
 ������ʽ��C10H12��������Ϊ��ɫҺ�壬�д̼�����ζ���е�207�棬������ˮ����һ���������ܼ�������Һ�巢����Ӧ��C10H12+4Br2��C10H8Br4+4HBr�����ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�
������ʽ��C10H12��������Ϊ��ɫҺ�壬�д̼�����ζ���е�207�棬������ˮ����һ���������ܼ�������Һ�巢����Ӧ��C10H12+4Br2��C10H8Br4+4HBr�����ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�
| �� |
| �� |
| �� |
| �� |


 ȫ�ܲ����ĩС״Ԫϵ�д�
ȫ�ܲ����ĩС״Ԫϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
![]()
���ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�
�ٰ�һ���������������⻯����ˮ�����ʵ��������У��������������ۣ�
����������Һ�壬���Ͻ��裬ֱ����Ӧ��ȫ��
��ȡ�·�Ӧ�����������������⻯����ֱ����Һ��ɫ��ʧ�����ˣ�����Һ�����Һ©�������ã�
�ܷ�Һ���õ��ġ�ˮ�㡱����������Һ��
�ش��������⡣
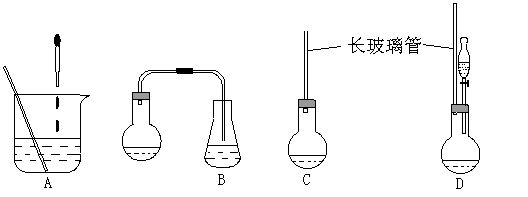
(1)��ͼ�е�װ�ã��ʺϲ���ٺ͢ڲ�������___________��

(2)�����������жϡ���Ӧ��ȫ�� _____________��
(3)������в����������⻯����Ŀ����_________________________________��
(4)������й��˺�õ��Ĺ���������_____________��
(5)��֪��ʵ�������£�������������Һ�������������������66%������廯��Ӧ������ȫ������������⻯����ˮ��������ԼΪ1��________(����С�����1λ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010���Ĵ��ɶ����и߶���ѧ�����л�ѧ�Ծ� ���ͣ������
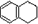
1��2��3��4�����⻯���Ľṹ��ʽ��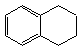 ������ʽ��C10H12��������Ϊ��ɫҺ�壬�д̼�����ζ���е�207�棬������ˮ����һ���������ܼ�������Һ�巢����Ӧ��C10H12+4Br2
������ʽ��C10H12��������Ϊ��ɫҺ�壬�д̼�����ζ���е�207�棬������ˮ����һ���������ܼ�������Һ�巢����Ӧ��C10H12+4Br2 C10H8Br4+4HBr�����ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�
C10H8Br4+4HBr�����ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�


�ٰ�һ�������Ȱ����⻯����ˮ�����ʵ��������У��������������ۡ�
����������Һ�壬���Ͻ��裬ֱ����Ӧ��ȫ��
��ȡ�·�Ӧ�����������������⻯����ֱ����Һ��ɫ��ʧ�����ˣ�����Һ�����Һ©�������á�
�ܷ�Һ���õ��ġ�ˮ�㡱����������Һ��
�ش��������⣺
��1������ʾ��ͼ�е�װ�ã��ʺϲ���ٺ͢ڲ������� ��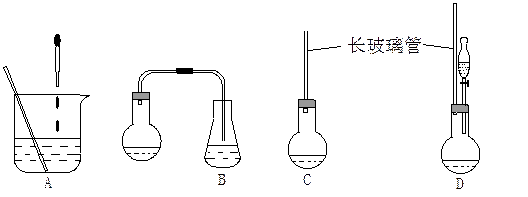
��2�������������жϡ���Ӧ��ȫ�� ��

 ��3��������в����������⻯����Ŀ���� ��
��3��������в����������⻯����Ŀ���� ��
��4��������й��˺�õ��Ĺ��������� ��
��5����֪��ʵ�������£�����������ˮ��Һ�������������������66%������廯��
Ӧ������ɣ�����������⻯����ˮ��������Լ��1�U ������С�����1λ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
1��2��3��4�����⻯���Ľṹ��ʽ��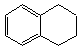 ������ʽ��C10H12��������Ϊ��ɫҺ�壬�д̼�����ζ���е�207�棬������ˮ����һ���������ܼ�������Һ�巢����Ӧ��C10H12+4Br2
������ʽ��C10H12��������Ϊ��ɫҺ�壬�д̼�����ζ���е�207�棬������ˮ����һ���������ܼ�������Һ�巢����Ӧ��C10H12+4Br2![]() C10H8Br4+4HBr�����ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�
C10H8Br4+4HBr�����ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�
�ٰ�һ�������Ȱ����⻯����ˮ�����ʵ��������У��������������ۡ�
����������Һ�壬���Ͻ��裬ֱ����Ӧ��ȫ��
��ȡ�·�Ӧ�����������������⻯����ֱ����Һ��ɫ��ʧ�����ˣ�����Һ�����Һ©�������á�
�ܷ�Һ���õ��ġ�ˮ�㡱����������Һ��
�ش��������⣺
��1������ʾ��ͼ�е�װ�ã��ʺϲ���ٺ͢ڲ������������� ��������������������
��2�������������жϡ���Ӧ��ȫ�������������������������������������� ������ ��
��3��������в����������⻯����Ŀ������������������������������������ ����
��4��������й��˺�õ��Ĺ��������������������������������������������� ��
��5����֪��ʵ�������£�����������ˮ��Һ�������������������66%������廯��
Ӧ������ɣ�����������⻯����ˮ��������Լ��1�U������ �� ������С�����1λ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
1��2��3��4�����⻯���Ľṹ��ʽ��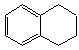 ������ʽ��C10H12��������Ϊ��ɫҺ�壬�д̼�����ζ���е�207�棬������ˮ����һ���������ܼ�������Һ�巢����Ӧ��C10H12+4Br2
������ʽ��C10H12��������Ϊ��ɫҺ�壬�д̼�����ζ���е�207�棬������ˮ����һ���������ܼ�������Һ�巢����Ӧ��C10H12+4Br2![]() C10H8Br4+4HBr�����ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�
C10H8Br4+4HBr�����ɵ����廯��������Ϊ��̬��������ˮ�����������⻯����Һ�塢����ˮ�ʹ�����Ϊԭ�ϣ��Ʊ�����������������Һ��ʵ�鲽�����£�
�ٰ�һ�������Ȱ����⻯����ˮ�����ʵ��������У��������������ۡ�
����������Һ�壬���Ͻ��裬ֱ����Ӧ��ȫ��
��ȡ�·�Ӧ�����������������⻯����ֱ����Һ��ɫ��ʧ�����ˣ�����Һ�����Һ©�������á�
�ܷ�Һ���õ��ġ�ˮ�㡱����������Һ��
�ش��������⣺
��1������ʾ��ͼ�е�װ�ã��ʺϲ���ٺ͢ڲ������� ��
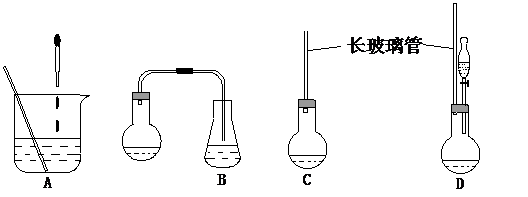
��2�������������жϡ���Ӧ��ȫ�� ����3��������в����������⻯����Ŀ���� ��
��4��������й��˺�õ��Ĺ��������� ��
��5����֪��ʵ�������£�����������ˮ��Һ�������������������66%������廯��
Ӧ������ɣ�����������⻯����ˮ��������Լ��1�U ������С�����1λ����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com