
��ȸʯ��Ҫ��Cu2(OH)2CO3,��������Fe��Si�Ļ����ʵ�����Կ�ȸʯΪԭ���Ʊ�CuSO4��5H2O��CaCO3���������£�
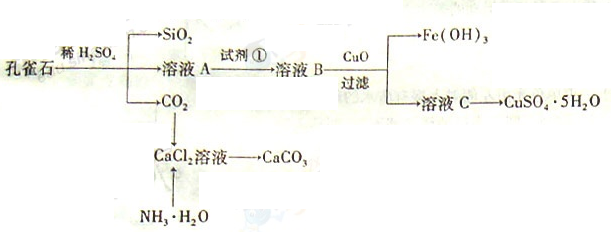
��ش��������⣺
��1����ҺA�Ľ���������Cu2+��Fe2+��Fe3+�������������Լ���ѡ��ʵ�鲽�����Լ���Ϊ______������ţ���������ҺA��Fe3+������Լ�Ϊ��������������ţ���
a��KMnO4����������b��(NH4) 2S c��H2O2 d��KSCN
��2������ҺC���CuSO4��5H2O����Ҫ���������������������� �����˵Ȳ��������ձ���©���⣬���˲������õ���һ�����������������ڴ˲����е���Ҫ�������������� ��
��3���Ʊ�CaCO3ʱ��Ӧ��CaCl2��Һ����ͨ�루���ȼ��룩�������� ���ѧʽ������ʵ��������а����ݳ���Ӧѡ�������������� װ�û��գ�����ţ���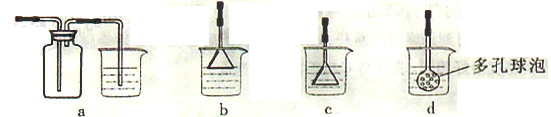
��4�����ⶨ��ҺA��Fe2+��Ũ�ȣ���Ҫ������ƿ����ij����Һ������ʱ����Ӧ ��ֱ�� ����KMnO4����Һ�ζ�ʱӦѡ�����������ζ��ܣ����ʽ����ʽ������
��1�� c (2��) d (2��) ��2����ȴ�ᾧ (2��) ���� (2��)
��3��NH3��H2O (2��) b (2��)
��4 ƽ�Ӱ�Һ�棨��ƽ�ӿ̶��ߣ���1�֣� ��Һ�����͵���̶������� (1��) ��ʽ (1��)
���������������1��Ҫ��ȡ����ͭ���壬�ͱ����ȥ��Һ�е��������ӣ����ó�������ȥ�����ӡ��������Ҫ����Һ�е����������������������ӣ���ͬʱ�ֲ��������µ����ʣ�˫��ˮ�Ļ�ԭ������ˮ�������������ʡ����Դ�ѡC��������ҺA��Fe3��������Լ���KSCN��Һ����2��Ҫ���CuSO4��5H2O����Ҫͨ��������������ȴ�ᾧ��Ȼ����˼��ɡ�����ʱ���ձ���©���⣬����Ҫ��������������3������CO2���ܽ�Ⱥ�С����������ͨ�백����ʱ��Һ�Լ��ԣ�Ȼ����ͨ��CO2���õ�̼��ƹ��塣���ڰ�����������ˮ������������Ҫ������ֹ��������˴�ѡb����4�����ⶨ��ҺA��Fe2+��Ũ�ȣ���Ҫ������ƿ����ij����Һ������ʱ����Ӧƽ�Ӱ�Һ�棨��ƽ�ӿ̶��ߣ���ֱ����Һ�����͵���̶������� ������KMnO4��Һ��ǿ�������ܸ�ʴ�ܣ��ζ�ʱӦѡ����ʽ�ζ��ܡ�
���㣺���⿼���������ӵļ��飬���ʵķ����ᴿ����ѡ������İ�ȫ���ռ��ζ�������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ૼ������ȵõ�����ͪ���������������ϣ��ɵõ��˼������ӿ���ϩ���й�ʵ��ԭ�����������£� 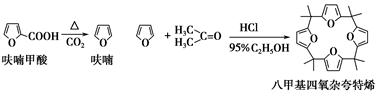
����1����Ʊ�
��Բ����ƿ�з���4.5 gૼ��ᣨ100 ��������ૼ�����133 �����ڣ�230��232 ����ڣ����ڴ��¶������ȣ�����ͼ��װ���������ȴ�����ʹૼ�������ۻ���Ȼ����ڼ���ǿ�ȣ��������У���ૼ������ȷ�Ӧ��ϣ�ֹͣ���ȡ�����ɫҺ��ૣ��е㣺31��32 �棬������ˮ����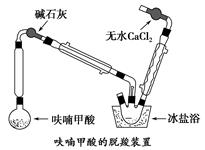
����2��������˼������ӿ���ϩ�ĺϳ�
��25 mL��ƿ�м���2.7 mL 95%�Ҵ���1.35 mLŨ���ᣬ���ȣ��ڱ�ԡ������5 �����£�Ȼ��3.3 mL��ͪ��1.35 mLĻ��ҺѸ�ٵ�����ƿ�У���ֻ��ȣ���ԡ��ȴ�����õ�һ��ɫ��״���塣���ˣ�����3 mL��ˮ�Ҵ�ϴ�ӣ��ñ��ؽᾧ���ð�ɫ�ᾧ�˼������ӿ���ϩ��
��1������1���ô���ټ��ȣ�����ҪĿ����____________________________��
��2��װ��ͼ�м�ʯ�ҵ�������__________________________________________��
��ˮ�Ȼ��Ƶ�������________________________________________________��
��3������װ�����ñ���ԡ��Ŀ����_____________________________________��
��4���ϳɰ˼������ӿ���ϩ���������Ŀ����_________________________��
��5��ȷ�۲�ƷΪ�˼������ӿ���ϩ����ͨ���ⶨ�е㣬���ɲ��õļ�ⷽ����__________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������ˮ�����������������ؾ���K3[Fe(C2O4)3]��3H2O��������Ӱ����ɫӡˢ������мΪԭ�ϵ��Ʊ��������£�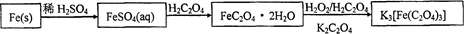
��ش��������⣺
��1����м�г�����Ԫ�أ���ϡ����ʱ������ж���H2S���壬��������������Һ���գ���������װ����ȷ���� ��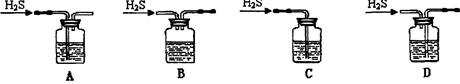
��2���Ƶõ�FeSO4��Һ�������������H2SO4�ữ��Ŀ���� ����Ҫ����Һ�еõ��̷�FeSO4��7H2O��������е�ʵ������� ����˳����д����
a������ϴ�� b������Ũ�� c����ȴ�ᾧ d������ e������
��3���þ�������110�����ȫʧȥ�ᾧˮ�����������¶ȿɷ����ֽⷴӦ��
�ٷֽ�õ����������������װ�ý���ʵ��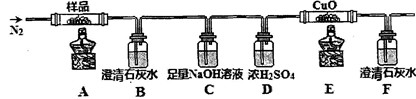
װ�ü�������Ժ���ͨһ��ʱ��N2����Ŀ��Ϊ ������ʵ��ʱ��Ϩ��ƾ�����ͨ��N2�����£���Ŀ��Ϊ ��ʵ������й۲쵽B��F�г���ʯ��ˮ������ǣ�E���к�ɫ�������ɣ������������ ��
�ڷֽ�õ��Ĺ�����ﺬ��K2CO3��FeO��Fe����ˮ�ܽ⡢���ˡ�ϴ�ӡ�����õ�������Ʒ���������������ʵ�鷽���Ը���Ʒ�������ʺ����ⶨ��
���ס�a g��Ʒ ��Һ
��Һ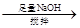
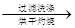 �ù���b g
�ù���b g
���ҡ�a g��Ʒ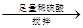
 ��������������Va mL
��������������Va mL
������a g��Ʒ 250 mL��Һ
250 mL��Һ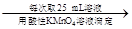 ����ƽ������0.1 mol��L��1����KMnO4��ҺVb mL����Ϊ���Ϸ����� ��ȷ����Ʒ����ɣ������� ��
����ƽ������0.1 mol��L��1����KMnO4��ҺVb mL����Ϊ���Ϸ����� ��ȷ����Ʒ����ɣ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��������������Ԫ��֮һ���С�����Ԫ�ء�֮�ơ�ʳ�üӵ�ʳ�ο�Ԥ����ȱ��������������֪���������������£�I- �ܱ�NO3- ������IO3-����H2O2��O2������ΪI2���� IO3- �ܱ�HSO3- ��ԭ��I2��
��������ʵ���ҳ���������������ѡ�Լ��������о�ijʳ����Ʒ�����ӵ�Ĵ�����ʽ��I2��I-��IO3- �е���һ�֡�
��ѡ�Լ����£�1.0 mol?L-1HNO3��Һ��1.0 mol?L-1H2SO4��Һ��1.0 mol?L-1NaHSO3��Һ��3%H2O2��Һ��1%������Һ������ˮ��
��1���������
����1����ʳ����Ʒ�к�I2��
����2����ʳ����Ʒ�к�I-��
����3�� ��
��2����Ʒ�����ʵ��̽��
������ʳ����Ʒ��������ˮ�Ƴ���Һ���밴Ҫ����д�±���
| ʵ�鲽�� | Ԥ����������� |
| ����1��ȡ����������Һע���Թ��У����뼸�ε�����Һ���� | ����Һ�� ������ɫ���������1������������1���������ٽ��в���2 |
| ����2�� | ����Һ����ɫ�������2��������Ӧ�����ӷ���ʽΪ ��������2���������ٽ��в���3 |
| ����3�� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij�о���ѧϰС����̽���ں�Mg2+��Al3+ �Ļ����Һ�еμ�NaOH��Һʱ������������������Ĺ��̡�
��ʵ�顿��0.1 mol?L��1 MgSO4��0.05 mol?L��1Al2(SO4)3�Ļ����Һ�еμ�0.5 mol?L-1NaOH��Һ�����������Ӵ���������Ӧ���������������Һ��pH��NaOH��Һ�ļ���仯�����ͼ��ʾ��
��1��Ϊ��ȷ���Ƽ���NaOH��Һ��������ɽ�NaOH��Һ���� �����������ƣ��еμӡ�
��2��ͼ��������pH���ӻ����ĽΣ���һ�Σ�a��ǰ����Ӧ��ʵ�������� ��
��3���Եڶ��Σ�b��c֮�䣩��ҺpH�仯�����ı��ʣ�С��ͬѧ���������Ʋ⣬�벹���Ʋ�2��3��
�Ʋ�1������Mg(OH)2����������OH����
�Ʋ�2�� ��
�Ʋ�3�� ��
���Ʋ�1����ʵ���������a��֮ǰ��Ӧ�����ӷ���ʽΪ ����ݴ�����Mg(OH)2��Al(OH)3����������ˮ��Һ���ܽ��ԵIJ��� ��
��4�������e�����Һ�нϴ������ڵĺ�����Ԫ�ص����Ӳ����ʵ����飨�ɲ���������
| �ϴ������ڵĺ�����Ԫ�ص����� | ���鷽�� |
| | |
| | |
| | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����ֲ���纣���������к��зḻ�ĵ�Ԫ�أ���Ԫ���Ե����ӵ���ʽ���ڡ�ʵ������Ӻ�������ȡ����������£�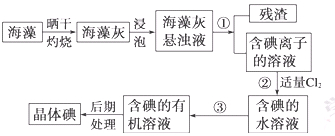
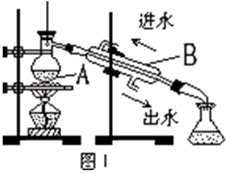
��1��ָ�������۵����ƣ� ���������г�������Cl2��Ŀ���� ��
��2����ȡ��Ĺ����У��ɹ�ѡ����Լ��� ( )
| A���ƾ� | B�����Ȼ�̼ | C������ | D������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��1���������廯�ƺͽ�ŨH2SO4�����Ϊԭ�ϣ���ʵ�����Ʊ�1���嶡�飬�����鷴Ӧ�IJ��ָ��������֪��NaCl+H2SO4(Ũ)=NaHSO4+HCl�������������װ�ã����мг�������������������ȴˮ��û�л�������ش��������⣺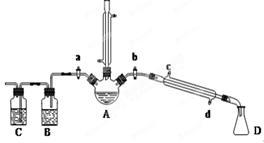
��1������D�������� ��
��2���ر�a��b����ͨ��ֱ�����ܵ�����ˮ����A����30���ӣ��Ʊ�1���嶡�顣д���÷�Ӧ�Ļ�ѧ����ʽ ��
��3�������ϣ�������Ӧ�������ﻹ�����У����ѡ�1����ϩ���廯��ȡ�Ϩ��A���ƾ��ƣ�����ֱ�������Ϸ��������ӣ���a���������ȼ�����Ӧֱ����ȴ��ͨ��B��Cװ�ü��鲿�ָ����B��C��Ӧʢ�ŵ��Լ��ֱ��� �� ��
��4����ʵ������У�����A��Һ������ɫ��ɺ�ɫ���ú�ɫ������Ũ���ᷴӦ�Ļ�ѧ����ʽΪ ��������ֱ�����ܵ��϶�����һ����װ���ռ���ʯ�ҵĸ���ܣ�������Ⱦ������
��5������л�����������£�
| ���� | �۵�/0C | �е�/0C |
| 1������ | -89��5 | 117��3 |
| 1���嶡�� | -112��4 | 101��6 |
| ���� | -95��3 | 142��4 |
| 1����ϩ | -185��3 | -6��5 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ��ȥ�����е�Ca2+��Mg2+��SO42-�Լ���ɳ�����ʣ�ijͬѧ�����һ���Ʊ����ε�ʵ�鷽�����������£����ڳ������Լ��Թ�����
��1���ж�BaCl2�ѹ����ķ����� ��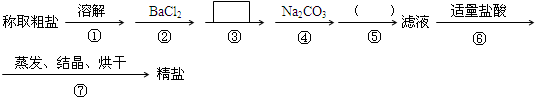
��2���ڢܲ��У�д����Ӧ�����ӷ���ʽ�� �� ____________________________.
��3���� ����д��ʹ�ó����Լ��Ļ�ѧʽ__________���ڣ� ���еIJ���������____ ___��
����д��ʹ�ó����Լ��Ļ�ѧʽ__________���ڣ� ���еIJ���������____ ___��
��4����ʵ����Ʒ����Ż��ĽǶȷ�������ںܿ͢ɷ�ߵ�____________����ǡ������������˵�����ɡ���ԭ���ǣ�_______________________ _______ _________________��
����ۺܿ͢ɷ�ߵ�____________��
��5�������������ٽ��в����ݣ�����ʵ��������Ӱ�죬��ԭ���ǣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��(Ti)����Ϊ21���ͽ�����ұ���ѵ���Ҫԭ���Ǻ�Fe2O3��������FeTiO3)���������������£�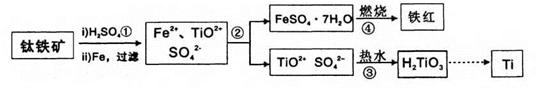
��֪��TiOSO4������ˮ����ˮ�⣬H2TiO3������ˮ���Իش��������⣺
(1)����ڵ���Ҫʵ���������ȴ���ᾧ�� (���������)���������ʵ�����г����Լ����� (���������ƣ��м�ǿ�ȡ�
(2)������м�����ˮ�������� ��
(3)�������������������(FeSO4��7H2O)�ڿ����������������졢ˮ����������д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
(4)��������õ���������������(FeSO4��7H2O)��Ŀǰ��ҵ�ϴ��������ж���ˮ���õĻ�ѧ�Լ������߷�Ӧ��(Cr��+6��ת��Ϊ+3��)����ת��Ϊ����Ҫ��ҵ��ֵ�������帴�����������FeO��FeyCrxO3��ʾ)�����Ʊ��������帴����������������Եĺ�����ˮ�У�����FeSO4��7H2O������ӦΪ��ˮ�����۸����൱��CrO3)������ ����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com