
ij��ѧ��ȤС������ͼһװ����ȡ������̽���������й����ʡ�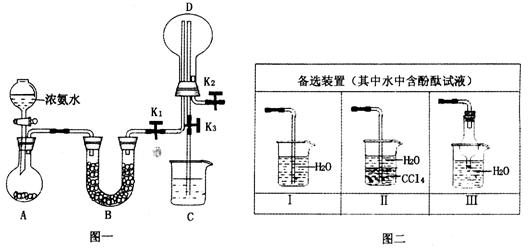
��1��װ��A����ƿ���Լ���ѡ��________������ţ�
a����ʯ�� b��Ũ���� c����ʯ�� d������������ e���ռ�
��2����̽���������ܽ��ԣ�����K2�ĵ���ĩ������ͼ��װ���е�_____װ�ã�����ţ�����װ��D�м��������ر�K1��K2����K3��������Ȫ��ʵ�������______________
___________________________________________________________________________��
��3����̽�������Ļ�ԭ�ԣ����K1��K2��K3������������ȡ����������������װ�á�
���ö���������Ũ������ȡ�����������������ͨ��ʢ��____________�Լ���ϴ��ƿ��
��D�а�����������Ӧ�������̣�ͬʱ����һ����ɫ��ζ�����壬�÷�Ӧ�Ļ�ѧ����ʽΪ___________________________________��
�۴�K3�������ݳ��������к�������Cl2����Cװ����Ӧʢ��_________��Һ���ѧʽ������Ӧ�����ӷ���ʽΪ____________________________________��
��1��ace(2�֣�ȫ�Ե�2�֣���ѡ���÷�)
��2������2�֣�����ë������ƿ����(1��)
��3���ٱ���ʳ��ˮ��Ũ���ᣨ2�֣�
��3Cl2+8NH3=6NH4Cl+N2��2�֣��ֲ�дҲ�ɣ�
��NaOH��1�֣�Cl2+2OH?=Cl?+ClO?+H2O
���������������1����ʯ�ҡ���ʯ�ҡ��ռ�ܴٽ�Ũ��ˮ�а����Ļӷ�����ace��ȷ��
��2�������ܷ�ֹ�����ĵ�������Ϊװ����û�н�ͷ�ιܣ���������ë��������ƿ��ʹ�������ͣ���ˮ�Ӵ��ܽ⣬�γ���Ȫ��
��3������ȡ�������к��Ȼ��⡢ˮ�֣��ñ���ʳ��ˮ�ɳ��Ȼ��⡢��Ũ����ɳ�ˮ�֣��õ�������������������ڲ����İ���Ϊ�Ȼ�泥���ɫ��ζ������Ϊ��������ƽ�ȵ÷���ʽ���������ж�������Ⱦ��������������������Һ����β���е���������ֹ��Ⱦ��
���㣺���⿼�黯ѧʵ����������ͻ��������Լ�ʵ������ͷ���ʽ����д��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧ������ȤС��̽��ͭ��Ũ����ķ�Ӧ�����ijЩ��������ʡ�
(��)�ס�����ͬѧ����������ʵ�飺ȡһ������ͭƬ��20 mL 18 mol/L��Ũ�������Բ����ƿ�й��ȣ�ֱ����Ӧ��ϣ��������ƿ�л���ͭƬʣ�࣬ͬʱ������ѧ��֪ʶ��Ϊ���н϶�����ʣ�ࡣ
��1��ͭ��Ũ���ᷴӦ�Ļ�ѧ����ʽ�ǣ�
����֤���������ʵ�鷽����_ (����ĸ)��
a���ټ�������NaNO3����B���ٵ���BaCl2��Һ C���ټ����� D���ٵ���Na2CO3��Һ
��2����ͬѧ���������Ũ�ȵ�ʵ�鷽���Dzⶨ����������������з����в����е��� (����ĸ)��
a�������������建��ͨ��Ԥ�ȳ�����ʢ�м�ʯ�ҵĸ���ܣ���Ӧ�������ٴγ���
b�������������建��ͨ�����Ը��������Һ���ټ�������BaCl2��Һ�����ˡ�ϴ�ӡ������������
c������ˮ���ⶨ�������������(����ɱ�״��)
d�����ű���NaHSO3��Һ�ķ�������������������(����ɱ�״��)
��3����ͬѧ��Ʋⶨ����Ũ�ȵ�ʵ�鷽���ǣ��ⶨ��Ӧ��Ļ��Һ��Cu2���������ڷ�Ӧ�����Һ�м�������Na2S��Һ����ַ�Ӧ���ˡ�ϴ�ӡ������������������ΪW g����÷�Ӧ����Һ�����ΪV mL����ʣ����������ʵ���Ũ��Ϊ mol/L(�ú�W��V�Ĵ���ʽ��ʾ)��
(��)��ͬѧ̽��SO2��BaCl2��Һ�ܷ�Ӧ���ɰ�ɫBaSO3������
��ͬѧ�Ƚ�����������ͨ��ʢ�б���NaHSO3��Һ��ϴ��ƿ���ٻ���ͨ��BaCl2��Һ�У��۲쵽��������ɫ�������ɣ������ɫ���������ֳ���ȫ��������ϡ���ᣬ�ó��������ɱ���SO2���� �ԡ���һ�����ӷ���ʽ�������ɴ˳�����ԭ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ʵ���ҿ���ͭ��Ũ������Ȼ�������������Ʒ�Ӧ��ȡ�������������������������Ʒ�Ӧ��ȡ��������ϣ���ܿ��Ʒ�Ӧ�ٶȣ���ͼ�п�ѡ�õķ���װ���� ����д��ĸ����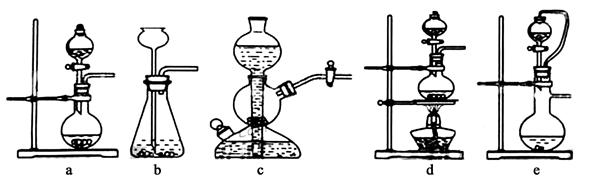
��Aͼ��ʾijѧ����SO2��Ư�۾�[80%Ca(ClO)2��]�ķ�Ӧ����ʵ��̽���Ĺ��̣��۲쵽�������У�
��.Һ���Ϸ����ְ�����
���Ժ��ֻ��ǣ���Һ��Ϊ����ɫ��
���Ժ���������ɫ����������ɫ��ȥ
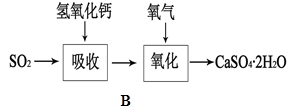
��1����ˮ�г���ͨ��SO2��δ�۲쵽�������Ʋ�������еİ�����HClСҺ���γɣ���������ʵ�飺
a����ʪ��ĵ⻯�ص�����ֽ����������ޱ仯��
b�����ữ��AgNO3��Һ���������������ɫ������
��ʵ��a��b�����жϰ����к���HCl�������� ��
��2�����д�����ɫ�����ijɷ��� ��
��3�����������Һ��Ϊ����ɫ�Ŀ���ԭ������Һ���Ե���ǿ��Ư�۾�����Ч�ɷֺ�C1-������Ӧ������Cl2��ͨ����һ��ʵ��ȷ�������ֿ����ԣ���ʵ�鷽���� ��
��4�������ӷ���ʽ����������л���ɫ��ȥ��ԭ�� ��
��5��Bͼ��ʾʯ��-ʯ�෨����SO2�Ĺ������̣�д����Ӧ�Ļ�ѧ����ʽ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��16�֣����������(Na2S2O3)������Ҫ����������Σ��׳ơ������������������մ�������ˮ���������Ҵ�������֯��Ư�ס������������й㷺Ӧ�á�
ijУ��ѧ�о���ѧϰС���������ѧϰ��˼�룬ͨ��ʵ��̽��Na2S2O3�Ļ�ѧ���ʡ�
����Ʒ�Ʊ���ʵ�����г����������ƺ�����Ʊ�Na2S2O3��5H2O��д����Ӧ�Ļ�ѧ����ʽ ��
��ӦҺ����ɫ�����ˡ�Ũ���ᾧ�����ˡ�ϴ�ӡ����T�ò�Ʒ�����þ������Ҵ�ϴ�ӵ�Ŀ���� ��
��������⡿Na2S2O3�Ƿ���Na2SO4�߱����Ƶ����������أ�
����٣� ��
����ڣ���Һ�����ԣ��Ҳ����ᷴӦ��
����ۣ���ԭ�ԣ����ܱ�������������
������̽����������������ڡ��ۣ����ʵ�鷽����
| | ʵ����� | ʵ������� Ԥ��ʵ������ | ������ͣ��� ���ӷ���ʽ��ʾ�� |
| ����� | ������ֽ�����ɫ������ [��Դ:ѧ*��*��Z*X*X*K] | ��ҺpH=8 | |
| ��pH=2�������� �μ�Na2S2O3��Һ | | 2S2O32- +2H+�T�T S��+SO2��+H2O | |
| ����� | ��������ˮ�еμ�����Na2S2O3��Һ | ��ˮ��ɫ��dz | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����������һ�ִ�����Ⱦ��о���NO2��SO2��CO�ȴ�����Ⱦ����Ĵ�������Ҫ���壬ij��ѧʵ�鰮��С����̽��SO2�����ʣ�������·�����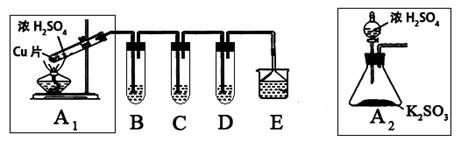
��1��B��C��D�ֱ����ڼ���SO2�Ļ�ԭ�ԡ������Ժ�Ư���ԡ�����B��C�ֱ�Ϊ��ˮ�������ˮ��Һ����D����ʢ�Լ�Ϊ_________��B�з�Ӧ�����ӷ���ʽΪ��_________________��
��2��Ϊ��ʵ����ɫʵ���Ŀ�꣬ijͬѧ���������������ͼA2����ȡװ��������A1װ�ã���A1װ����ȣ�A2װ�õ��ŵ��ǣ�________________________________����дһ�㼴�ɣ���
��3��E���ð�ˮ����β���е�SO2��������Һ���п��ܺ���OH����SO32����SO42����HSO3���������ӡ���֪����������һ��������ˮ��SO2Ҳ������ˮ�������������Լ�Ϊ��С�ձ����Թܡ�����������ͷ�ιܡ�����װ�ú���ֽ��2mol/L���ᡢ1mol/L BaCl2��Һ��1mol/L Ba(OH)2��Һ��Ʒ����Һ������ˮ��
�����ʵ��֤��������Һ���д���SO32����HSO3��������±���ʵ�������Ԥ������ͽ��ۣ�
| ʵ����� | Ԥ����������� |
| ����1��ȡ����������Һ����С�ձ��У��ý�ͷ�ι�ȡ1mol/L BaCl2��Һ��С�ձ��μ�ֱ�������� | �����ְ�ɫ���ǣ�����Һ�д���SO32���� SO42���� |
| ����2����С�ձ��е���Һ���ˡ�ϴ�ӣ���������ˮ�Ѹ�����ֽ�ϵĹ��������һС�ձ��У�����µĹ��� ___________________________________________________ | _________________________ ______________________________________________ |
| ����3��_______ _______________________ ___________________________________________________ | _________________________ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijУ��ѧ��ȤС����ѧϰ�˹����������ʺ�һ���о�����SO2��Ӧ����������������֪������������CO2��Ӧ���������ɣ�����SO2ͨ��������Ʒ�ĩ��Ҳ���������ɡ���ͬѧ��Ϊ��CO2��SO2��Ȼ���������������SO2���н�ǿ�Ļ�ԭ�ԣ�CO2��ԭ�ԣ���Ӧԭ��Ӧ�ò���ͬ�����������ʵ�����̽����
��1��[�������]��һ�����Ĺ������ƹ�����ͨ��������SO2���Է�Ӧ��Ĺ������ɷּ���Ӧԭ��������¼��裺
����һ����Ӧ�������ֻ��Na2SO3��֤��SO2δ��������
���������Ӧ�������ֻ��Na2SO4��֤��SO2��ȫ��������
����������Ӧ�������____________ _____��֤�� ��
��2��[�����о�]
Ϊ��֤�������Ƿ��������ȤС����������о�����������±�������
| ʵ�鲽�裨��Ҫ��д������������̣� | Ԥ�ڵ�ʵ������ͽ��� |
| ȡ��Ӧ��Ĺ�������Թ��У����� | |
| SO2�������ij̶� | V��m1��m2�Ĺ�ϵ |
| ��ȫ������ | V=0 |
| ���ֱ����� | |
| ����� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ��̽��SO2��Na2O2�ķ�Ӧ�Ƿ�������CO2����ͬѧ�������ͼʵ��װ�á�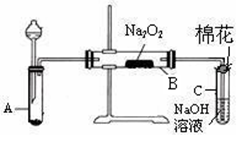
�ش��������⣺
��1����ȡSO2�ĺ����Լ��� ��
a��80%��H2SO4(aq) b��10%��H2SO4(aq)
c��Na2SO3(s) d��Na2SO3��aq��
��1��װ��C��NaOH(aq)�������� ��
��1���ƿ������������ǵ�ľ������C�Թܿڣ�δ��ľ����ȼ����ͬѧ�����ΪSO2��Na2O2�ķ�Ӧ��ͬ��CO2���밴��ͬѧ�Ĺ۵�д����Ӧ�Ļ�ѧ����ʽ ��
��1�����鷴Ӧ��Bװ����������ķ����� ��
��1����ͬѧ��Ϊ���۷�Ӧԭ����Σ����ն���O2��������ͬѧ�������� ��������ͬѧ�Ĺ۵㣬��װ�û��������¸Ľ��� ������Ҫ��������ָ�������װ���������Լ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijУ��ȤС������ͼ��װ����ȡƯ��Һ���������Ѽ��飬�Լ������ӣ������о���������ʡ�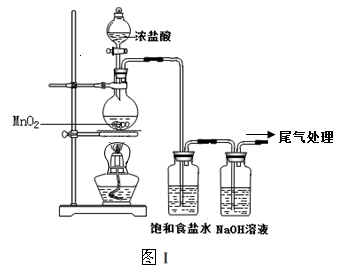

ʵ�����������Һ©���Ļ����������μ�һ����Ũ���ᣬ��ȼ�ƾ��ƣ�һ��ʱ��رշ�Һ©���Ļ�����Ϩ��ƾ��ơ�
��1����ƿ�з�Ӧ�Ļ�ѧ����ʽ�� ��
��2������ʳ��ˮ�������� ��
��3������ͼ��װ���ռ�������������������߿��ڻ�����װ�ü�ͼ��
��4����С��ͬѧ������װ���Ƶõ�Ư��Һ��NaClO��Ũ��ƫ�͡�
�������ϣ�����Һ�з�����Ӧ��
��a��Cl2��g����2NaOH��aq���T NaCl��aq����NaClO��aq����H2O��l��??H1=��101.1kJ/mol
��b��3NaClO��aq���T2NaCl��aq����NaClO3��aq��??H2=��112.2kJ/mol
��Ӧ��a���ķ�Ӧ���ʺܿ죬��Ӧ��b���������½���
�������ϣ����ͼ��װ������Ľ����飺 ��
��5���ķ�����С��ͬѧ�Ƶ��˽ϸ�Ũ�ȵ�NaClO��Һ�����ǰ�Ư��Һ�͵��з�̪�ĺ�ɫNa2SO3��Һ��Ϻõ���ɫ��Һ��
������룺����NaClO��Na2SO3������
����NaClO�ѷ�̪������
����NaClO��Na2SO3�ͷ�̪��������
������ʵ�鷽���п���֤��NaClO������Na2SO3���� ��
a�����Ϻ����Һ�м����������
b�����Ϻ����Һ�м���������ᣬ�ټ����Ȼ�����Һ
c�����Ϻ����Һ�м���������ᣬ�ټ�����������Һ
d�����Ϻ����Һ�м�������������Һ���ټ����������
��Ϊ֤��NaClO�����˷�̪���ɽ��е�ʵ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��֪һ��������A��B��C��D֮���ת����ϵ����ͼ��ʾ������˵����ȷ���ǣ� ��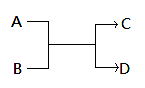
A����AΪFe��DΪ��������Bһ��Ϊ��
B����A��DΪ�����BΪˮ����Cһ�������嵥��
C����A��B��C��D��Ϊ������÷�Ӧһ�����ڸ��ֽⷴӦ
D����A��B��C��D��Ϊ10����������C�ǿ�ʹʪ��ĺ�ɫʯ����ֽ���������壬��D������һ����Һ̬
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com