
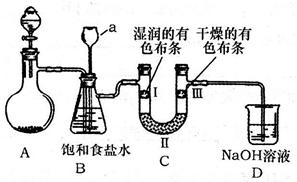
ij��ȤС���������ͼʵ��װ�ý���ʵ�顣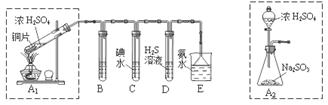
��̽��������Ⱦ��SO2������
��1��Ϊ��ʵ����ɫ������Ŀ�꣬�ܷ�����ͼA2����A1װ�� ����ܡ�����
��2��B��C��D�ֱ����ڼ���SO2��Ư���ԡ���ԭ�Ժ������ԣ���B����ʢ�Լ�Ϊ ��C�з�Ӧ�����ӷ���ʽΪ ��D�з�Ӧ�Ļ�ѧ����ʽΪ ��
��̽��ͭƬ��ŨH2SO4��Ӧ�IJ���
ʵ�������������ͭƬ���渽�ź�ɫ���塣�������ϵ�֪���˺�ɫ������ܺ���CuO��CuS��Cu2S��������CuS��Cu2S��������ϡ���ᣬ�ڿ��������ն�ת��ΪCu2O��SO2����С��ͬѧ�ռ�һ������ɫ���壬������ʵ�鷽��̽����ɷ֣�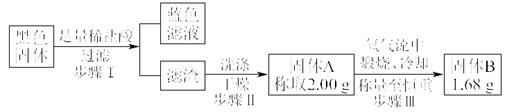
��3������� �м�������ϴ�Ӹɾ���ʵ�鷽����_____________________________��
��4����ɫ����ijɷ���________________��
�������
�ð�ˮ����β���е�SO2��������Һ���п��ܺ���OH����SO32����SO42����HSO3���������ӡ�
��5����ˮ���չ���SO2�ķ�Ӧ�����ӷ���ʽΪ ��
��6����֪����������һ��������ˮ��SO2Ҳ������ˮ��
�����������Լ�Ϊ��С�ձ����Թܡ�����������ͷ�ιܡ�����װ�ú���ֽ��2 mol/L���ᡢ
2 mol/LHNO3��1 mol/LBaCl2��Һ��l mol/LBa(OH)2��Һ��Ʒ����Һ������ˮ�������ʵ��֤��������Һ���д���SO32����HSO3��������±���ʵ�������Ԥ������ͽ��ۣ�
| ʵ����� | Ԥ����������� |
| ����1��ȡ����������Һ����С�ձ��У��ý�ͷ�ι�ȡl mol/L BaCl2��Һ��С�ձ��μ�ֱ�������� | �����ְ�ɫ���ǣ�������Һ�� �д���SO32���� SO42���� |
| ����2����С�ձ��е���Һ���ˡ�ϴ�ӣ���������ˮ�Ѹ�����ֽ�ϵĹ��������һС�ձ��У�����µĹ��� �� | �� ������Һ���д��� SO32���� |
| ����3�� �� | �� ������Һ���д��� HSO3���� |
��1���ܣ�1�֣�
��2��Ʒ����Һ��1�֣� SO2��I2��2H2O��SO42����2 I����4H+��2�֣�
SO2��2H2S="3S��+" 2H2O��2�֣�
��3��ȡ���һ��ϴ��Һ�������μ�����AgNO3��Һ����������������ϴ�Ӹɾ���2�֣���
��4��CuO��CuS��Cu2S��2�֣�
��5��NH3��H2O��SO2��NH4+��HSO3����2�֣�
��6������1�֣�ʵ����� Ԥ����������� ����2������1�Σ���������Ʒ����Һ���ٵ������2mol/L���ᣬ�� ��Ʒ����ɫ���������ݡ�����������ܽ⣩�� ����3�����Թ�ȡ������Һ�������е��������1mol/LBa(OH)2��Һ [�����1�Σ���������Ʒ����Һ���ٵ���2-3�Σ����������2mol/L����]���� �����ְ�ɫ��������Ʒ����Һ��ɫ���������ݣ�
���������������B��ʢ��Ʒ�죬�ܼ���SO2��Ư���ԡ����ˮ����SO2��I2��2H2O��SO42����2 I����4H+֤���仹ԭ�ԡ�D�з�ӦSO2��2H2S="3S��+" 2H2O��֤���������ԣ�
��ȡ���һ��ϴ��Һ�������μ�����AgNO3��Һ����������������ϴ�Ӹɾ�������1�����������ɫ��Һ��ȷ����CuO,�ڿ��������ն�ת��ΪCu2O��SO2, ��Ӧ����ʽΪ��4CuS+3O2=2Cu2O+4SO2
��2Cu2S+3O2=2Cu2O+2SO2����2g��������AΪCuS,��Ϸ���ʽ�ټ���ù�������BΪ1.47g�������������AΪCu2S����Ϸ���ʽ�ڼ���ù�������B������Ϊ1.8g������B������Ϊ1.68g������ͬʱ����CuS��Cu2S��
��ˮ���չ�����SO2�����ӷ���ʽΪNH3��H2O��SO2��NH4+��HSO3-֤��SO32���Ĵ��ڣ�������������м������ᣬ��Ʒ������Ƿ����SO2��֤��HSO3-��������ȿ����ᷴӦ��Ҳ����Ӧ�����ʡ�
���㣺��Ԫ�ػ�������صĻ�ѧʵ�顣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼ��ʾΪ���������Ʊ������������֤��β�������IJ�������װ�ã����ȼ��̶�װ�þ���ȥ��������װ�ÿ�����ѡ��Ҫʱ���ظ�ѡ�á�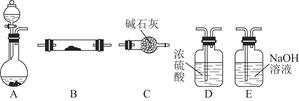
��ش��������⣺
��1����װ��A��Բ����ƿ��װ��пƬ����Һ©����װ��ϡ���ᣬ����֤H2�Ļ�ԭ�Բ��������������
������˳��A��D��B��B��C����ʱ��������ʹ�õ�װ��B����ʢ��ҩƷ�Ļ�ѧʽ����ΪCuO�� ���ѧʽ����
�ڼ���װ�õ������Ժ��ڼ���װ��CuO��װ��Bǰ������еIJ����� ��
��2����װ��A����ƿ��װ��Na2O2����Һ©����װ��Ũ��ˮ��װ��B��װ�����������Խ��а��Ĵ�������
����װ�õ�����˳��ΪA��C��B��C����װ��B�а��Ĵ�������Ӧ�Ļ�ѧ����ʽΪ ��װ��B�п��ܹ۲쵽�������� ��
������װ��A�Ƶõ�������Cl2�����һ�𣬲������̣�д����Ӧ�Ļ�ѧ����ʽ ���÷�Ӧ�����˰��� �ԡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧʵ��С���ͬѧΪ̽���ͱȽ�SO2����ˮ��Ư���ԣ���������µ�ʵ��װ�á�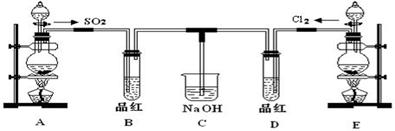
��1��ʵ������װ��A�Ʊ�SO2��ijͬѧ��ʵ��ʱ���ִ�A�ķ�Һ©��������©����Һ��δ���£�����Ϊԭ������� ��
��2��ʵ������װ��E�Ʊ�Cl2���䷴Ӧ�Ļ�ѧ��ѧ����ʽΪ��
MnO2+4HCl��Ũ�� MnCl2+Cl2��+2H2O������6 mol��HCl�μӷ�Ӧ����ת�Ƶĵ�������Ϊ ��
MnCl2+Cl2��+2H2O������6 mol��HCl�μӷ�Ӧ����ת�Ƶĵ�������Ϊ ��
��3���ٷ�Ӧ��ʼһ��ʱ��۲쵽B��D�����Թ��е�Ʒ����Һ���ֵ������ǣ�B�� ��D�� ��
��ֹͣͨ����,�ٸ�B��D�����Թֱܷ���ȣ������Թ��е�����ֱ�ΪB�� ��D�� ��
��4����һ��ʵ��С���ͬѧ��ΪSO2����ˮ����Ư���ԣ�����Ϻ��Ư���Կ϶����ǿ�����ǽ��Ƶõ�SO2��Cl2��1��1ͬʱͨ�뵽Ʒ����Һ�У����������ɫЧ���������������������������������ԭ���û�ѧ����ʽ��ʾ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��Ϥʵ������������ȷ����ʵ����������û�ѧʵ���ǰ�ᡣ
��1�������й�ʵ�������ʵ����ʵ����������ȷ���� (�����)��
A��ʵ������Ũ����Ӧ��������ɫϸ��ƿ�У���������ͼ��ʾ��ǩ |
| B����50mL��Ͳ��ȡ5��6mLŨ���� |
| C���к͵ζ�ʵ��ʱ����ƿϴ�Ӹɾ����ñ�Һ��ϴ����ע�����Һ |
| D�������Ȼ�̼��ȡ��ˮ�еĵ⣬��Һʱ�л���ӷ�Һ©�����¶˷ų� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧС�������ͼʵ��װ�ã��г�װ����ȥ���Ʊ�Cl2��̽��������������ʣ�
��1����װ��A�еĹ���ҩƷΪKClO3����Ӧ��ÿ����3mol Cl2 ʱת�Ƶ��ӵ����ʵ���
Ϊ ��
��2��װ��B���ܳ�ȥ�����е��Ȼ��⣬���ܼ��ʵ�������װ��C�Ƿ�����������C�з���������B�н��۲쵽�������� ��
��3��װ��C����������֤�����Ƿ����Ư���ԣ�l����ʪ�����ɫ���������Ӧ��������ʷֱ��� �� ��
��4�����װ��D��E��Ŀ���DZȽ��ȡ��塢��ķǽ����ԡ��������˵���ȡ��塢��ǽ�����ǿ����ʵ����������� ��
��5���û�ѧ����ʽ˵��װ��F������ ��
��6����ͬѧ�����װ��F�е��Լ��ɸ���������Na2SO3��Һ����ͬѧ����˼������Ϊ�˷������С��������ӷ���ʽ��������Ϊ�����е�ԭ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ȤС���������ͼʵ��װ�ý���ʵ�顣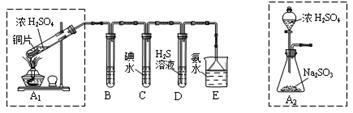
��̽��������Ⱦ��SO2������
��1��Ϊ��ʵ����ɫ������Ŀ�꣬�ܷ�����ͼA2����A1װ�� ����ܡ�����
��2��B��C��D�ֱ����ڼ���SO2��Ư���ԡ���ԭ�Ժ������ԣ���B����ʢ�Լ�Ϊ ��C�з�Ӧ�����ӷ���ʽΪ ��D�з�Ӧ�Ļ�ѧ����ʽΪ ��
��̽��ͭƬ��ŨH2SO4��Ӧ�IJ���
ʵ�������������ͭƬ���渽�ź�ɫ���塣�������ϵ�֪���˺�ɫ������ܺ���CuO��CuS��Cu2S��������CuS��Cu2S��������ϡ���ᣬ�ڿ��������ն�ת��ΪCu2O��SO2����С��ͬѧ�ռ�һ������ɫ���壬������ʵ�鷽��̽����ɷ֣�
��3��������м�������ϴ�Ӹɾ���ʵ�鷽����_____________________________��
��4����ɫ����ijɷ���________________��
�������
�ð�ˮ����β���е�SO2��������Һ���п��ܺ���OH-��SO32-��SO42-��HSO3-�������ӡ�
��5����ˮ���չ���SO2�ķ�Ӧ�����ӷ���ʽΪ ��
��6����֪����������һ��������ˮ��SO2Ҳ������ˮ�������������Լ�Ϊ��С�ձ����Թܡ�����������ͷ�ιܡ�����װ�ú���ֽ��2 mol/L���ᡢ2 mol/LHNO3��1 mol/LBaCl2��Һ��l mol/LBa(OH)2��Һ��Ʒ����Һ������ˮ�������ʵ��֤��������Һ���д���SO32-��HSO3-������±���ʵ�������Ԥ������ͽ��ۣ�
| ʵ����� | Ԥ����������� |
| ����1��ȡ����������Һ����С�ձ��У��ý�ͷ�ι�ȡl mol/L BaCl2��Һ��С�ձ��μ�ֱ�������� | �����ְ�ɫ���ǣ� ������Һ���д���SO32-�� SO42-�� |
| ����2����С�ձ��е���Һ���ˡ�ϴ�ӣ���������ˮ�Ѹ�����ֽ�ϵĹ��������һС�ձ��У�����µĹ��� �� | �� ������Һ���д��� SO32-�� |
| ����3�� �� | �� ������Һ���д��� HSO3-�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������Ϸ��֣�N2��ȡ�����в�ͬ������
a����������������NH3��ԭCuO�Ƶô�����N2�ͻ���ͭ��
�ⷽ��������NaNO2��NH4Cl��Ũ��Һ�Ƶ�
c������������������ͨ�����ȵ�ͭ���Ƶýϴ���N2
����ʵ�����й�ѡ������¼�����������ȡN2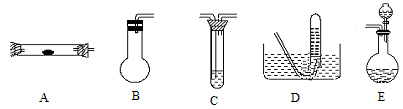
��1������ ������N2ʱ����Ҫ�İ�������ʯ�Һ�Ũ��ˮ��ԭ����ȡ�����˲������������е�____________����A��B��C������ͬ����NH3�ķ�������Ҫ��ȡ���ռ�N2������ѡ�õ���������___________��
������N2ʱ����Ҫ�İ�������ʯ�Һ�Ũ��ˮ��ԭ����ȡ�����˲������������е�____________����A��B��C������ͬ����NH3�ķ�������Ҫ��ȡ���ռ�N2������ѡ�õ���������___________��
��2��д��b�����з�Ӧ�Ļ�ѧ����ʽ_________________________
��3������ ������ȡN2��������һ�����к����������� ______________________
������ȡN2��������һ�����к����������� ______________________
��4��������N2�����������У� �������ʹ��Խ��Խ�ܵ����ǵĹ�ע�����ַ�����
�������ʹ��Խ��Խ�ܵ����ǵĹ�ע�����ַ����� ������ȣ�����Խ������____________��
������ȣ�����Խ������____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�����������������ѧ��ѧ�еij������壬̽�����ߵ��Ʒ���������ʮ����Ҫ�Ŀ��⡣
��1��ʵ���ҿ��ɶ���;����ȡSO2��
;��I��Na2SO3�������Ũ�����ᣨԼ70%����Ӧ��ȡ��
;��II��ͭ��Ũ���������ȡ��
���Ҫ��ʵ������ȡSO2��ѡ���������ַ����е� ���;��I����;��II��������������������� (���һ��Ϳ�)��
��2����ͼ����KMnO4��Ũ���ᷴӦ��ȡ���������ļ���װ�á�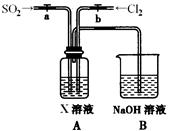
��֪��2 KMnO4+16HCl��Ũ��= 2KCl+2MnCl2+5Cl2��+8H2O��װ��B��C��D�����÷ֱ��ǣ�
B�� ��
C�� ��
D�� ��
��3��ijͬѧ���������װ��̽���������������Ļ�ѧ���ʡ�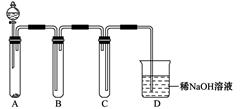
ͨ�����Ƶ��ɼ�a��b����װ��A�зֱ�ͨ�벻ͬ���壬������������⣺
�����ر�b����a����XΪƷ����Һ����A�е�����Ϊ�� ��˵������������� �ԡ�
�����ر�a����b����XΪ��ɫʯ����Һ����A�е�����Ϊ�� ��ԭ���� ��
����ͬʱ��a��b����ͨ������������Ϊ1:1����XΪ��ɫʯ����Һ����A�е�����Ϊ ���������ͬ��ԭ���� ����д��Ӧ�Ļ�ѧ����ʽ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�ף��������о���ѧϰС��Ϊ�ⶨ�������е�����ԭ�Ӹ����ȣ����������ʵ�����̣�
��ͼA��B��CΪ�ף�����С����ȡ����ʱ�����õ���װ�ã�DΪʢ��Ũ�����ϴ��ƿ��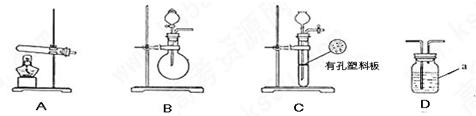
ʵ�鿪ʼǰװ���еĿ������ž�����С���ã���Ӧǰ����ͭ������Ϊ ������ͭ��Ӧ��ʣ����������Ϊ
������ͭ��Ӧ��ʣ����������Ϊ �����ɵ����ڱ�״���µ����
�����ɵ����ڱ�״���µ���� ����С����ϴ��װ��Dǰ������������ɵ����ڱ�״���µ������
����С����ϴ��װ��Dǰ������������ɵ����ڱ�״���µ������
��1��д������a�����ƣ� ��
��2���ף�����С��ѡ���˲�ͬ������ȡ�������뽫ʵ��װ�õ���ĸ��ź��Ʊ�ԭ����д���±��ո��С�
| | ʵ��װ�� | ʵ��ҩƷ | �Ʊ�ԭ�� |
| ��С�� | A | �������ƣ������ | ��Ӧ�Ļ�ѧ����ʽΪ �� |
| ��С�� | �� | Ũ��ˮ���������� | �û�ѧƽ��ԭ�������������Ƶ����ã� �� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com