
(12��)��һ���¶��£���a.���ᡡb�����ᡡc�����������
(1)�����������ʵ���Ũ����ͬʱ��c(H��)�ɴ�С��˳����________________��
(2)ͬ�����ͬ���ʵ���Ũ�ȵ������ᣬ�к�NaOH�������ɴ�С��˳����______________��
(3)������c(H��)��ͬ�����Ҳ��ͬʱ���ֱ����������п����ͬ״���²������������ɴ�С��˳����___________��
(4)��c(H��)��ͬ�������ͬʱ��ͬʱ������״���ܶȡ�������ȫ��ͬ��п����������ͬ�����H2(��ͬ״��)����ʼʱ��Ӧ���ʵĴ�С��ϵΪ____________����Ӧ����ʱ��ij��̹�ϵ��__________��
��5)��c(H��)��ͬ�����������ˮϡ����ԭ����100����c(H��)�ɴ�С��˳����____________________________________________________________________��
��12�֣�ÿ��2�֣���1��b��a��c ��2��b��a��c ��3��c��a��b
��4��a��b��c a��b��c ��5��c��a��b
����������1�������Ƕ�Ԫǿ�ᣬ������һԪǿ�ᣬ������һԪ���ᣬ���Ե����������ʵ���Ũ����ͬʱ��c(H��)�ɴ�С��˳����b��a��c ��
��2��ͬ�����ͬ���ʵ���Ũ�ȵ������ᣬ�����������ӵ����ʵ�����࣬����ʹ�������ʵ�����ͬ�������к�NaOH�������ɴ�С��˳����b��a��c ��
��3��������c(H��)��ͬ�����Ҳ��ͬʱ������������������ӵ����ʵ�����ͬ������������Ҳ����ͬ�ġ������������ᣬ��Ũ��������ɵ�������࣬������ͬ״���²������������ɴ�С��˳����c��a��b��
��4����Ӧ��ʼʱ�����ӵ�Ũ����ͬ�����Կ�ʼʱ��Ӧ���ʵĴ�С��ϵΪa��b��c �����ڴ��������ᣬ�ڷ�Ӧ�����л��ܼ�������������ӣ����Դ���ķ�Ӧ��������������������ͬ�ģ�����Ӧ����ʱ��ij��̹�ϵ��a��b��c��
��5�����������ᣬϡ�ʹٽ����룬����ϡ�����У����ỹ�ܵ�������ס������ϡ����ͬ�ı��������������ǿ�������������ͬ������c(H��)�ɴ�С��˳����c��a��b��


 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2011������������2011�������һ�ε��в��ԣ����ۣ���ѧ�Ծ� ���ͣ������
(16��)
���û�ѧ��Ӧԭ���о���������Ԫ�صĵ��ʼ��仯����ķ�Ӧ����Ҫ���塣 ��(1)һ���¶Ⱥ�ѹǿ�£���ӦN2(g) + 3H2(g)
��(1)һ���¶Ⱥ�ѹǿ�£���ӦN2(g) + 3H2(g)  2NH3(g)�ﵽ��ѧƽ��״̬������ƽ����ϵ��ͨ�������ƽ�� (����������ҡ�����)�ƶ�����ʹ�û��Ը�ǿ�Ĵ������÷�Ӧ�Ħ�H (���������С�����ı䡱)��
2NH3(g)�ﵽ��ѧƽ��״̬������ƽ����ϵ��ͨ�������ƽ�� (����������ҡ�����)�ƶ�����ʹ�û��Ը�ǿ�Ĵ������÷�Ӧ�Ħ�H (���������С�����ı䡱)�� (2)��һ���¶Ⱥ�ѹǿ�£���֪��
(2)��һ���¶Ⱥ�ѹǿ�£���֪��
O2 (g) = O2+ (g) + e-�� H1=" 1175.7" kJ/mol
H1=" 1175.7" kJ/mol PtF6 (g) + e- = PtF6- (g)��
PtF6 (g) + e- = PtF6- (g)�� H2=" �D771.1" kJ/mol
H2=" �D771.1" kJ/mol O2PtF6 (s) = O2+ (g) + PtF6- (g)��
O2PtF6 (s) = O2+ (g) + PtF6- (g)��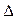 H3=" 482.2" kJ/mol
H3=" 482.2" kJ/mol ��ӦO2 (g) + PtF6 (g) = O2PtF6(s)��
��ӦO2 (g) + PtF6 (g) = O2PtF6(s)�� H="_____________" kJ/mol��
H="_____________" kJ/mol�� �����г����µ�������Һ����0.01 mol/L CH3COOH��Һ����0.01 mol/L HCl��Һ����pH=12�İ�ˮ����pH=12��NaOH��Һ����0.01 mol/L CH3COOH��Һ��pH=12�İ�ˮ�������Ϻ�������Һ����0.01 mol/L HCl��Һ��pH=12��NaOH��Һ��������������Һ��
�����г����µ�������Һ����0.01 mol/L CH3COOH��Һ����0.01 mol/L HCl��Һ����pH=12�İ�ˮ����pH=12��NaOH��Һ����0.01 mol/L CH3COOH��Һ��pH=12�İ�ˮ�������Ϻ�������Һ����0.01 mol/L HCl��Һ��pH=12��NaOH��Һ��������������Һ��
(1)�ݡ�����Һ�Ƚϣ�pH�ϴ���� ��
(2)������Һ�У�ˮ�ĵ���̶���ͬ����______________��
(3)���ڡ��ۻ�Ϻ�������ҺpH=7����������Һ���������________ ��(ѡ�����������������)��
(4)ϡ����ͬ��������Һ��pH���� �ڣ��� ��(ѡ�����������������)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�����ʡ��Ѩ��ѧ������ѧ��11�·��¿���ѧ�Ծ� ���ͣ���ѡ��
(12��)��һ���¶��£���a.���ᡡb�����ᡡc�����������
(1)���������� �ʵ���Ũ����ͬʱ
�ʵ���Ũ����ͬʱ ��������Һ��ˮ�ĵ���̶��ɴ�С��˳����____________������a��b��c��ʾ����ͬ��
��������Һ��ˮ�ĵ���̶��ɴ�С��˳����____________������a��b��c��ʾ����ͬ��
(2)ͬ�����ͬ���ʵ���Ũ�ȵ������ᣬ�к�N aOH�������ɴ�С��˳����___________��
aOH�������ɴ�С��˳����___________��
(3)������c(H��)��ͬʱ�����ʵ���Ũ���ɴ�С��˳����__________________��
(4)������c(H��)��ͬ�����Ҳ��ͬʱ���ֱ����������п����ͬ״���²������������ɴ�С��˳����________ __________��
__________��
(5)��c(H��)��ͬ�������ͬʱ��ͬʱ������״���ܶȡ�������ȫ��ͬ��п����������ͬ�����H2(��ͬ״��)����Ӧ����ʱ��ij��̹�ϵ��______________________��
(6)��c(H��)��ͬ�����������ˮϡ����ԭ����100���� ��c(H��)�ɴ�С��˳����_________________��
��c(H��)�ɴ�С��˳����_________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012����ױ���������ʯ��ҵһ�и߶���ѧ����ĩ���Ի�ѧ�Ծ����������� ���ͣ�ʵ����
(12��)������һ���¶��µ�ijNaOH��Һ����֪:����Һ���ΪV mL����Һ�ܶ�Ϊd g/cm3����������(�����ٷֱ�Ũ��)Ϊw%�����ʵ���Ũ��Ϊc mol/L����Һ�к��������Ƶ�����Ϊm g. �Իش��������⣮
(1)���㣺��w��d��ʾ��Һ�����ʵ����ʵ���Ũ��cΪ________________��
(2)ijѧ����������ƽ����С�ձ�������(�ձ���ʢNaOH)������ǰ��������ڱ�ߵ���̶ȣ���ƽ��ֹʱ����ָ���ڷֶ��̵�ƫ��λ�ã���ʱ��ߵ����̽�________________(����ڡ����ڡ�)�ұߵ����̣���ʹ��ƽƽ�⣬�����еIJ���Ϊ____________ ________________ ���ٶ����ճ���С�ձ�������Ϊ________________(�32.6 g����32.61 g��)��
(3)�ڱ��(ͼ)�ϻ�������λ��(����������ʾ)��
(4)���ݺ���Һ���ȣ�����ʱ����Һ����ڿ̶��ߣ������ּ�����ˮ���̶��ߣ�������Һ�����ʵ���Ũ�Ƚ�__________________(�ƫ����ƫС������Ӱ�족)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012����ױ����и߶���ѧ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ�ʵ����
(12��)������һ���¶��µ�ijNaOH��Һ����֪:����Һ���ΪV mL����Һ�ܶ�Ϊd g/cm3����������(�����ٷֱ�Ũ��)Ϊw%�����ʵ���Ũ��Ϊc mol/L����Һ�к��������Ƶ�����Ϊm g. �Իش��������⣮
(1)���㣺��w��d��ʾ��Һ�����ʵ����ʵ���Ũ��cΪ________________��
(2)ijѧ����������ƽ����С�ձ�������(�ձ���ʢNaOH)������ǰ��������ڱ�ߵ���̶ȣ���ƽ��ֹʱ����ָ���ڷֶ��̵�ƫ��λ�ã���ʱ��ߵ����̽�________________(����ڡ����ڡ�)�ұߵ����̣���ʹ��ƽƽ�⣬�����еIJ���Ϊ____________ ________________ ���ٶ����ճ���С�ձ�������Ϊ________________(�32.6 g����32.61 g��)��
(3)�ڱ��(ͼ)�ϻ�������λ��(����������ʾ)��

(4)���ݺ���Һ���ȣ�����ʱ����Һ����ڿ̶��ߣ������ּ�����ˮ���̶��ߣ�������Һ�����ʵ���Ũ�Ƚ�__________________(�ƫ����ƫС������Ӱ�족)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012�갲��ʡ��У�����о���߶����ʲ��Ի�ѧ�� ���ͣ�ʵ����
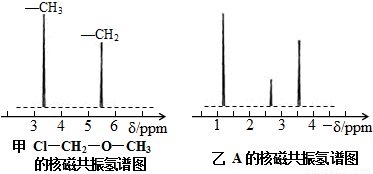
(14��)Ϊ�ⶨij�л�������A�Ľṹ����������ʵ�顣
������ʽ��ȷ����
�Ž��л���A�����������г��ȼ�գ�ʵ���ã�����5.4gH2O��8.8gCO2����������6.72L����״���£���������ʵ�ʵ��ʽ�� ��
�������Dzⶨ�л���A����Է�������Ϊ46��������ʵķ���ʽ�� ��
��Ԥ��A�Ŀ��ܽṹ��д���ṹ��ʽ ��
���ṹʽ��ȷ����
�Ⱥ˴Ź�����ԭ�ӹ����ܶ��л�������в�ͬλ�õ���ԭ�Ӹ�����ͬ�ķ�ֵ���źţ������ݷ�ֵ���źţ�����ȷ����������ԭ�ӵ��������Ŀ�����磺���ȼ��ѣ�Cl�DCH2�DO�DCH3����������ԭ�ӣ�����ͼ�������ⶨ���л���A�ĺ˴Ź�������ͼ������ͼ����A�Ľṹ��ʽΪ ��
������ʵ�顿
��A��һ����������ˮ������B��B�ɺϳɰ�װ����C����д��Bת��ΪC�Ļ�ѧ��Ӧ����ʽ�� ��
�����������е��˶�Ա����Ť��ʱ����ҽ�漴�������飨�е�Ϊ12.27�棩�����˲�λ���оֲ��䶳����������Bѡ����ʵķ����Ʊ������飬Ҫ��ԭ��������Ϊ100%����д���Ʊ���Ӧ����ʽ�� ��
��A��ͨ����ʳ��һ���������Ƶã�����ʳ�Ƶõ�A��һ���¶����ܱմ��棬��Ϊ����һϵ�еĻ�ѧ�仯����ø����㡣��д�����һ����Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com