��2011?ʯ��ɽ��һģ������������Ԫ��A��B��C��D��E��ԭ��������������AԪ�صĵ��ʳ��³�ѹ������������壬BԪ�����γɻ�����������࣬C������������Ӧˮ�����������̬�⻯�����ܹ������γ��α���DԪ�ص����Ӱ뾶��ͬ����Ԫ���γɵļ���������С�ģ�
��1����֪�������֮��������±仯��
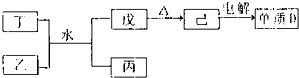
�ٶ����Һ�ˮ��Ӧ������ͱ������ӷ���ʽΪ
Al3++3NH3?H2O=Al��OH��3��+3NH4+
Al3++3NH3?H2O=Al��OH��3��+3NH4+
�������ʼ����õ�����D�Ļ�ѧ����ʽΪ
��
��0.1mol/L�ı���Һ����������Ũ���ɴ�С����˳��Ϊ
C��NO3-����C��NH4+����C��H+����C��OH-��
C��NO3-����C��NH4+����C��H+����C��OH-��
�������£�Ϊʹ����Һ���ɱ����������������Ũ����ȣ�Ӧ����Һ�м���һ�������ҵ�ˮ��Һ��
��ҺPH=7
��ҺPH=7
��
��2����֪E���仯���������±仯��
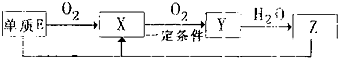
д������E�뻯����Z��һ�������·�Ӧ����X��ˮ�Ļ�ѧ����ʽ
_����A��B��C��D��E5��Ԫ���е�����Ԫ�أ����γɼȺ����Լ��ֺ��Ǽ��Լ���18���ӵķ��ӣ��÷��ӵķ���ʽΪ
C2H6��N2H4
C2H6��N2H4
����дһ�����ɣ���
��3��C�ж������������֮һ��һ����ɫ���壬�ڿ�����Ѹ�ٱ�ɺ���ɫ����һ�������£�2L�ĸ���ɫ������0.5L���������ϣ����û�����屻������NaOH��Һ��ȫ���պ�û����������������ɵ�C�ĺ������εĻ�ѧʽ��
NaNO2
NaNO2
��





 ����������Ԫ��A��B��C��D��E��ԭ������������������ԭ�Ӻ���ĵ��Ӳ���֮��Ϊ10��BԪ�صĻ���������࣬��Ŀ�Ӵ�C��D����Ԫ���γɵĵ����ǿ����к����������ʣ�D��E��Ԫ�ؿ����������ֲ�ͬ�����ӻ����
����������Ԫ��A��B��C��D��E��ԭ������������������ԭ�Ӻ���ĵ��Ӳ���֮��Ϊ10��BԪ�صĻ���������࣬��Ŀ�Ӵ�C��D����Ԫ���γɵĵ����ǿ����к����������ʣ�D��E��Ԫ�ؿ����������ֲ�ͬ�����ӻ���� NH3?H2O+H+
NH3?H2O+H+ NH3?H2O+H+
NH3?H2O+H+ 2NO2
2NO2 2NO2
2NO2