
�ߴ���ϸ�������������ںϳ�����﮵�ص缫���ϣ���ҵ�Ͽ�������ȡ�Ѱ۵ĸ���Ʒ�̷���FeSO4��7H2O��ͨ�����з�Ӧ�Ʊ���
FeSO4��2NH3��H2O��Fe(OH)2����(NH4)2SO4
Fe(OH)2��H2C2O4��FeC2O4��2H2O
��1���̷��к���һ������TiOSO4���ʡ����̷�����ϡ���ᣬ�������ۡ����衢��ַ�Ӧ������һ��ʱ�䣬���ˣ��ɵô�����FeSO4��Һ�������������У�TiOSO4����ˮ��Ӧת��ΪH2TiO3������д���÷�Ӧ�Ļ�ѧ����ʽ�� ���������۵������� �� ��
��2���ɴ�����FeSO4��Һ��ȡFeC2O4ʱ��������ջ����½��У�ԭ���� ��
FeC2O4���ɺ�Ϊ��߲�Ʒ���ȣ����������ҺpH��2����pH���ͣ�����
FeC2O4�IJ��� ���ƫ�ߡ�����ƫ�͡�����Ӱ�족����
��3��������FeC2O4�Ļ��Һ���ˣ�����Ʒ����ˮϴ�ӣ�������ˮ�Ҵ���ϴ����ˮ�Ҵ��������� �� ��
��4��ij�о�С������ij������������Ҫ�ɷ�ΪFe2O3��SiO2��Al2O3�����������Ʊ��ϴ�����FeSO4��Һ���ٺϳ�FeC2O4���벹�������ɸû��������Ʊ��ϴ�����FeSO4��Һ��ʵ�鲽�裨��ѡ�õ��Լ������ۡ�ϡ�����NaOH��Һ������һ�����û��������м���������ϡ�����ַ�Ӧ�����ˣ� �����ˣ��õ��ϴ�����FeSO4��Һ��
��1��TiOSO4��2H2O��H2TiO3����H2SO4��2�֣�
��ֹFe2+��������������ҺpH����ȥ���ʣ������𰸾��ɣ�
��2���������ɵ�Fe(OH)2�����ʱ������������𰸾��ɣ���2�֣� ƫ�ͣ�2�֣�
��3����ȥ�����������ʡ�ʹ��Ʒ���ٸ�����ٲ�Ʒ����ĵȣ�2�֣�ÿ���1���1�֣�
��4������Һ�еμӹ�����NaOH��Һ�����ˣ����ϴ�ӹ��壬������м�������ϡ������������ȫ�ܽ⣬�ټ������������ۣ���ֽ����3�֣�
���������������1����Һ�е����������ױ������е��������������Լ������۷�ֹFe2+����������������Һ�������ӷ�Ӧ���ٽ�TiO2+ˮ����ȫ�����Ӷ���ȥ����2�����Ʊ������з�Ӧ���ɸ��ױ�������Fe(OH)2���������������Ƿ�ֹ������������ΪFe(OH)3��ʵ�����������Һ���Թ�ǿ��������ʽ�������������²���ƫ�͡���3���Ҵ����н�ǿ�ӷ��ԣ�������ˮϴ�������Ҵ�ϴ����Ȼ��ʹ��Ʒ���ٸ����ֹ�ڼ��Ȼ��ڿ����л����������Ӧ�Բ�Ʒ����������Ӱ�죻��4���Ʊ�������FeSO4��Һ��Ҫ��ȥ���ʣ��ȼ����ܽ���˳�ȥ�������裬�ټӹ������ȥ�����ӣ��ټ����ᡢ���۵ȵõ�������FeSO4��Һ��
���㣺�����ۺ�ʵ���й��̵Ŀ��ơ�ԭ���������й����⡣


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����̿�MnCO3��������Fe2O3��FeO��HgCO3��2HgO�����ʣ���ҵ�������̿���ȡ�̣������������£�
��ش��������⣺
��1�����Һ1�м����ˮ�����Ҫ �������ܴﵽ����Ҫ��
��2���������õĿ�������Ĥ���뷨�Ʊ��ĸ����������÷�����ԭ���� ��
��3����������Ҫ�ɷ�Ϊ(NH4)2S����Һ2�з�����Ҫ��Ӧ�����ӷ���ʽΪ ��
��4��д�������ĵ缫��Ӧʽ ��˵�����Һѭ����ԭ�� ��
��5��д�����ȷ����̵Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������(LiFePO4)����Ϊ������ǰ;������ӵ���������ϡ�ij��ҵ���ø�������Һ�����������,�����˴�������������Һһ����;��������Ҫ��������: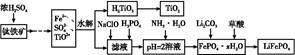
��֪:H2TiO3����������ˮ�����ʡ�
(1)��������Ũ���ᴦ��֮ǰ,��Ҫ����,��Ŀ���� ����
(2)TiO2+ˮ������H2TiO3�����ӷ���ʽΪ ����
(3)����NaClO������Ӧ�����ӷ���ʽΪ ����
(4)��ʵ����,����Һ�й��˳�H2TiO3��,������Һ����,Ӧ��β����� ��
(5)Ϊ�ⶨ�����������ĺ���,ijͬѧȡ��Ũ����ȴ�������Һ(��ʱ�������е�����ȫ��ת��Ϊ����������),��ȡKMnO4��Һ�ζ�Fe2+�ķ���:(������KMnO4���������ʷ�Ӧ)�ڵζ�������,��δ�ñ�Һ��ϴ�ζ���,��ʹ�ⶨ�����������(�ƫ�ߡ�����ƫ�͡�����Ӱ�족),�ζ��յ���������� ���ζ�����ʱ,��ȡa g������,������,��c mol/L KMnO4��Һ�ζ�,����V mL,����Ԫ�ص����������ı���ʽΪ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������أ� ������ǿ�����ԣ�������ԭΪ����أ���80 �����������ֽ⡣ʵ����ģ�ҵ�ϳɹ�����ص��������£�
������ǿ�����ԣ�������ԭΪ����أ���80 �����������ֽ⡣ʵ����ģ�ҵ�ϳɹ�����ص��������£�
��1������狀��������Ƴɵ��Һ���Բ����缫���е�⣬���ɹ��������Һ��д�����ʱ������Ӧ�����ӷ���ʽ_____________________________________
___________________________________��
��2����֪������ʵ��ܽ����������ͼ��ʾ����ʵ�������ᴿ������شֲ�Ʒ��ʵ������������Ϊ����������شֲ�Ʒ��������ˮ�У�________________�����
��3����Ʒ�й�����صĺ������õ��������вⶨ��ʵ�鲽�����£�
����1����ȡ���������Ʒ0.300 0 g�ڵ���ƿ�У�����30 mLˮ�ܽ⡣
����2������Һ�м���4.00 0 g KI���壨�Թ�������ҡ�ȣ��ڰ�������30 min��
����3���ڵ���ƿ�м�������������Һ�ữ���Ե�����Һ��ָʾ������0.100 0 mol��L��1Na2S2O3����Һ�ζ����յ㣬������Na2S2O3����Һ21.00 mL��
����֪��Ӧ��I2��2S2O32-=2I����S4O62-��
��������2��δ������ƿ���ڰ�������30 min�����������в���3����ⶨ�Ľ������________��ѡ�ƫ����ƫС��������Ӱ�족������������3�еζ��յ��������____________________________________________��
�ڸ�����������ɼ��������Ʒ�й�����ص���������Ϊ_______________��
��Ϊȷ��ʵ������ȷ�ԣ�����Ϊ����Ҫ____________________________��
��4����0.40 mol���������0.20 mol�������Ƴ�1 L��Һ����80 �������¼��Ȳ���tʱ������Һ�еμ�������FeCl3��Һ���ⶨ��Һ�и��ɷֵ�Ũ����ͼ��ʾ��H��Ũ��δ��������ͼ������X�Ļ�ѧʽΪ________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������(K2FeO4)��һ�ּ�������������������һ������Ͷ��ˮ������,��������������:
��֪:��2KOH+Cl2 KCl+KClO+H2O(����:�¶Ƚϵ�)
KCl+KClO+H2O(����:�¶Ƚϵ�)
��6KOH+3Cl2 5KCl+KClO3+3H2O(����:�¶Ƚϸ�)
5KCl+KClO3+3H2O(����:�¶Ƚϸ�)
��2Fe(NO3)3+3KClO+10KOH 2K2FeO4+6KNO3+3KCl+5H2O
2K2FeO4+6KNO3+3KCl+5H2O
�ش���������:
(1)����������Ӧ����������(��¶Ƚϸߡ����¶Ƚϵ͡�)������½��С�
(2)д����ҵ����ȡCl2�Ļ�ѧ����ʽ�� ��
(3)K2FeO4����Ϊ���Ͷ��ˮ��������ԭ���� ��
(4)����KOH��Һʱ,����ÿ100 mLˮ���ܽ�61.6 g KOH����(����Һ���ܶ�Ϊ1.47 g��mL-1),�������ʵ���Ũ��Ϊ��������
(5)�ڡ���ӦҺ���м���KOH�����Ŀ����:�� ;��������������������
;��������������������
(6)�ӡ���ӦҺ���з����K2FeO4��,����Ʒ����������(д��ѧʽ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������������������С������������������ˮ��Դ�ḻ���ش��������⣺
��1����ˮ����Ҫ���� �����ӣ�д���ӷ��ţ����ٴ��4�֣���
��2����ˮ������һ����Ч��ȥ�������������SO2�ķ������乤����������ͼ��ʾ��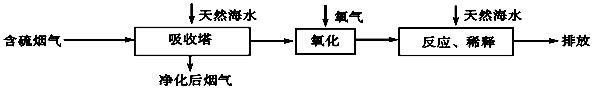
��Ȼ��ˮ�����˺���������Ҫ��O2���������������䷴Ӧ�Ļ�ѧ����ʽ�ǣ� ��������ġ���ˮ����Ҫ�ô�������Ȼ��ˮ��֮��Ϻ�����ŷţ��ò�������ҪĿ���� ��
��3����ͼ�Ǻ�ˮ�ۺ����õ�һ�����档
��ش��������⣺
I.�ٲ���Ҫ��Ӧ�����ӷ���ʽ�� ��
�弰�仯�������;�кܶ࣬д�����е�һ�� ��
II���ڢ۲���Ҫ���� ����ǰ������������þ�IJ�����Ϊ ����þ���ڶ�����̼��ȼ�յIJ���Ϊ ��
III������ˮ�Ժ��п����Ե�CaCl2��MgCl2��Na2SO4�����ʣ�ͨ�����¼���ʵ�鲽�裬���Ƶô�����ʳ��ˮ���� �����Թ�����Na2CO3��Һ���� �����Թ�����NaOH��Һ���� �����Թ�����BaCl2 ��Һ���ܵ���ϡ�����������ݲ������� ���� ��ȷ�IJ���˳����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ѧʵ���Ҳ����ķ�Һ�к��д�������Ⱦ���������ʣ�Ϊ�˱�����������Щ��Һ���뾭����������ŷš�ij��ѧʵ���Ҳ����ķ�Һ�к������ֽ������ӣ�Fe3+��Cu2+����ѧС�����������ͼ��ʾ�ķ����Է�Һ���д������Ի��ս���������������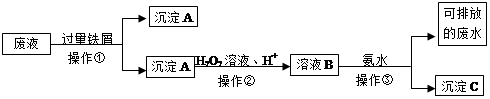
��1�������ٵ������� ��
��2������A�к��еĽ��������� ��
��3�����������������·�����Ӧ�����ӷ���ʽ ��
��4��������ҺB�к��еĽ��������ӳ��õ��Լ��� ��
��5���������з�����Ӧ�����ӷ���ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ϼʯ�ң���ѧʽΪ KNa3[AlSiO4]4����Ҫ�ɷ�Na2O��K2O��Al2O3��SiO2����̼���ơ�̼��غ��������Ĺ����������£�
��֪��NaHCO3��Һ��pHԼΪ8��9��Na2CO3��Һ��pHԼΪ11��12���ܽ���˹����������Һ�к��ơ��غ����Ŀ��������࣬�ƺ����������������ϼʯ���С��������ʵ��ܽ�ȼ���ͼ��
�Իش��������⣺
��1�����յõ�����M�Ļ�ѧ����ʽ��________________________________��
��2��X������___________����ҺW����Ҫ���е�������____________����д���֣�
��3��������õ�̼���ƾ���IJ���Ϊ���� �� �� ��ϴ�ӡ����
��4��̼�ữ���з�����Ҫ��Ӧ�����ӷ���ʽ��__________________________��
��5��̼�ữ�����pH��8��Ŀ����_______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
NiSO4��6H2O��һ����ɫ������ˮ�ľ��壬�㷺���ڻ�ѧ������������صȣ����ɵ�Ʒ������������⣬�����У�Cu��Zn��Fe��Cr�����ʣ�Ϊԭ�ϻ�á������������£�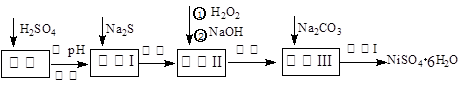
��1����Na2S��Ŀ���dz�ȥͭ��п�����ʣ���д����ȥCu2+�����ӷ���ʽ__________ __________
��2�� ��6%��H2O2ʱ���¶Ȳ��ܹ��ߣ�����Ϊ�� _____ ________ ��
��3�� ������������H2O2�������������NaOH����pHֵ3��4��Χ�������������������������������У�����������NaClO3���棬��д��������������Fe2+�����ӷ���ʽΪ��___________________________________________________________________________
��4��������������Һ�����Ҫ�ɷ��ǣ��� �������ѧʽ��
��5��������������¹��̣����ˣ��� �����Լ���ѧʽ���ܽ⣬ ��ϴ�ӻ�ò�Ʒ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com