
X��Y��Z��W��Ԫ�����ڱ�ǰ�������еij���Ԫ�أ��������Ϣ���±���
| Ԫ�� | �� �� �� Ϣ |
| X | X����������ˮ��������̬�⻯������γ�һ���� |
| Y | ���������õİ뵼����ϣ��㷺Ӧ���ڹ����Ϣ���� |
| Z | Z��һ�ֺ���������Ϊ27��������Ϊ14 |
| W | ����������Ӧ��ˮ������һ�ֲ�����ˮ����ɫ���� |
��1���� ����A �� �� �� ��
��2��1s22s22p63s23p63d104s1 ��[Ar]3d104s1 ��NH3���Ӽ���������
��3��ClO��+ 2NH3 �� N2H4 + Cl��+ H2O��
��4��2Cu2O(s)+ O2(g)=4CuO(s) ��H = -292.0KJ��mol-1 ��
����������������Ƚ���Ԫ���ƶϡ�X������������ˮ��������̬�⻯������γ�һ���Σ�˵��X�Ƿǽ���Ԫ�أ�������������ˮ�������ᣬ��̬�⻯���ˮ��Һ�Լ��ԡ���X��NԪ�أ���Ӧ����ΪNH4NO3�����������õİ뵼����ϣ��㷺Ӧ���ڹ����Ϣ�����Ԫ����Si��Y��Si��Z��һ�ֺ���������Ϊ27��������Ϊ14����������Ϊ27-14=13.��AlԪ�ء�W������������Ӧ��ˮ������һ�ֲ�����ˮ����ɫ���壬��W��Cu����1��Al�ĺ�������Ų�Ϊ2��8��3��������Ԫ�����ڱ���λ�ڵ������ڵ�IIIA��Al��Si���ǵ������ڵ�Ԫ�أ�����ԭ������������ԭ�Ӱ뾶��С������ԭ�Ӱ뾶Al>Si����Ԫ�صĺ�������Ų�ʽΪ1s22s22p3,2p������ڰ�������ȶ�״̬��ʧȥ�����ѣ���SiԪ�صĺ�������Ų�ʽΪ1s22s22p63s23p2, 3p��������ȶ���״̬���Ƚ�����ʧȥ���ӣ�����ʧȥ���ӽ����ס�X�ĵ�һ�����ܱ�Y�Ĵ�2��29��Ԫ��Cu��̬ԭ�ӵĺ�������Ų�ʽΪ1s22s22p63s23p63d104s1 ��[Ar]3d104s1��NH3���ɷ��ӹ��ɵķ��Ӿ��壬�ڷ���֮����˴���һ��ķ��Ӽ����������������������˷���֮�������ã����Կ˷����Ӽ�������ʹ�����ۻ���������Ҫ�������ϸߡ����۵㡢�е�ϸߡ���3��ʹNaClO��Һ��NH3��Ӧ��N2H4�Ļ�ѧ����ʽΪ��NaClO + 2NH3 �� N2H4 + NaCl+ H2O������д�����ӷ���ʽΪClO�� + 2NH3 �� N2H4 + Cl�� + H2O����4���ڡ�2-�ٿɵã�2Cu2O(s)+ O2(g)=4CuO(s) ��H =" -292.0KJ/mol" ��
���㣺����Ԫ�ص��ƶϡ�Ԫ�����ڱ���Ԫ�������ɡ�ԭ�ӽṹ������ṹ�����ӷ���ʽ���Ȼ�ѧ����ʽ����д��֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij�̬������W��X��Y��Z 4�ֶ�����Ԫ����ɣ�����W��ԭ�Ӱ뾶��С��
��.��Y��Zͬ���壬ZY2���γ��������Ҫ����֮һ��
��1����X��Y��Z��Ԫ�ط���������ͼ��ʾԪ�����ڱ����ֲ����е���Ӧλ���ϡ�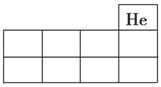
��2��X������������Ӧˮ�����ϡ��Һ��ͭ��Ӧ�Ļ�ѧ����ʽΪ ��
��3��һ�������£�1 mol XW3������O2��ȫ��Ӧ����XԪ�صĵ��ʺ�Һ̬ˮ���ų�382.8 kJ�������÷�Ӧ���Ȼ�ѧ����ʽΪ __��
��.��Z���γɻ�������������Ԫ�ء�
��4���õ��ʵ������� __����һ�֣���
��5��HR�Ǻ�ZԪ�ص�һԪ�ᡣ����ʱ����0.250 mol��L��1NaOH��Һ�ζ�25.0 mL HR��Һʱ����Һ��pH�仯�����ͼ��ʾ��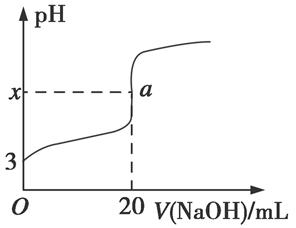
���У�a���ʾ��������ǡ����ȫ��Ӧ��
��ͼ��x �������������������7��
������ʱ��HR�ĵ��볣��Ka�� ������ֵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ѧ��ѧ����Ԫ��ԭ�ӽṹ�����������ʾ��
| ��� | Ԫ�� | �ṹ������ |
| �� | A | A�����������г������������������Ȼ����Է����������35.5 |
| �� | B | Bԭ���������������ڲ���������� |
| �� | C | C�dz������ʵ���ҪԪ�أ����ʳ����³���̬ |
| �� | D | D���ʱ���Ϊ����Ϣ�����Ĵ��������dz��õİ뵼����� |
| �� | E | ͨ������£�Eû�������ϼۣ�A��B��C��D��F������E�γɻ����� |
| �� | F | F�����ڱ��п������ڢ�A�壬Ҳ����������ڢ�A�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��Y��Z��WΪ���ֶ�����Ԫ�ء�Xԭ�������������Ǻ�����Ӳ�����3����Y�������������۵Ĵ�����Ϊ6��Z�ڵؿ��еĺ�������������Y��Z��Wͬ���ڣ�W�ij������ϼ�Ϊ+3���ش��������⣺
(1)Y-�Ľṹʾ��ͼΪ ��
(2)д������10�����ӵ�Xԭ�ӵ�ԭ�ӷ��� ��
(3)Z�������������Zͬ����ĵ���A�ܷ����û���Ӧ�������Ӧ�ڹ�ҵ���ǻ��Z��һ����Ҫ��Ӧ��д���ò���Ӧ�Ļ�ѧ����ʽ�����õ����ŷ��������ת�Ƶķ�������Ŀ ��
(4)��WY3��Һ�м���������NaOH��Һ����Ӧ�����ӷ���ʽΪ ��
(5)Y����̬�⻯���백���ڿ����������а������ɣ��÷�Ӧ�Ļ�ѧ����ʽΪ ��
(6)ZX2�д��ڵĻ�ѧ��Ϊ (����Ӽ������ۼ���)��ZX2��NaOH��Һ��Ӧ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ǰ������ԭ���������������Ԫ��A��B��C��D�У�A��B�ļ۵��Ӳ���δ�ɶԵ��Ӿ�ֻ��1��������A����B+�ĵ������Ϊ8����Bλ��ͬһ���ڵ�C��D�����Ǽ۵��Ӳ��е�δ�ɶԵ������ֱ�Ϊ4��2����ԭ���������Ϊ2���ش��������⣺
��1��D2+�ļ۲�����Ų�ͼΪ_______________��
��2������Ԫ���е�һ������С����_________________���縺��������________________������Ԫ�ط��ţ�
��3��A��B��D����Ԫ����ɵ�һ��������ľ�����ͼ��ʾ��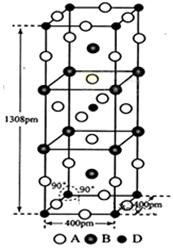
�ٸû�����Ļ�ѧʽΪ_____________��D����λ��Ϊ_______��
����ʽ����þ�����ܶ�Ϊ___________g��cm-3������ʽ�����㣩
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�輰�仯����㷺Ӧ����̫���ܵ����á����ά�������Ʊ��ȡ�
�����Ĺ��Ǵ���Ȼ���е�ʯӢ��ʯ����Ҫ�ɷ�ΪSiO2������ȡ�ġ���������ȡ���������·�Ӧ������1����
��SiO2(s)+2C(s) Si(s)+2CO(g)
Si(s)+2CO(g)
��Si(s)+2Cl2(g) SiCl4(g)
SiCl4(g)
��SiCl4(g)+2H2(g) ��Si(s)+4HCl(g)
���������գ�
��1����ԭ�Ӻ����� �ֲ�ͬ�ܼ��ĵ��ӣ�������p������ ����������
��2������̼ͬ���壬���ʵĻ�ԭ�ԣ�̼ �裨��д��ͬ�ڡ�����ǿ�ڡ������ڡ�������Ӧ��֮�����ܽ��е�ԭ���� ��
��3����Ӧ�����ɵĻ�����ĵ���ʽΪ ���÷���Ϊ ���ӣ���д�����ԡ��Ǽ��ԡ�����
��4��ij�¶��£���Ӧ�����ݻ�ΪV�����ܱ������н��У��ﵽƽ��ʱCl2��Ũ��Ϊa mol/L��Ȼ��Ѹ����С�����ݻ���0.5V����t������´ﵽƽ�⣬Cl2��Ũ��Ϊb mol/L����a b����д�����ڡ��������ڡ���С�ڡ�����
��5����t���ڣ���Ӧ����v(SiCl4)= ���ú�a��b�Ĵ���ʽ��ʾ����
��6����ҵ�ϻ�����ͨ�����·�Ӧ��ȡ���裨����2����
��Si(��) ��3HCl(g)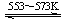 SiHCl3(l)��H2(g) + Q��Q��0��
SiHCl3(l)��H2(g) + Q��Q��0��
��SiHCl3(g)��H2(g) Si(��)��3HCl(g)
Si(��)��3HCl(g)
��߷�Ӧ����Si(��)�IJ��ʣ��ɲ�ȡ�Ĵ�ʩ�У� ��ѡ��2������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��C��D�Ƕ����ڵ�����Ԫ�أ�ԭ��������������Aԭ�ӵ����������4�����ӣ�B�������Ӻ�C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����Ԫ�صĵ��ʷ�Ӧ��������һ�ֵ���ɫ�Ĺ���E��D��L�����������K��M�������Ӳ��ϵĵ�����֮�͡��Իش�
��1��Aԭ�ӵĵ���ʽΪ ��BԪ�����γɵ�����ͬ������������ʵĻ�ѧʽΪ ��CԪ�ص����ӽṹʾ��ͼΪ ��
��2��������E�������еĻ�ѧ���� �������� �����
��3��д��A��B��Ԫ���γɵĻ�������E��Ӧ�Ļ�ѧ����ʽ�� ��
��4��д��D������������ˮ�����A���ʷ�Ӧ�Ļ�ѧ����ʽ�� ��
�˷�Ӧ�����������뻹ԭ��������ʵ���֮��Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������������Ԫ�صĵ�һ�����ĵ���������(��λ�� kJ��mol��1)���ش����и��⣺
| Ԫ�ش��� | I1 | I2 | I3 | I4 |
| Q | 2 080 | 4 000 | 6 100 | 9 400 |
| R | 496 | 4 562 | 6 912 | 9 543 |
| S | 738 | 1 451 | 7 733 | 10 540 |
| T | 578 | 1 817 | 2 745 | 11 575 |
| U | 420 | 3 100 | 4 400 | 5 900 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��Y��Z��M��Q��G���ֶ�����Ԫ�أ�ԭ��������������X��Zͬ���壬���γ����ӻ�����ZX��Y��Mͬ���壬Y��һ��ͬλ��ԭ�ӳ����ڲⶨ����������Q�γɵĵ���Ϊ����ɫ���塣��ش��������⣨�漰���ʾ��û�ѧʽ��ʾ����
��1�����ӻ�����ZX��X���ӵĽṹʾ��ͼΪ �� Y��Ԫ�����ڱ��е�λ����_______________��
��2������Ԫ�ص�����������Ӧ��ˮ����������ǿ����_______________��Q��G����̬�⻯�ﻹԭ�Ը�ǿ����__________________��
��3����ҵ���Ʊ�M�ĸߴ��ȵ��ʣ�����һ����Ҫ��Ӧ�ǣ���MXG3��X2�ڸ����·�Ӧ���÷�Ӧ���̱��������ˮ��������ΪMXG3��ˮ���ҷ�Ӧ����H2�� �� ������������������ĺ���� ��
��4��X2Q��ȼ����Ϊa kJ·mol-1������X2Qȼ�շ�Ӧ���Ȼ�ѧ����ʽ��ȷ���� ��
| A��2X2Q(g) + O2(g) =" 2Q(s)" + 2X2O(g)��H=" -2a" kJ·mol-1 |
| B��X2Q(g) + 2O2(g) = QO3(g) + X2O(l)��H=" +a" kJ·mol-1 |
| C��2X2Q(g)+ 3O2(g) = 2QO2(g) + 2X2O(l)��H=" -2a" kJ·mol-1 |
| D��X2Q(g) + 2O2(g) = QO3(g) + X2O(l)��H=" -a" kJ·mol-1 |
 Fe + 2ZG �ŵ�ʱ����ص�������ӦʽΪ: �õ�صĵ����Ϊ___________________��
Fe + 2ZG �ŵ�ʱ����ص�������ӦʽΪ: �õ�صĵ����Ϊ___________________��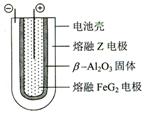
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com