
| ʵ����� | �� | �� | �� |
| �Ͻ�����/mg | 255 | 385 | 459 |
| �����������/mL | 280 | 336 | 336 |
| V |
| Vm |
| 336mL |
| 280mL |
| 0.336L |
| 22.4L/mol |
| 0.03mol |
| 0.03L |
| 0.28L |
| 22.4L/mol |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��������۲����� |
| B�����岻��ͨ����ֽ |
| C���������Ӳ�ͣ���������˶� |
| D�����岻�ȶ�����ֹ�������γɳ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
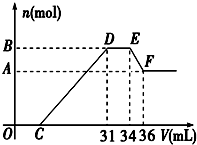 ��֪�������������������ԭ��Ӧ��ʱ��һ������Ũ��Խϡ����Ӧ�Ļ�ԭ�����е��Ļ��ϼ�Խ�ͣ�����һ�������������Ͻ���һ����ϡHNO3��ַ�Ӧ����Ӧ���������κ�����ų����ڷ�Ӧ���������Һ�У���μ���4mol/L NaOH��Һ������NaOH��Һ�������V����������������ʵ�����n����ϵ��ͼ��ʾ����
��֪�������������������ԭ��Ӧ��ʱ��һ������Ũ��Խϡ����Ӧ�Ļ�ԭ�����е��Ļ��ϼ�Խ�ͣ�����һ�������������Ͻ���һ����ϡHNO3��ַ�Ӧ����Ӧ���������κ�����ų����ڷ�Ӧ���������Һ�У���μ���4mol/L NaOH��Һ������NaOH��Һ�������V����������������ʵ�����n����ϵ��ͼ��ʾ�����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| Ԫ�� ���� | I1 | I2 | I3 | I4 |
| Q | 2080 | 4000 | 6100 | 9400 |
| R | 500 | 4600 | 6900 | 9500 |
| S | 740 | 1500 | 7700 | 10500 |
| T | 580 | 1800 | 2700 | 11600 |
| U | 420 | 3100 | 4400 | 5900 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ijУ�о���ѧϰС���ͬѧѧϰ�굪���й����ʵ�����֮�Ե�Ԫ�ص��⻯�������������ʽ����˸�������о���
ijУ�о���ѧϰС���ͬѧѧϰ�굪���й����ʵ�����֮�Ե�Ԫ�ص��⻯�������������ʽ����˸�������о���
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A���٢ڢۢ� | B���٢ڢ� |
| C���٢� | D���ڢۢ� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com