

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 9N2(g)��6CO2(g)��6H2O(g)��
9N2(g)��6CO2(g)��6H2O(g)�� 9N2(g)��12CO2(g)��12H2O(g)��
9N2(g)��12CO2(g)��12H2O(g)��
| A��������� | B�������¶� |
| C�������H2O(g) | D������ѹǿ |
 O2��Hb��CO K��220
O2��Hb��CO K��220�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

 CH3OH(g)��H����90.7 kJ/mol ��
CH3OH(g)��H����90.7 kJ/mol �� CH3OCH3(g)��H2O(g)��H����23.5 kJ/mol ��
CH3OCH3(g)��H2O(g)��H����23.5 kJ/mol �� CO2(g)��H2(g)��H����41.2 kJ/mol ��
CO2(g)��H2(g)��H����41.2 kJ/mol �� O2(g)
O2(g)  CO(g)��2H2(g) ��H����35.6 kJ/mol���÷�Ӧ�� ��Ӧ(��Է������Է���)��
CO(g)��2H2(g) ��H����35.6 kJ/mol���÷�Ӧ�� ��Ӧ(��Է������Է���)�� CH3OCH3(g)��CO2(g)�Ħ�H�� ��830��ʱ��Ӧ�۵�K��1.0�����ڴ���Ӧ���з�Ӧ�۵�K 1.0(�>������<������)��
CH3OCH3(g)��CO2(g)�Ħ�H�� ��830��ʱ��Ӧ�۵�K��1.0�����ڴ���Ӧ���з�Ӧ�۵�K 1.0(�>������<������)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| A��;��II��ˮú��ʱ�����ܺģ���;��II����������ȡ |
| B����;��I��ȣ�;��II���Լ��ٶԻ�������Ⱦ |
| C����;��I��ȣ�;��II�������ú��ȼ��Ч�� |
| D����úת��Ϊˮú������ͨ���ܵ��������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��4a��4b��4c | B��2a��2b��6c |
| C��2a��2b��2c | D��2a��6b��2c |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 CH3OH(g)����H1
CH3OH(g)����H1 CH3OH(g)��H2O(g)����H2
CH3OH(g)��H2O(g)����H2| �¶� | 250 �� | 300 �� | 350 �� |
| K | 2.041 | 0.270 | 0.012 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
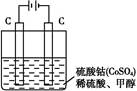

�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com