
��һƿ��ɫ�������Һ�����п��ܺ�Na+��Mg2+��H+��Fe3+��CO32-��Cl-��Br-�е�һ�ֻ��֣�ȡ����Һ��������ʵ�飺
����PH��ֽ���飬������Һ��ǿ����
��ȡ������Һ������������CCl4���������Ƶ���ˮ����CCl4���ԳȺ�ɫ
�۽��ڵõ�����Һ�μ���������Һ���а�ɫ�������ɣ��μ�ϡ����������ܽ⡣
����ȡ������Һ����μ���ϡNaOH��Һ��ʹ��Һ��������ת��Ϊ���ԣ��ڵμӹ����м��μ���Ϻ���Һ�о���������
��������ʵ����ʵȷ�����ش�
��1������Һ�У��϶����ڵ������� ��
��2���϶������ڵ������� ��
��3�����ܴ��ڵ������� ��
��1��H+ Br-
��2��Mg2+ Fe3+��CO32-
��3��Na+ Cl-
���������������1������Һ��ɫ��˵������Fe3+����Һ��ǿ���ԣ�˵����Һ�д���H+���������䷴Ӧ��CO32-�����ڣ��ڵ����Ƶ���ˮ����CCl4���ԳȺ�ɫ��˵����Һ�д���Br-���۲���֤��ԭ��Һ����Cl-����Ϊ�ϲ��е���Һ��ˮ�к������Ӣ���Һ�о���������˵����Һ�в���Mg2+�����Ϸ�������Һ�У��϶����ڵ�������H+ Br-
��2���϶������ڵ�������Mg2+ Fe3+��CO32-
��3�����ܴ��ڵ�������Na+ Cl-
���㣺������Һ�����Ӽ�ķ�Ӧ��������ɫ���ж�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͭ����Ҫ�ɷ��Ƕ�������ͭ��CuFeS2������ͭ�����������պ�õ���ͭ��¯����ұ�����̵���Ҫ��Ӧ�У�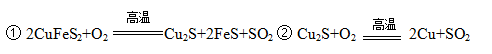
��1����������ͭҲ���Ա�ʾΪCuS��FeS��������Ԫ�صĻ��ϼ��� ��
��2����Ӧ���л�ԭ���� ��
��3��ijУѧϰС������ͭ������¯������Fe2O3��FeO��SiO2��Al2O3�ȣ��Ʊ����죬��������ʵ�顣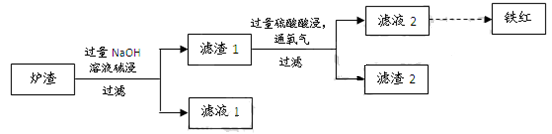
�� ¯�����ʱ��Ӧ�����ӷ���ʽ�� �� ��
�� ����1�м������Ტͨ��������ʹFeOת��ΪFe3+���÷�Ӧ�����ӷ���ʽ�� ��Ϊ������Ԫ���Ƿ�������ȫ��Ӧ���е�ʵ���ǣ�ȡ������Һ2���Թ��� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
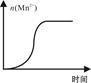
KMnO4������Һ����ᣨH2C2O4����Һ��Ӧʱ����Һ��ɫ������ȥ��ij̽��С���òⶨ�˷�Ӧ��Һ��ɫ��ʧ����ʱ��ķ������о���������Է�Ӧ���ʵ�Ӱ�졣��ʵ����������������
������KMnO4������Һ��Ũ�ȿ�ѡ��0.02 mol��L-1��0.002 mol��L-1��
������H2C2O4��Һ��Ũ�ȿ�ѡ��0.2 mol��L-1��0.4 mol��L-1��
��ÿ��ʵ��ʱKMnO4������Һ��������Ϊ4 mL��H2C2O4��Һ��������Ϊ2mL��
��1����Ҫ̽����Ӧ��Ũ�ȡ��¶ȡ������Է�Ӧ���ʵ�Ӱ�죬ͨ���任��Щʵ��������������Ҫ���____ ��ʵ����жԱȼ��ɵó����ۡ�
��2��������������ͬ������£�ijͬѧ�ı�KMnO4������Һ��Ũ�ȣ����ʵ�����ݣ��ӻ�����ȿ�ʼ��ʱ�����±���ʾ��
| KMnO4������ҺŨ�� ��mol��L-1�� | ��Һ��ɫ����ʱ�䣨min�� | | ||
| ��һ�� | �ڶ��� | ������ | ||
| 0.02 | 14 | 13 | 11 | |
| 0.002 | 6.7 | 6.6 | 6.7 | |
| KMnO4������Һ | H2C2O4��Һ | ||
| Ũ��/ mol/L | ���(ml) | Ũ��/ mol/L | �����ml�� |
| 0.02 | 2 | b | 4 |
| a | 2 | c | 4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij���������̿�MnO2Լ70%��Al2 O3������п��ZnSԼ80%��FeS����ͬ����MnO2����Zn���ɵ��ԭ�ϣ����������£�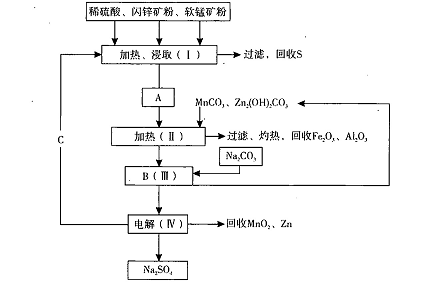
��֪����A��MnSO4��ZnSO4��Fe2(SO4)3��Al2(SO4)3�Ļ��Һ��
��IV�еĵ�ⷴӦʽΪMnSO4+ZnSO4+2H2O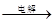 MnO2+Zn +2H2SO4��
MnO2+Zn +2H2SO4��
��1��A�����ڻ�ԭ������� ��
��2������MnCO3��Zn2(OH)2CO3�������� ��C�Ļ�ѧʽ�� ��
��3���������г��õ�Na2SO4��S�ȸ���Ʒ�⣬���ɵõ��ĸ���Ʒ�� ��
��4������ƷS�����������ᣬת�������ǣ�S��SO2��SO3��H2SO4��д���ڶ���ת���Ļ�ѧ����ʽ ��
��5��Ҫ��Na2SO4��Һ�еõ�â���� Na2SO4��10H2O��������еIJ���������Ũ���� ��
���ˡ�ϴ�ӡ�����ȡ�
��6��������MnO2��Zn�ĽǶȼ��㣬���̿����п��Ͷ�ϵ������ȴ�Լ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(����Ԥ����)(1)���ڵ��۵⻯����Һ�У��μ������������Ƽ�����Һ�������ῴ����Һ����ɫ��������Ϊ________�����ӷ���ʽΪ__________________________��
���ڵ�͵����γɵ���ɫ��Һ�У��μ��������Ƽ�����Һ��������ɫ����ʧ��������Ϊ______________________________�����ӷ���ʽ��_______________________________��
�۶ԱȢٺ͢�ʵ�����õĽ������I2��ClO����SO42������ǿ������˳������Ϊ_____________________________��
(2)������Ƭ��ͭƬ�����ʵ��֤��������ʵ����д����Ӧ�Ļ�ѧ����ʽ��
��Ũ����������Ա�ϡ����ǿ��________________________________��
���Ȼ�����Һ��Fe3���������Ա�����ͭ��Һ�е�Cu2��ǿ��__________________________��
�����Ļ�ԭ�Ա�ͭǿ��
___________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ũҩ��Ⱦ�ϡ��ӡ��軯��Լ�����ɫ�ȡ���ζ�ķ�ˮ�����û�ѧ���������д��������õ������������ࣨ��Һ�ȡ�������ơ��������Ƶȣ������ࣨ��������������������⡢������صȣ���һ������ʵ���������������������о綾���軯���CN�����ķ�ˮ���ڼ��������£�pH��8.5��11���������ɽ��軯����CN������Ϊֻ��������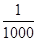 �������Σ���CNO������
����������CNO������
��1��д����CN����ˮ�������������������ε����ӷ���ʽ��________________________________________________________________��
��2������CNO���ķ�ˮ����ͨ����������ʹCNO��ת��Ϊ�������壬д�������Ӧ�����ӷ���ʽ��______________________________________________��
��3������Һ�Ȳ���ĵ���������Ư�۴�����CN���ķ�ˮ������������ΪCNO���������ӷ���ʽΪ__________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��Һ���ܺ���Na����K����Mg2����Cu2���������Ӽ�MnO4����SiO32����AlO2����CO32����HCO3����SO42����Cl���������ӣ���֪���ٸ���Һ����ɫ���ھ��ⶨ��Һ��pH��12����ȡ������Һ������100 mL 2 mol��L��1ϡ��������ữ���а�ɫ�������ɣ����õ�һ����ɫ��ζ�����壬������ʹ����ʯ��ˮ(����)����ǡ����ữ�����Һ���ˣ��õ���Һ�ס�
(1)�ɢ٢ڢۿ��жϣ�ԭ��Һ��һ�������ڵ�������________��һ�����ڵ�������________��
(2)����Һ�ֳ����ȷݣ�һ������μ��백ˮ�������а�ɫ��״������˵��ԭ��Һ��һ����________(�����ӷ���)���տ�ʼ���백ˮʱ��û�г���������ԭ����____________________________________(�����ӷ���ʽ��ʾ)����һ���м���������Ba(NO3)2��Һ���а�ɫ�������ɣ�˵��ԭ��Һ��һ����________(�����ӷ���)�����˵õ���Һ�ҡ�
(3)����Һ���м���������AgNO3��Һ�����ˡ�ϴ�ӡ�����ù���26.5 g����ԭ��Һ���Ƿ���Cl����________(��ǡ���)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪ 0.4 mol Һ̬�º�����H2O2��Ӧ���ɵ�����ˮ����ʱ�ų�256.64 kJ��������
(1)д���º�H2O2��Ӧ���Ȼ�ѧ����ʽ: ��
(2)��֪H2O(l)=H2O(g) ��H="+44" kJ/mol,��16 gҺ̬��������˫��ˮ��Ӧ���ɵ�����Һ̬ˮʱ,�ų��������� ��
(3)������ӦӦ���ڻ���ƽ���,���ͷų����������Ϳ��ٲ�������������,����һ����ͻ�����ŵ��� ��
(4)�����������Һ��ͨ��һ�����ʵ����İ�����������,д����Ӧ�����ӷ���ʽ: ,�÷�Ӧ�Ļ�ԭ������ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ˮ��������ʱ,����˫����(H2Dz,��Ԫ����)�ѽ���������ϳɵ����Ե�����,����CCl4��ȡ�����,�Ӷ��ѽ������Ӵ�ˮ��Һ����ȫ�������������˫����(H2Dz)~CCl4������ˮ�е�Cu2+ʱ,�ȷ�����Ϸ�Ӧ:Cu2++2H2Dz Cu(HDz)2+2H+,�ټ���CCl4,Cu(HDz)2�ͺ����ױ���ȡ��CCl4�С�
Cu(HDz)2+2H+,�ټ���CCl4,Cu(HDz)2�ͺ����ױ���ȡ��CCl4�С�
(1)д��˫�����Fe3+��ϵ����ӷ���ʽ�� ,
��ȡFe3+�Ĺ�����Ҫ�������˵����,�����Һ��pH����,����������������
(2)��ͼ����˫����(H2Dz)~CCl4�����ȡijЩ�������ӵ��������,����ӳ����ȡijЩ��������ʱ���˵�pH��Χ��E%��ʾij�ֽ����������������ʽ��ȡ����İٷ��ʡ�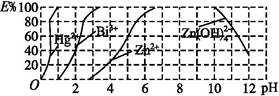
ij��ҵ��ˮ�к���Hg2+��Bi3+��Zn2+,��˫����(H2Dz)~CCl4�����ȡ��������ˮ��
������ȫ����ˮ�е�Hg2+�������,�������Һ��pH=����������
�ڵ�����pH=2ʱ,��(Bi)�Ĵ�����ʽ����������,�����ʵ���֮��Ϊ����������
����ȡ��CCl4�е�Zn(HDz)2��Һ��,��������NaOH��Һ,�����,п��ת��ˮ��Һ�С�д����Ӧ�����ӷ���ʽ:
(3)��ˮ�е��ǹ�����(H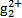 )����ת���ɹ�����(Hg2+)������˫������ϡ�ij������ˮ�к��н϶���Ȼ��ǹ�(Hg2Cl2),����������(K2S2O8)������H
)����ת���ɹ�����(Hg2+)������˫������ϡ�ij������ˮ�к��н϶���Ȼ��ǹ�(Hg2Cl2),����������(K2S2O8)������H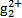 �������ṯ,д���÷�Ӧ�Ļ�ѧ����ʽ:����
�������ṯ,д���÷�Ӧ�Ļ�ѧ����ʽ:����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com