
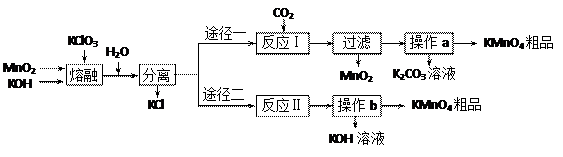
 ��֪����3MnO2+KClO3+6KOH
��֪����3MnO2+KClO3+6KOH 3K2MnO4+KCl+3H2O
3K2MnO4+KCl+3H2O| �¶� | �ܽ��/g | ||
| K2CO3 | KOH | KMnO4 | |
| 20�� | 111 | 112 | 6.38 |
| 60�� | 127 | 154 | 22.1 |
| A�������� | B���ձ� | C���ƾ��� | D�������� |
 �Ʒ�Ӧ�������������뻹ԭ��������ʵ���֮��Ϊ �� ���÷�Ӧ�п���ѭ�����õIJ����� �� ��
�Ʒ�Ӧ�������������뻹ԭ��������ʵ���֮��Ϊ �� ���÷�Ӧ�п���ѭ�����õIJ����� �� ��
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ����ʯ��ˮ��ˮ������� | 1:0 | 1:1 | 1:2 | 1:3 | 1:5 | 1:7 |
| ������CaCO3���������g/100ˮ�� | A | 0.110 | 0.073 | 0.055 | 0.037 | 0.028 |
| ����ѧ�¶�/K | 282 | 298 | 308 |
| CaCO3�ܽ�ȣ�g/100ˮ�� | 0.130 | 0.094 | 0.076 5 |
| P(CO2)/Pa | 0 | 1.40��104 | 9.95��104 |
| CaCO3�ܽ�ȣ�g/100ˮ�� | 0.001 3 | 0.023 3 | 0.108 6 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ��� | �缫���� | �������Һ | ����ָ��ƫת���� |
| 1 | Mg Al | ϡ���� | ƫ��Al |
| 2 | Al Cu | ϡ���� | ƫ��Cu |
| 3 | Al ʯī | ϡ���� | ƫ��ʯī |
| 4 | Mg Al | NaOH��Һ | ƫ��Mg |
| 5 | Al Zn | Ũ���� | ƫ��Al |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 ����������ʵ��̽����
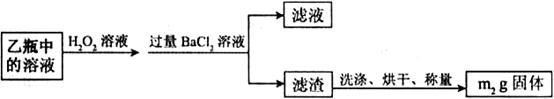
����������ʵ��̽���� g�û�������Ʒ�������в������������������ͼ��ʾװ�ã��гֺͼ���װ��ʡ�ԣ���ʯӢ���У���a�����ϵػ���ͨ�˿�������������ʯӢ���еĻ�������Ʒ����Ӧ��ȫ��ʯӢ���з�����Ӧ�Ļ�ѧ����ʽΪ��
g�û�������Ʒ�������в������������������ͼ��ʾװ�ã��гֺͼ���װ��ʡ�ԣ���ʯӢ���У���a�����ϵػ���ͨ�˿�������������ʯӢ���еĻ�������Ʒ����Ӧ��ȫ��ʯӢ���з�����Ӧ�Ļ�ѧ����ʽΪ��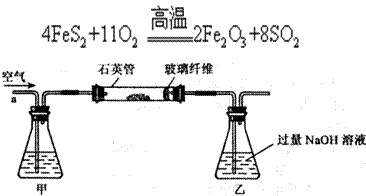
 O
O ��Һ�������������ǣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
��Һ�������������ǣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�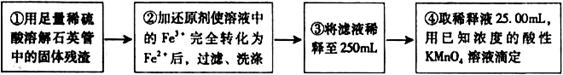
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| A��1��0 mol��L-1HNO3 | B��1��0 mol��L-1���� | C��1��0 mol��L-1 NaOH | D��3%H2O2��E���� 0��1mol��L-1KI��F���� 0��1 mol��L-1CuSO4��G���� 20��KSCN��H��������ʯ��ˮ��I��������ͭ��J��������ˮ��K���� |
| ʵ�鲽�� | Ԥ����������� |
| ����1��ȡ��������������Թ��У�������������ˮ�������ʹ̼�����ȫ�ܽ⡣���벻�ܹ�������Һ��������ˮ���ϴ�Ӳ��ܹ��� | ������ﲿ���ܽ� |
| ����2�����Թ��м�����������ͭ��Һ���ټ�������������1�еIJ��ܹ��壬����� | ��1������ɫ��Һ��ɫ������IJ��ܹ��������Ա仯������� ������ ��2������ɫ��Һ��ɫ���Ըı䣬���а���ɫ�������ɣ���֤���� ���ڡ� |
| ����3����������2�е�(2)�����й�Һ���룬������ˮϴ�ӹ�����ϴ��Һ��ɫ��ȡ�����������Թ��У��μӹ���HCl�����ã�ȡ�ϲ���Һ���μ�����H2O2�������μ�KSCN | ��ϲ���2�еģ�2���� ��1������Һ������ɫ������裨1������ ��2���� ������� ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����������ˮ�к���Cl�� | B�����鼦�����к���̼���� |
| C������ӵ�ʳ���мӵIJ��ǵⵥ�� | D����ȥ��ˮƿ�е�ˮ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
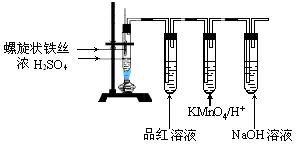
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com