
����ʯ����ȡ�طʺ�������������Ҫԭ�ϣ�����ʯ����ɺ��������ƣ�����������������������������ʡ�����ʵ�鲽������ͼ��ʾ��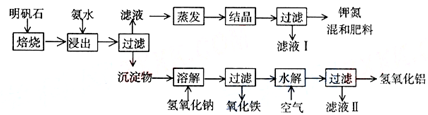
��������ͼʾ�����������գ�
��1������ʯ���պ���ϡ��ˮ����������500mLϡ��ˮ��ÿ������39.20g������ҪȡŨ��ˮ��ÿ������251.28g����__________mL���ù��Ϊ_______mL��Ͳ��ȡ��
��2����ˮ������õ���������ϵ�����ˣ���Һ�г�K����SO42���⣬���д�����NH4��������NH4���ķ�����______________________________________________��
��3��д�����������������ʵĻ�ѧʽ________________________________________��
��4����ҺI�ijɷ���ˮ��______________��
��5��Ϊ�ⶨ��Ϸ���K2SO4��(NH4)2SO4�мصĺ��������������в��裺
�ٳ�ȡ�ص�������������ˮ����������______��Һ��������ɫ������
��___________��__________��_________(������дʵ���������)��
����ȴ�����ء�
��6��������Ϊmg�����������ʵ���Ϊnmol����������K2SO4�����ʵ���Ϊ��___________mol���ú���m��n�Ĵ���ʽ��ʾ����

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| (m-132n) |
| 42 |
| (m-132n) |
| 42 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ʯ����ȡ�طʺ�������������Ҫԭ�ϣ�����ʯ����ɺ��������ƣ�����������������������������ʡ�����ʵ�鲽������ͼ��ʾ��

��������ͼʾ�����������գ�
��1������ʯ���պ���ϡ��ˮ����������500mLϡ��ˮ��ÿ������39.20g������ҪȡŨ��ˮ��ÿ������251.28g����__________mL���ù��Ϊ_______mL��Ͳ��ȡ��
��2����ˮ������õ���������ϵ�����ˣ���Һ�г�K����SO42���⣬���д�����NH4��������NH4���ķ�����______________________________________________��
��3��д�����������������ʵĻ�ѧʽ________________________________________��
��4����ҺI�ijɷ���ˮ��______________��
��5��Ϊ�ⶨ��Ϸ���K2SO4��(NH4)2SO4�мصĺ��������������в��裺
�ٳ�ȡ�ص�������������ˮ����������______��Һ��������ɫ������
��___________��__________��_________(������дʵ���������)��
����ȴ�����ء�
��6��������Ϊmg�����������ʵ���Ϊnmol����������K2SO4�����ʵ���Ϊ��___________mol���ú���m��n�Ĵ���ʽ��ʾ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ�긣��ʡ��������У�����ڶ���������ѧ�Ծ��������棩 ���ͣ������
����ʯ����ȡ�طʺ�������������Ҫԭ�ϣ�����ʯ����ɺ�����[KAl(SO4)2?12H2O]���ƣ�����������������������������ʡ�����ʵ�鲽������ͼ��ʾ��

��������ͼʾ�����������գ�
��1������ʯ���պ���ϡ��ˮ����������500mlϡ��ˮ��ÿ������39.00g������ҪȡŨ��ˮ��ÿ������250g����__________ml���ù��Ϊ_______ml��Ͳ��ȡ��
��2����ˮ������õ���������ϵ�����ˣ���Һ�г�K����SO42���⣬���д�����NH4��������NH4���ķ�����_______________________________________________________��
��3��д�����������������ʵĻ�ѧʽ________________________________________��
��4����Һ�ijɷ���ˮ��______________��
��5��Ϊ�ⶨ��Ϸ���K2SO4��(NH4)2SO4�мصĺ��������������в��裺
�ٳ�ȡ�ص�������������ˮ����������______��Һ��������ɫ��������_______��______������(��ʵ���������)������ȴ�����ء�
��6��������Ϊmg�����������ʵ���Ϊn mol����������K2SO4�����ʵ���Ϊ��______���ú���m��n�Ĵ���ʽ��ʾ����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com