
NaCl��NaClO�����������¿ɷ�����Ӧ��ClO��+Cl��+2H+ = Cl2��+H2O��ijѧϰС�����о�����Һ(��Ҫ�ɷ�ΪNaCl��NaClO)�ı��������
��1��������Һ��NaClO�����տ����е�CO2����NaHCO3��HClO�����ʡ�д����ѧ��Ӧ����ʽ ��
��2��ȡ��������Һ�����Թ��У���������һ��Ũ�ȵ����ᣬ������ų���ͨ������װ�ü�������ijɷֿ����ж�����Һ�Ƿ���ʡ�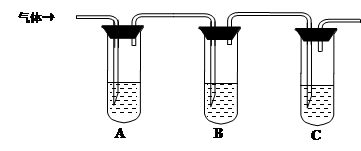
��ѡ�Լ���98%Ũ���ᡢ1%Ʒ����Һ��1.0 mol��L��1 KI-������Һ��1.0 mol��L��1NaOH������ʯ��ˮ������NaCl��Һ
���������ʵ�鷽����
| �����Լ� | Ԥ������ͽ��� |
| �Թ�A�м������� �� �Թ�B�м�1%Ʒ����Һ�� �Թ�C�мӢ� �� | ��A����Һ����ɫ��B����Һ����ɫ��C����Һ����ǡ�������Һ���ֱ��ʣ� �� ������Һδ���ʣ� �� ������Һ��ȫ���ʡ� |
��16�֣�
��1��NaClO+CO2+H2O==NaHCO3+HClO ��3�֣�
��2��(��8��) �����Լ� Ԥ������ͽ��� �� 1.0mol/L KI-������Һ��2�֣�
������ʯ��ˮ��2�֣�����A����Һ����ɫ��B����Һ����ɫ(�ޱ仯)��C����Һ�������(�ޱ仯)��������Һδ���ʣ�2�֣�
����A����Һ������ɫ(�ޱ仯)��B����Һ����ɫ(�ޱ仯)��C����Һ�����������Һ��ȫ���ʣ�2�֣�
��3������ʽ ��2�֣�
�ۣ�2ab �C 5cv��/ 50 ��3�֣�
���������������1�������⣬��Ҫ��Ӧ��ΪNaClO��CO2������ΪNaHCO3��HClO��������Ԫ�ػ��ϼ۱仯�����ݹ۲취��ƽ�ɵã�NaClO+CO2+H2O==NaHCO3+HClO����2������Һ�м�������ϡ����ʱ�����ܷ����ķ�ӦΪClO��+Cl��+2H+=Cl2��+H2O��HCO3��+H+=CO2��+H2O����ų���������ܺ���������������̼����A����Һ�������ɴ��ƶ��Թ�A�м����������1.0mol/LKI��������Һ����Ϊ������KI�ܷ����û���Ӧ���������û����ĵ��ʵ������B��Ʒ����Һ����ɫ��˵��������ȫ��A����Һ���ģ���C�б���ǣ�˵��������һ���ж�����̼���壬�Թ�C�м�����������ʯ��ˮ����Ϊ������̼������������Һ��Ӧ����̼��Ƴ�����ˮ���۸�����֪����ͽ����ƶϣ���A����Һ����ɫ��B����Һ����ɫ��C����Һ������ǣ�������Һδ���ʣ�����A����Һ������ɫ��B����Һ����ɫ��C����Һ����ǣ�������Һ��ȫ���ʣ���3���٢����Ը��������Һ�����ԣ����Ӧ��װ����ʽ�ζ����У������⣬����n=c?V�������������ܵ����ʵ���Ϊab��10��3mol��ÿ�εζ����ĵĸ������Ϊ cv��10��3mol������2KMnO4+5Na2SO3+3H2SO4=K2SO4+2MnSO4+5Na2SO4+3H2O�и����ʵ�ϵ��֮�ȵ������ʵ���֮�ȣ����������������������Ϊ5cv/2��10��3mol��������������������������Ϊ��ab��10��3��5cv/2��10��3��mol������NaClO+Na2SO3=NaCl+Na2SO4�и����ʵ�ϵ��֮�ȵ������ʵ���֮�ȣ����������Ϊ��ab��10��3��5cv/2��10��3��mol������c=n/V��V=0.025L����������Ƶ����ʵ���Ũ��Ϊ��ab��10��3��5cv/2��10��3��mol��0.025L ="(2ab" �C 5cv)/ 50mol/L��
���㣺���黯ѧʵ�鷽������ƺͲⶨ��Ʒ���ȵĻ�ѧ���㣬�漰����������Һ�������̼�ķ�Ӧԭ�������ʵ�鷽��̽������������Һ�Ƿ���ʡ��ζ��ܵ�ѡ�ⶨ����������Һ�����ʵ���Ũ�ȡ����ʵ����ڻ�ѧ����ʽ�е�Ӧ�õȡ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ̽��Cl2��Ư�۵��Ʊ����й����ʣ�ij��ȤС����Ʋ�����������ʵ��̽������ش��������⣺
��1��ʵ������������װ���Ʊ����﴿�����������밴������������������ķ��������������ӣ�H��_______��_______��_______��_______��_______�����й��ƿ���е��Լ�Ϊ_______��

��2��д����ҵ����������ʯ������ȡƯ�۵Ļ�ѧ��Ӧ����ʽ_______��
��3��ʵ������һƿ�ܷⲻ�ϵ�Ư����Ʒ�����п϶�����CaCl2�������ʵ�飬̽������Ʒ�г�CaCl2����е������������ʡ�
������������衣
����1����Ư��δ���ʣ�������Ca(ClO)2
����2����Ư��ȫ�����ʣ�������______��
����3����Ư�۲��ֱ��ʣ�������Ca(ClO)2��CaCO3��
�����ʵ�鷽��������ʵ�顣�����±���д��ʵ�鲽�衢Ԥ������ͽ��ۡ�
��ѡ�õ�������ҩƷ���Թܡ��ιܡ������ܵĵ�����������ˮ������ˮ��Ʒ����Һ��1 mol��L-1 HCl��Һ�����Ƴ���ʯ��ˮ������ʾ�����ؼ���Ca2+��Cl-����
| | ʵ�鲽�� | Ԥ����������� |
| ����1 | ȡ��������Ư�����Թ��У��ȼ��� �ܽ���ٰ����ɵ�����ͨ�� �� | �� �������1������ �� �������2�����3������ |
| ����2 | ��ȷ��Ư�۱��ʣ�����ȡ��������Ư�����Թ��У��ȼ�������1 mol��L-1 HCl��Һ���ټ��� �� | �� �������2������ �� �������3������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�����Ȼ����ڹ�ҵ�Ͽ������Ʊ����ֺ��صĻ�������������գ�
��1����ҵ�Ȼ����к���Fe3+��SO42����Br�����������ӣ��ɰ����²�����о��ƣ���ɸ������ݡ�
���ܽ⣻�ڼ����Լ���Fe3+��SO42��������ȫ����У���_____________________���ܼ����������pH��
��___________________����Br���������������ա�
������У����μ���ij�������NH3��H2O��________��________��
֤��Fe3+�ѳ�����ȫ�IJ�����_________________________________________________��
��2�����˳����ù�ҵ�ƴ���ԭ�����Ʊ�K2CO3������KCl��Һ������ͨ��������______��______�������壬��ַ�Ӧ���а�ɫ�������������õ��İ�ɫ����ϴ�Ӻ����գ�������κι���������Ҳ�����������ʹ����ʯ��ˮ����ǡ�
д�����ɰ�ɫ����Ļ�ѧ����ʽ��___________________________________________��
�����÷����ò���K2CO3��ԭ�������_______________________________________��
��3�� ���Ȼ����Ʊ��������صij��÷��������ӽ���Ĥ��ⷨ������������_________����������Ϊ�˱�����������䷢������Ӧ��λ�ڵ����м�����ӽ���ĤӦ��ֹ_______��������������������С�������ͨ����
��4����ѧ�����������һ�����Ȼ������������صķ������䷴Ӧ�ɷ�Ϊ5�������ɲ��Ѻϲ���������ʡ�ԣ�����д���ڢݲ���Ӧ�Ļ�ѧ����ʽ��
�ڢ١��ڲ����ϲ�����2KCl + 4HNO3�� 2KNO3 + Cl2 + 2NO2 + 2H2O
�ڢۡ��ܲ����ϲ�����4KNO3 + 2H2O �� 4KOH + 4NO2 + O2
�ڢݲ���____________________________________________
�ܷ�Ӧ��4KCl + O2 + 2H2O �� 4KOH + 2Cl2
���ⷨ��ȣ��÷��������ƿ�����______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijʵ��С�����ͨп�̷ϸɵ���ڵĺ�ɫ�������̽����������·�����
��֪��I����ͨп�̵�صĺ�ɫ������Ҫ�ɷ�ΪMnO2��NH4Cl��ZnCl2�����ʡ�
II��������пΪ��ɫ��ĩ��������ˮ�������ᡢǿ����Һ�Ͱ�ˮ��
��ش��������⣺
��1���ڲ�����������___________��
��2��ijͬѧ������ҺA�ijɷֺ���NH4Cl��ZnCl2���������һ��ʵ�鷽������֤�������ȷ��Ҫ���ڴ���ϰ��±���ʽд��ʵ�������Ԥ������ͽ��ۡ�
��ѡ�Լ�������ˮ��2moL��L��1 HCI ��2 moL��L��1 HNO3 ��2 moL��L��1 NH3��H2O��6 moL��L��1 NaOH��0.1 moL��L��1 KSCN��0.1 moL��L��1 BaCl2��0.1 moL��L��1 AgNO3����ɫʯ����Һ����ɫʯ����ֽ
| ʵ����� | Ԥ������ | ���� |
| ����1����ȡ������ҺA��װa��b��c��֧�Թܣ���a�Թܣ�__ __________________________ | �а�ɫ�������� | ˵����ҺA����Cl�� |
| ����2����b�Թܣ�__________ __________________________ | ______________________ | _______________________ |
| ����3����c�Թܣ�__________ __________________________ | �Ȳ���_______________, ��____________________ | ˵����ҺA����Zn2+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij������Ni���ϴ�������Ҫ����Ni��������Al��Al203��Fe�������������ᡢ������ʡ�����������������������ʽ��ȫ����ʱ��Һ��pH���£�
���Ӻ����ϴ������Ƶ�NiSO4��7H2O���壬���������£�
�����������Ϣ������ͼ���ش��������⣺
��1�����������Ŀ���dz�ȥ�����ϴ����е�___ _��
��2���������ʱ�����������___ _���������Һ���п��ܺ��еĽ���������___ ���������ӷ��ű�ʾ����
��3������pHΪ2��3��Ŀ����___ _��
��4������A�IJ���˳���Ǽ���Ũ������ȴ��____��____��
��5��NiSO4��ǿ����Һ����NaC1O���������Ƶü������ӵ�ص缫����NiOOH���÷�Ӧ�����ӷ���ʽΪ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���ͷ���һ����������Ʒ����ʳƷ�Ļ�ѧ���ɼ�����С�մ���(̼�����)�������е�����������ɡ�ij�о���ѧϰС��Ϊ̽����ͬƷ�Ƶķ��ͷ۵Ļ�ѧ�ɷ֣���������ʵ�顣
��������衿
��1������1����С�մ�ͳ�����ɣ�
����2����С�մ��������ɣ�
����3����________________��ɡ�
�����������̡�
Ϊ̽��ijƷ�Ƶķ��ͷ۵Ļ�ѧ�ɷ֣�ijͬѧ�������ʵ�飬�õ���������
��2���÷��ͷ۵ijɷ�Ϊ________ (�ѧʽ����
��3����һƷ�Ƶķ��ͷ۵Ļ�ѧ��ɿ���Ϊ����2������������ʵ����֤��д��ʵ�鲽�衢Ԥ������ͽ��ۡ�
| ʵ�鲽�� |  Ԥ��������� Ԥ��������� |
| 1��ȡ������Ʒ����ϡ�������Һ�ֳ����� | |
| 2�� _______________________________________ | |
| 3�� ________________________________________ | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ҵ��������Ĺ������£�
���������գ�
��1������ˮ������������Mg2+��Ca2+�������������A��B������(A��Դ��ʯ��Ҥ�������������B �Ļ�ѧʽΪ ��
��2��ʵ����ģ������Һ�Ʊ�������װ�����£�
��ͼ1��װ�ú�ͼ2��װ�õ����ӷ���Ϊa�� ��b�� ��f��c��
��ͼ2���Լ�ƿ�ڷ����Ļ�ѧ��Ӧ����ʽΪ ��
��ʵ����Ҫ��ͨ���NH3����֮����ͨ��CO2���壬����ͨ���NH3�ѹ�����ʵ������� ��
��3�����������պ�Ĵ����к���δ�ֽ��̼�����ơ�ijͬѧ��ȡ�ô�����Ʒm g���ٳ�ּ������������ٱ仯ʱ�Ƶ�ʣ����������Ϊn g������Ʒ��̼���Ƶ���������Ϊ ��
��4������25���£�0.1mol/LNH3��H2O��Һ��0.1mol/LNH4Cl��Һ����������Һ�������ϲ����Һ��pH=9������˵����ȷ���� ������ţ���
a��0.1mol/L NH4Cl��Һ���Ϻ���Һ�е������ӵ��������Ŀ����ͬ
b����Ϻ����Һ�У�c(NH3��H2O)��c(Cl-)��c(NH4+)��c(OH-)��c(H+)
c���������֪��NH3��H2O�ĵ���̶ȴ���ͬŨ�ȵ�NH4Cl��ˮ��̶�
d�����ǰ������Һ��pH֮�ʹ���14
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ʵ���ܴﵽ��ӦĿ�ĵ���
| A����ͼ��װ����ȡ���ռ����� | B����ͼ��װ����ȡ���ռ���ϩ |
| C����ͼ��װ�ý��������ճɻ� | D����ͼ��װ����ȡ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ҩƷ��װ�ú������������Ӧʵ����ǣ� ��

| A����Ȫʵ�� |
| B��ʵ������ȡ���ռ����� |
| C���Ʊ����������� |
| D����֤�����Ƿ���̼̼˫�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com