ṹ�뵥�������ƣ������ʿ���E���Ȼ�����NaB3�ڸ����·�Ӧ�Ƶã������ɵ���B2���÷�Ӧ��ѧ����ʽΪ_______________________________________________������8.4 g B2���ɣ���ת�Ƶ�����Ϊ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������Ҫ�İ뵼����ϣ��������ִ����ӹ�ҵ�Ļ������ش��������⣺
(1)����Ҫ�Թ����Ρ�____________�Ȼ��������ʽ�����ڵؿ��С�
(2)���ʹ��������ʯ�ṹ���Ƶľ��壬����ԭ����ԭ��֮����________���ϣ��侧���й���8��ԭ�ӣ�����������λ�ù���________��ԭ�ӡ�
(3)���ʹ��ͨ������(SiH4)�ֽⷴӦ���Ʊ�����ҵ�ϲ���Mg2Si��NH4Cl��Һ�������з�Ӧ�Ƶ�SiH4���÷�Ӧ�Ļ�ѧ����ʽΪ______________________________________��
(4)̼����йػ�ѧ������������ʾ����Ҫ�����ͽ��������й���ʵ��
| ��ѧ�� | C—C | C—H | C—O | Si—Si | Si—H | Si—O |
| ����/ (kJ·mol��1) | 356 | 413 | 336 | 226 | 318 | 452 |
�ٹ���̼ͬ�壬Ҳ��ϵ���⻯�������������������϶�Զ���������࣬ԭ����________________________________________________________________________��
��SiH4���ȶ���С��CH4���������������ԭ����________________________________
________________________________________________________________________��
(5)�ڹ������У�SiO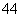 ������[����ͼ(a)]ͨ�����ö��������ӿ��γɵ�״����״����״���Ǽ���״�Ĵ���ṹ��ʽ��ͼ(b)Ϊһ�����������ṹ�Ķ�����������Siԭ�ӵ��ӻ���ʽΪ__________��Si��O��ԭ����֮��Ϊ__________����ѧʽΪ__________��
������[����ͼ(a)]ͨ�����ö��������ӿ��γɵ�״����״����״���Ǽ���״�Ĵ���ṹ��ʽ��ͼ(b)Ϊһ�����������ṹ�Ķ�����������Siԭ�ӵ��ӻ���ʽΪ__________��Si��O��ԭ����֮��Ϊ__________����ѧʽΪ__________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����˵����ȷ����
A��NaHCO3��H2 HCOONa��H2O��Ӧ�У����⡢������̾��������仯
HCOONa��H2O��Ӧ�У����⡢������̾��������仯
B����ͼ��ʾij���ȷ�Ӧ�ֱ����С�����������·�Ӧ�����е������仯
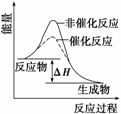
C����ѧ��Ӧ�����������ɣ�����ѭ�����غ㶨�ɺ������غ㶨��
D��2SO2(g)��O2(g)===2SO3(g)��4SO2(g)��2O2(g)===4SO3(g)�Ħ�H���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ӦA��B�D��C(��H��0)���������У���A��B�D��X(��H��0)����X�D��C(��H��0)����ʾ��ͼ�У�����ȷ��ʾ�ܷ�Ӧ�����������仯����
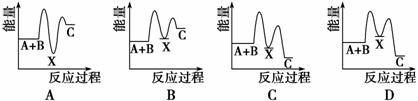
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʹ�õ���(CuSO4��5H2O)��0.1 mol��L��1������ͭ��Һ����ȷ�IJ���(����)
A�����������ȳ�ȥ�ᾧˮ��ȡ16 g�ܽ���1 Lˮ��
B����ȡ����25 g���ܽ���1 Lˮ��
C����25 g��������ˮ��Ȼ����Һϡ����1 L
D����16 g��������ˮ��Ȼ����Һϡ����1 L
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
NaCl ��һ�ֻ���ԭ�ϣ������Ʊ�һϵ�����ʣ���ͼ��ʾ������˵����ȷ����(����)
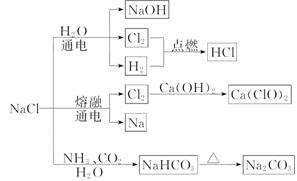
A��25��ʱ��NaHCO3��ˮ�е��ܽ�ȱ� Na2CO3�Ĵ�
B��ʯ������ Cl2 �ķ�Ӧ�У�Cl2 ���������������ǻ�ԭ��
C�������¸���� Cl2 ���ø�ƿ���ˣ����� Cl2 ��������Ӧ
D��ͼʾת����Ӧ����������ԭ��Ӧ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com