
ʵ������ͭ��ȡ����ͭ������������ֶ�μ��뵽ͭ����ϡ����Ļ�����У�����ʹ֮��Ӧ��ȫ��ͨ���������ᾧ�õ�����ͭ���塣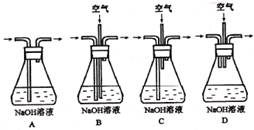
��1��Ϊ�˽�Լԭ�ϣ��������������ʵ���֮�����Ϊ:_ ��Ϊ�����ո÷�Ӧ�в�����β����Ӧѡ������װ��(����ĸ)��
��2��Ϊ������ɫ��ѧ��Ҫ��ij�о���ѧϰС��������������ַ���:
����1:�Կ���Ϊ����������ͭ��������B�з������գ�ʹͭ�������ַ�Ӧ��������ͭ���ٽ�����ͭ��ϡ���ᷴӦ��
����2:������������ֱ��ͨ�˵�ͭ����ϡ����Ļ�����У������ڳ����¼�������Ӧ����ӦҺ�м�����FeSO4����������Ӧ����������ͭ����Ӧ��ȫ�����ʼ���pH����Ԫ��ȫ������(һ����Ϊ�����ӵ�Ũ���½���10-5mol��L-1������Ϊ������ȫ)��Ȼ����ˡ�Ũ�����ᾧ��
��֪:Ksp(Cu(OH)2]=10-22, Ksp[Fe(OH)2]=10-16��Ksp[Fe(OH)3]=10-38
��ش��������⣺
�ٷ���1�е�B����������_��
��Ϊ��ʹ��Ԫ��ȫ��������Ӧ����pH����Ϊ_��
�۷���2�м����ʿ�ѡ�õ���_��
| A��CaO | B��NaOH | C��CuCO3 | D��Cu2(OH)2CO3 E�� Fe2(SO4)3 |
 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д� Ӣ�żƻ���ĩ����ϵ�д�
Ӣ�żƻ���ĩ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��15�֣���ͭ���ǹ�ҵ��ͭ����Ҫԭ�ϣ�����Ҫ�ɷ�ΪCuFeS2������һ����Ȼ��ͭ������SiO2����Ϊ�˲ⶨ�û�ͭ��Ĵ��ȣ�ijͬѧ���������ʵ�飺
�ֳ�ȡ��ϸ�Ļ�ͭ����Ʒ1.150g���ڿ��������½������գ�����Cu��Fe3O4��SO2���壬ʵ���ȡd����Һ�� ������ƿ�У���0.05mol/L������Һ���еζ������ı���Һ20.00ml����ش��������⣺
������ƿ�У���0.05mol/L������Һ���еζ������ı���Һ20.00ml����ش��������⣺
��1��������Ʒ���õ�����Ϊ_____(�������ƽ��������ƽ��)������Ʒ��ϸ���ٷ�Ӧ����Ŀ����_______ ��
��2��װ��a��c�����÷ֱ���____��____�����ţ���
A��ȥSO2���� B��ȥ�����е�ˮ���� C������������
D�����ڹ۲�������� E��ȥ��Ӧ����������
��3��������Ӧ����������ͨһ��ʱ��Ŀ�������Ŀ����___________��
��4��ͨ�������֪���û�ͭ��Ĵ���Ϊ________��
��5��������ͼװ���������ʵ��װ��d��ͬ�����Դﵽʵ��Ŀ�ĵ���____������ţ���
��6������ԭװ��d�е���Һ��ΪBa(OH)2����õĻ�ͭ�����Ϊ��1%������ʵ���������ȷ�����ܵ�ԭ����Ҫ��_____________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������ȣ�ClO2����һ�ֹ��ס���Ч��ɱ���������������������Ƶ�ǿ�Ҵ̼�����ζ��������ˮ���е�11�棬���Ȳ��ȶ��ֽⷢ����ը��һ�ֹ�ҵ�Ʒ���������(��Ҫ�ɷ�ΪFeS2)��ԭ��������ȡ�������ȣ�ij�о�С����ʵ�����������װ���Ʊ�ClO2��ˮ��Һ��
��1��������ƿ�м���NaClO3��Һ��ŨH2SO4��ͨ����������ں�������60�棬ͨ������������������ټ����������ĩ����Ӧ��ʼʱ��ƿ�ڷ�����Ӧ�����ӷ���ʽ���£�����ƽ�÷���ʽ����ClO3�� ����FeS2����H������ClO2����Fe3������SO42������H2O
��2��װ��D�������� ��
��3����������Ӧ���ķ�Һ(�����������⣬����Al3����Mg2����H����Cl����)���Ʊ�Ħ���Σ���(NH4)2Fe(SO4)2��6H2O��������������£�
�ش��������⣺
�ٲ����������Ϊ�� ��
���Լ�X�� ��д����������Ҫ��Ӧ�����ӷ���ʽ ��
��Ϊ�ⶨ(NH4)2Fe(SO4)2��6H2O���������ĺ�����ijʵ��С����������ʵ�飺�õ�����ƽȷ����5.000g��������茶��壬���Ƴ�250mL��Һ��ȡ������Һ25.00mL����ƿ�У���ϡH2SO4�ữ����0.01000mol/L KMnO4��Һ�ζ���Fe2+ǡ��ȫ��������Fe3+��ͬʱ��MnO4������ԭ��Mn2+���������KMnO4��Һ��ƽ�����Ϊ21.00mL����þ�����������������Ϊ ��������4λ��Ч���֣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��.(1)ij��ѧ��ȤС����������װ����ѡȡ��Ҫ��װ����ȡ��NH4��2SO4��Һ�����ӵ�˳���ýӿ������ĸ��ʾ���ǣ�a ��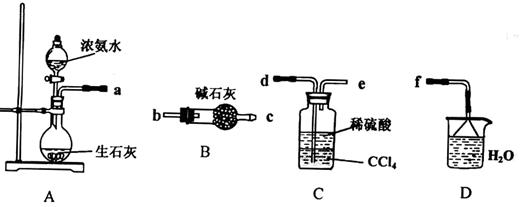
(2)��װ��C������Һ����뿪�IJ���������___ __��װ��D�������� ��
��.�������ƿ������ڸ��Ƶر�ˮ�ʡ��������ؽ������ӷ�ˮ�������ೱ��Ҳ������Ӧ�������ȡ���ҵ�������������Ƶ���Ҫ�������£�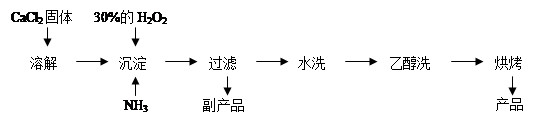
��֪CaO2��8H2O�ʰ�ɫ������ˮ��I2+2S2O32��= 2I��+S4O62��
��1��������������ȡCaO2��8H2O�Ļ�ѧ����ʽ�� ��
��2�����顰ˮϴ���Ƿ�ϸ�ķ����� ��
��3���ⶨ��Ʒ��CaO2�ĺ�����ʵ�鲽���ǣ�
��һ����ȷ��ȡa g��Ʒ����ƿ�У�������������ˮ������b g KI���壬�ٵ�������2 mol/L��H2SO4��Һ����ַ�Ӧ��
�ڶ�������������ƿ�м��뼸�ε�����Һ��
����������μ���Ũ��Ϊc mol��L��1��Na2S2O3��Һ����Ӧ��ȫ������Na2S2O3��ҺV mL��
���жϴ˵ζ�ʵ��ﵽ�յ�ķ����ǣ� ��
��CaO2����������Ϊ (����ĸ��ʾ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ˮ�Ȼ�����һ����Ҫ���л��ϳɴ������������ڽ���ұ�������ͺϳɣ�ʳƷ����ˮ�Ȼ���Ҳ���������ɼ����������ȡ�
ij�о���ѧϰС��������Ϻ�������ͼ��ʾװ���Ʊ���ˮ�Ȼ�����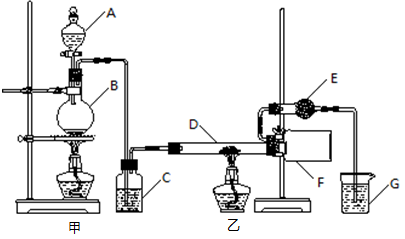
������ʾ���Ȼ���Ϊ��ɫ��������ɫ����dz��ɫ�Ľᾧ�Է�ĩ����178���������������ǵϷ��ӣ�Al2Cl6�����ڿ�����������ˮ�֣�����ˮ�⡣
��1����д�������������ƣ�A ��B ��
��2��װ��C��ʢװ���Լ��� ��װ��G��ʢװ���Լ��� ��
��3����ͬ�����£��Ȼ�������������������ܶ��� ��
��4��װ��F�е������� ��
��5��װ��E�������� ��
��6��ʵ�������Ӧ�ȳ�ȥ ����ס����ҡ������ľƾ��ơ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ѧʵ�����������⻯ѧ֪ʶ���γɻ�ѧ������̽���봴��������������ѧ������
��1����ʵ��������Ũ������MnO2������ȡCl2���������ʵ�顣
��д���÷�Ӧ�����ӷ���ʽ_________________________��
�������ռ�Cl2��װ����ȷװ����_________________________��
�۽�Cl2ͨ��ˮ�У�������Һ�о��������Եĺ���������________________________��
�����ʵ��Ƚ�Cl2��Br2�������ԣ������������ǣ�ȡ����������ˮ��CCl4���Թ��У�____________
_______________________________________________________________________________________��
��2��ij��ѧ��ȤС��Ϊ��̽��AgNO3�����ȶ��ԣ����������ʵ�顣
����ͼ��ʾ��ʵ��װ��A����AgNO3���壬��������ɫ���壬��װ��D���ռ�����ɫ���塣����Ӧ�����Ժ��Թ��в�������Ϊ��ɫ��
��װ��B��������___________________________��
�ھ�С�����۲���֤����ɫ����ΪO2������֤������_____________________��
���������ϡ�Ag2O�ͷ�ĩ��Ag��Ϊ��ɫ��Ag2O�����ڰ�ˮ��
��������衿�Թ��в����ĺ�ɫ��������ǣ�i Ag��ii Ag2O��iii Ag��Ag2O
| ʵ���� | ���� | ���� |
| a | ����������ˮ���� | ��ɫ���岻�ܽ� |
| b | ��������ϡ���ᣬ�� | ��ɫ�����ܽ⣬����������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������ͼװ��̽�������Ͱ���֮��ķ�Ӧ������A��F�ֱ�Ϊ�����������ķ���װ�ã�CΪ�ϴ�������������백����Ӧ��װ�á�
��ش��������⣺
��1��װ��F�з�����Ӧ�����ӷ���ʽΪ ��
��2��װ��A�е���ƿ�ڹ�����ѡ�� ��ѡ������ѡ��Ĵ��ţ�
A����ʯ�� B����ʯ�� C���������� D������������ E���ռ�
��3����A��B��C��F��E��D��C�����ڸ����߿��ڻ��Ϻ��ʵ�װ��ͼ��ע����װ�Լ����ƣ�
��4����C�й۲쵽�������̣�ͬʱ����һ�����嵥�ʣ��÷�Ӧ�Ļ�ѧ����ʽΪ ����Ӧ���������뻹ԭ�������ʵ�����Ϊ �����һ��ʵ�鷽�������ù������Ȼ�泥�Ҫ��д�������������ۣ��� ��
��5����װ��C��G���ݳ���β�����ܺ�����Ⱦ���������壬��δ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ʵ��������ϩʱ��������������ʹBr2�����Ȼ�̼��Һ��ɫ���ס���ͬѧ������ʵ����֤�����������Ѽ��飬���ּг�װ���ԣ���
ʵ�����������
| �� �� | �� �� |
| ��ȼ�ƾ��ƣ� ������170�� | ��A����ƿ��Һ�彥����� ��B����������ð������Һ����ɫ |
| ���� | |
| ʵ����ϣ� ��ϴ��ƿ | ��A����ƿ�ڸ���������ɫ����״��д̼�����ζ�ݳ� |
| | �� �� | �� �� |
| �� | ��A��B������һ��װ��ij���Լ���ϴ��ƿ | Br2��CCl4��Һ��ɫ |
| �� | ��A���ӵ�װ�����£� | D����Һ�ɺ���ɫ��Ϊdz����ɫʱ��E����Һ��ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ʵ��Ŀ���ܴﵽ����( )
| A����Zn�����ԭ����������ͨ����Zn�������Լ���ЩZn������ϡ���ᷴӦʱ�ų�H2����� |
| B����Na2CO3��NaHCO3�������Na2CO3��������������ͨ������Ʒ����������Ʒ����Ba(OH)2��Һ��Ӧ�ij��������� |
| C�����к���ʱ��������ϡ��ˮ����NaOH��Һ |
| D����������Һ����ij���Һ�У������Ƿ��е⻯�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com