
ij��ѧС��������װ�ö������仯��������ʽ���̽�����ش��й����⣺
��1����С��ͬѧ���Ƶ�����������������ϳ�ʱ�䣬����ͼ��a����ʾװ�ö�����в�����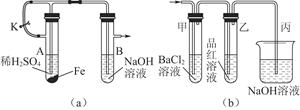
��ijͬѧ�IJ����ǣ��ȼн�ֹˮ��K����ʹA�ܿ�ʼ��Ӧ��ʵ������B���й۲쵽��������
��
��Ϊ�ﵽʵ��Ŀ�ģ���ȷ�IJ����� ��
B���з�����Ӧ�����ӷ���ʽ�� ��
��2��������װ�õ�ʵ�������ȡA���ڷ�Ӧ���õ���Һ����������С�����ɺ�õ�FeSO4���ٸ������գ��й�װ�úͲ���������ȥ����������º���ɫ���壬���ֽ�ʱ���������尴ͼ��b����ʾװ������ͨ��ϴ��װ�ã�����Թܼ��ڳ��ְ�ɫ�������Թ�����Ʒ����Һ��ɫ��ȥ���ش�
���÷���ʽ��ʾ�Թܼײ�����ɫ������ԭ��
��
��Ϊ��֤����ɫ����ɷ֣��ɽ������²���
��
��ͼ��b���б�װ�õ������� ��
�ܸ���ʵ������д��A������Һ���ɺ��ڸ������շֽ�ʱ��������Ӧ�Ļ�ѧ����ʽ ��
��1����A����Һ����B�У���ʼ������ɫ������Ȼ���Ϊ����ɫ������ɺ��ɫ
�ڴ�ֹˮ��a��ʹA�ܷ�Ӧһ��ʱ����B�е����о������ݲ������ټн�ֹˮ��a
Fe2����2OH��=Fe��OH��2�� ���������кͷ�Ӧд��д���ɣ�
��2����SO3��H2O=H2SO4��
H2SO4��BaCl2=BaSO4����2HCl
��SO3��H2O��BaCl2=BaSO4����2HCl
��ȡ��������������ٵμ�KSCN��Һ���۲쵽��Һ���ɫ
������SO2
��2FeSO4 Fe2O3��SO3����SO2��
Fe2O3��SO3����SO2��
����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��70 g�������ƺ������ƵĻ�����98 gˮ��ַ�Ӧ����������������Һ����������Ϊ50%��
��1����ԭ������й������ƺ������Ƶ���������2���������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������������(FeSO4��7H2O)��ҽҩ������Ѫ����ij����С���ͬѧ���ⶨ�ò�Ѫ����
��Ԫ�صĺ�����ʵ�鲽�����£�
��ش��������⣺
(1)֤���������Һ�к���Fe2���ķ����ǣ�ȡ�����ȵμ�KSCN��Һ���ٵμ�________���ù��̵�����Ϊ________��
(2)����ڼ������H2O2��Ŀ���ǣ�______________________________________��
(3)������з�Ӧ�����ӷ���ʽ�ǣ�_______________________________________��
(4)�������һϵ�д����IJ������裺���ˡ�________�����ա�________��������
(5)��ʵ����������ģ���ÿƬ��Ѫ���к���Ԫ�ص�����Ϊ________g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�ú���Al2O3��SiO2������FeO��xFe2O3�������Ʊ�Al2(SO4)3��18H2O��������������(���ֲ�����������)��
��.�������м������ϡH2SO4�����ˣ�
��.����Һ�м������KMnO4��Һ��������Һ��pHԼΪ3��
��.���ȣ�����������ɫ���������ã��ϲ���Һ���Ϻ�ɫ��
��.����MnSO4���Ϻ�ɫ��ʧ�����ˣ�
��.Ũ�����ᾧ�����룬�õ���Ʒ��
��1��H2SO4�ܽ�Al2O3�����ӷ���ʽ��____________________________________��
��2����MnO4-����Fe2�������ӷ���ʽ����������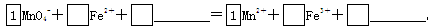
��3����֪��
�����������������pH
| | Al(OH)3 | Fe(OH)2 | Fe(OH)3 |
| ��ʼ����ʱ | 3.4 | 6.3 | 1.5 |
| ��ȫ����ʱ | 4.7 | 8.3 | 2.8 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Fe2����I�������ֳ����Ļ�ԭ�����ӡ�
��1����FeSO4��Һ�еμ���ˮ����Һ��dz��ɫ��ɻ�ɫ����Ӧ�����ӷ���ʽΪ ����KI��Һ�еμ���ˮ����Һ����ɫ��ɻ�ɫ����Ӧ�����ӷ���ʽ�� ��
��2������FeSO4��Һ��KI��Һ����ˮΪ�Լ���֤I���Ļ�ԭ��ǿ��Fe2�������ʵ�鷽�����������ʵ�鲽�衢Ԥ������ͽ��ۡ�������ѡ�Լ���3 mol��L��1 H2SO4��0.01 mol��L��1 KMnO4��20% KSCN��3%H2O2��������Һ����ɫʯ����Һ��
| ʵ�鲽�� | Ԥ����������� |
| ����1��ȡ2mLFeSO4��Һ��2mLKI��Һ������Թ��У��ٵμ�1��2����ˮ�� | �� |
| ����2��____________________________________ ____________________________________�� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼʵ��װ��������֤ijЩ���ʵ����ʡ����Թ�A��װ�������Ĺ���NaHCO3,DΪ�̶������ӲֽƬ���Իش��������⣺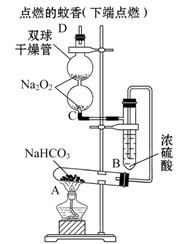
��1����ʵ���ʵ��Ŀ����__________________________________________��
��2����A�Թ��ڷ�����Ӧ�Ļ�ѧ����ʽ��_______________________________��
��3��Bװ�õ�������_________________________________________________��
��4����˫�������ڷ�����Ӧ�Ļ�ѧ����ʽΪ____________________________��
��5��ʵ��ʱ�۲쵽��ʵ��������_______________________________________��
����ʵ������˵��___________________________________________________��
��6������������ڵ�Na2O2����Na2O����ʵ��ʱ�۲쵽��ʵ��������___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijѧϰС����ͨ��ʵ��̽���̷���FeSO4��7H2O���ȷֽ�IJ��
��ʵ��ǰ����
��1�����۷��� С���Ա�������Ϸ�������Ϊ���зֽ���ﲻ���ܵ��� ��
a��Fe2O3��SO3��H2O b��Fe2O3��SO2��SO3��H2O
c��FeO��Fe2O3��SO2��SO3��H2O
��2���������� ��ѹ��SO3�۵�16��8�棬�е�44��6��
��ʵ��̽����
������Ͽ��ܵ���ϲ��룬��ѧϰС���������ͼ��ʾ��ʵ��װ�ã�ͼ�м��ȡ��г������Ⱦ�ʡ�ԣ���
��3��ʵ�����
���������Ӻ��װ��A��B�����ԵIJ���Ϊ ��
��ȡһ�����̷���������A�У�ͨ��N2������װ���ڵĿ������ر�k���þƾ��Ƽ���
˫ͨ�ܡ�
�۹۲쵽A �й��������ɫ��B���Թ��ռ�����ɫҺ�壬C����Һ��ɫ��
�ܴ�A�з�Ӧ��ȫ����ȴ�����º�ȡ������Ӧ��������Թ��У����������ܽ⣬ȡ
�������뼸��KSCN��Һ����Һ���ɫ��
����Bװ�õ��Թ��е��뼸��BaCl2��Һ����Һ����ǡ�
( 4��ʵ��������
����1��B���ռ�����Һ���� ��
����2��C����Һ��ɫ������֪�������� ��
����3���ۺϷ�������ʵ��ۺܿ͢���֪�������һ����Fe2O3��
��ʵ�鷴˼��
��5����ָ����С����Ƶ�ʵ��װ�õ����Բ��㣺 ��
��6���ֽ��Ĺ����п��ܺ�������FeO��ȡ����ʵ����������ܽ�����Һ�������Թ��У�
ѡ��һ���Լ����𣬸��Լ�����ʵ��� ��
a����ˮ��KSCN��Һ b������KMnO4��Һ c�� H2O2 d�� NaOH��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����������FeC2O4�������������Լ�����Ӱ���Լ����͵�ز�����������﮵����������������ڸ�������ʱ�����ܹ��ֽ�,��ȤС��Բ��������ķֽ���������ʵ���̽��������֪��CO�����Ȼ���[PdC12]��Һ��Ӧ���ɺ�ɫ���ٷۡ���
��1�������������ֽ�������������ͨ������ʯ��ˮ���Ȼ�����Һ���۲쵽����ʯ��ˮ����ǣ��Ȼ�����Һ���к�ɫ�������ɡ�˵������������� �����ѧʽ��
��2��̽�����������ֽ�õ��ĺ�ɫ�����������Ԫ�صĴ�����ʽ��
���������⡿
���������ֽ��õ��ĺ�ɫ������ʲô��
��������衿
����1�� ������2��FeO������3��FeO��Fe�Ļ���
��ʵ�鷽����
��ѡ�Լ������ᡢ��ˮ��CuSO4��Һ��KSCN��Һ������ˮ��
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ����1�����Թ��м��������������ټ������� ������� | ����Һ��ɫ���Ըı䣬 ���к�ɫ�������ɡ� | ��Fe���ڡ� |
| ����2��������1�еõ�����Һ���ˣ���������ˮ������ϴ����ϴ��Һ����ɫ�� | | |
| ����3��ȡ����2�õ��������������Թ��У��μӹ������ᣬ���ú�ȡ�ϲ���Һ�� �� | �� | ��FeO���ڡ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijУ��ѧ��ȤС��Ϊ̽������Ũ����ķ�Ӧ,�����ͼ1��ͼ2��ʾװ�ý���ʵ�顣 
��1����˵����SO2���������ʵ�������� ��
��2��ͼ2�е�����e����Ҫ����Ϊ ��
��3������װ����ͼ2�е�NaOH��Һ������SO2β������ֹ��Ⱦ���罫�����Ϊ����KMnO4��Һ��ͬ�����ԴﵽĿ�ģ���д������KMnO4��Һ��SO2��Ӧ�Ļ�ѧ����ʽ��
��
��4���Ա�����ʵ��װ�ã����ѷ���ͼ2װ�ó����ܸ��õ������ж�����SO2��ֹ����Ⱦ�����⣬����һ���dz����Ե��ŵ㣬����Ϊ�� ��
��5����Ӧһ��ʱ���ֹͣ��Ӧ������ȴ���ý�ͷ�ι���ȡA�Թ��е���Һ���뵽����ˮ����Ϊ�����������������������ӵijɷ����������ֿ��ܣ�
��ֻ����Fe3+����ֻ����Fe2+�� ����Fe3+����Fe2+��
Ϊȷ����Һ�ijɷ֣�ѡ�������Լ���
| A��ϡHCl��Һ | B��ϡ���� | C��KSCN��Һ | D������KMnO4��Һ |
| ʵ�鲽�� | ʵ�������� |
| 1��ȡһ֧�ྻ���Թܣ��μ�1-2mL��������Һ�������Թ��еμӼ���KSCN��Һ | ��1�� ����˵��������� ��2�� ����˵����Һ�д���Fe3+������������ |
| 2�� | �� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com