
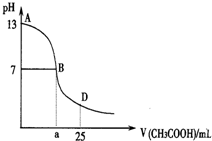
 ��ͼΪ��25mL 0.1mol?L-1NaOH��Һ����εμ�0.2mol?L-1CH3COOH��Һ��������ҺpH�ı仯���ߣ���ش�
��ͼΪ��25mL 0.1mol?L-1NaOH��Һ����εμ�0.2mol?L-1CH3COOH��Һ��������ҺpH�ı仯���ߣ���ش�| 0.1000mol/L��20ml |
| 60ml |
| 1 |
| 30 |
| 1 |
| 30 |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��2H2��g��+O2��g��=2H2O��l����H=+571.6kJ?mol-1 | ||
| B��2H2��g��+O2��g��=2H2O��l������H=-571.6kJ?mol-1 | ||
C��H2��g��+
| ||
D��H2��g��+
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
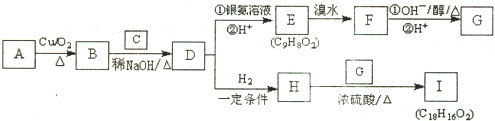
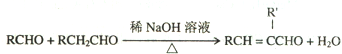
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| +X��һ������ |
| ��Ӧ�� |
| +X��һ������ |
| ��Ӧ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��c��OH-����c��HA����c��HB����c��H+�� |
| B��c��OH-����c��A-����c��B-����c��H+�� |
| C��c��OH-����c��B-����c��A-����c��H+�� |
| D��c��OH-����c��HB����c��HA����c��H+�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �� | HClO4 | H2SO4 | HCl | HNO3 |
| Ka | 1.6��10-5 | 6.3��10-9 | 1.6��10-9 | 4.2��10-10 |
| A���ڱ��������������ᶼû����ȫ���� |
| B���ڱ������и�������������������ǿ���� |
| C���ڱ�����������ĵ��뷽��ʽΪ H2SO4=2H++SO42- |
| D��ˮ�������������ǿ��û����������������������������������ǿ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
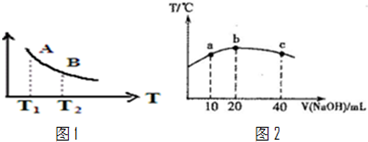
| һ������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
A��
| ||
B��
| ||
C��
| ||
D��
|
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com