
ij������Ni���ϴ�������Ҫ����Ni��������Al��Al203��Fe�������������ᡢ������ʡ�����������������������ʽ��ȫ����ʱ��Һ��pH���£�
���Ӻ����ϴ������Ƶ�NiSO4��7H2O���壬���������£�
�����������Ϣ������ͼ���ش��������⣺
��1�����������Ŀ���dz�ȥ�����ϴ����е�___ _��
��2���������ʱ�����������___ _���������Һ���п��ܺ��еĽ���������___ ���������ӷ��ű�ʾ����
��3������pHΪ2��3��Ŀ����___ _��
��4������A�IJ���˳���Ǽ���Ũ������ȴ��____��____��
��5��NiSO4��ǿ����Һ����NaC1O���������Ƶü������ӵ�ص缫����NiOOH���÷�Ӧ�����ӷ���ʽΪ________��
��13�֣���1��Al��Al2O3��2�֣� ��2��H2SO4��2�֣���Ni2����Fe2����2�֣�
��3����ֹ��Ũ���ᾧ������Ni2+ˮ�⣨2�֣� ��4���ᾧ�����ˣ�2�֣�
��5��2Ni2+��ClO-��4OH-��2NiOOH����Cl-��H2O��3�֣�
���������������1�����������������Ϊ�˳�ȥ������������Al2O3�����������������Ժ�ǿ�Ӧ���ܽ�õ�ƫ�����Σ���Ӧ�����ӷ���ʽ�ֱ�Ϊ2Al+2OH-+2H2O��2AlO2-+3H2����Al2O3+2OH-��2AlO2-+3H2O��
��2���������ʱ��Ҫ���ܽ��������������ʼ�������������Ʊ�Ŀ���ǵõ�NiSO4?7H2O����˼������������µ����ʣ�������Ҫ������������������������ϡ���ᷴӦ����������������������Һ�������Һ���п��ܺ��еĽ���������Ni2+��Fe2+��
��3����������Һ��Ҫ����Ũ���ᾧ������Ϊ��ֹ������ˮ����������������������Ҫ������ҺpH��
��4������A�IJ���˳���Ǽ���Ũ������ȴ���ᾧ�����ˡ�
��5��NiSO4��ǿ����������NaClO���������Ƶü������ӵ�ص缫����NiOOH����˸÷�Ӧ�����ӷ���ʽ��2Ni2+��ClO-��4OH-��2NiOOH����Cl-��H2O��
���㣺�������ʵķ�����ᴿ����ѧʵ�����������ʵ�鷽������Լ�ʵ���������Ƶ��ۺ�Ӧ�õ�


 ��ѧ�̸̳����¿α�ϵ�д�
��ѧ�̸̳����¿α�ϵ�д� Сѧ��ʱ��ѵϵ�д�
Сѧ��ʱ��ѵϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ľ���к��п����Լ���(��Ҫ�ɷ���K2SO4��K2CO3��KCl)��ijѧ�������в�����ȡ��ľ���еļ��Σ���ȡ��ľ�Ҽ�ˮ�ܽ⣻�ڹ���ȡ��Һ����������Һ������ȴ�ᾧ��
(1)�ڲ����١��ڡ����о��õ�����������������Ŀ�������� �� �� ��
(2)Ϊ�����ľ���е������ӣ�ȡ������������ˮ��������Һ�ֳ����ȷݣ�
��ȡһ����Һ����������HCl���۲쵽 ��֤������̼�����
��ȡ��һ����Һ��Ϊ����SO42����Ӧ����Һ�м��� ��
��ȡʣ���һ����Һ�������������������۲쵽�г��������������ɴ�֤����Һ��һ����Cl��������Ϊ�������õĽ����Ƿ����ܣ� ��Ϊʲô�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
S2Cl2�ǹ�ҵ�ϳ��õ�����ʵ�����Ʊ�S2Cl2�ķ�����2�֣�
�� CS2+3Cl2 CCl4+S2Cl2���� 2S+Cl2
CCl4+S2Cl2���� 2S+Cl2 S2Cl2��
S2Cl2��
��֪S2Cl2����Ԫ����+1�ۣ�����ʽ�� �������ȶ�����ˮ������᪻���Ӧ��һ������Ԫ�ؼ�̬���ߣ�һ���ֽ��ͣ�����Ӧ�漰�ļ������ʵ��۷е����£�
�������ȶ�����ˮ������᪻���Ӧ��һ������Ԫ�ؼ�̬���ߣ�һ���ֽ��ͣ�����Ӧ�漰�ļ������ʵ��۷е����£�
| ���� | S | CS2 | CCl4 | S2Cl2 |
| �е�/�� | 445 | 47 | 77 | 137 |
| �۵�/�� | 113 | -109 | -23 | -77 |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�����״���һ����Ҫ�л��ϳ��м��壬����ͨ������ԭ�����кϳɣ� 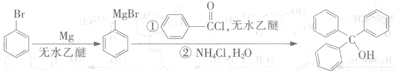
ʵ�鲽�����£�
����ͼ1��ʾ����������ƿ�м���A,1.5 gþм���ڵ�Һ©���м���,6.7 mL�屽����ˮ���ѣ��ȼ���1��3���Һ����ƿ�У���þм���������ݲ���ʱ����ʼ���裬����μ������µĻ��Һ��ʹ��ӦҺ������״̬������Ӧ��ȫ��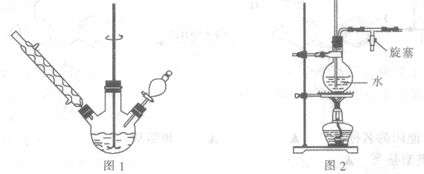
�ڼ�����,A,3.7 g�������Ⱥ���ˮ���ѣ�ˮԡ����l h ������ˮԡ�������μ��Ȼ�淋ı�����Һ30.0 mL��
�۽�������ƿ�ڵ�����ˮԡ������ȥ���ѣ�Ȼ��ӣ�A,30.0 mLˮ����ˮ��������(ͼ2����ˮ��������װ��)��ֱ������״��������
����ȴ���ˣ���������80.0�����Ҵ���Һ�ؽᾧ�����յõ���Ʒ��
��1��������С���μ������µĻ��Һ����Ϊ�˷�ֹ ��
��2����ˮ��������װ���г��ֶ���������ͼ2��������ˮλ��ͻȻ���ߣ���ʱӦ ���������ų����ٽ�������
��3�������õ�����Ҫ���������á� ������������ͨ���˵������ص��� ��
��4�����������80.0�����Ҵ���Һ�ؽᾧ��Ŀ���� ��
��5���ù����ɼ������ò����Ƿ�������������÷����к��к��ֻ�ѧ����������Ϣ�ķ��� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ư����һ�ֳ��õ���������
��1����ҵ������Ư�۷�Ӧ�Ļ�ѧ����ʽΪ��________________ __��Ư�۵���Ч�ɷ�Ϊ ��
��2��ij̽��С����г��Ϲ�����һ����װ�����Ư�ۣ��Ը�Ư�۵ijɷֽ���̽�������������Լ������ʵ�鷽��������ʵ�顣���ڴ�������ʵ�鱨�档
��ѡ�Լ���2mol��L��1NaOH��Һ��2mol��L��1HCl��Һ��2mol��L��1HNO3��Һ��0.5mol��L��1BaCl2��Һ��0.01mol��L��1AgNO3��Һ������ʯ��ˮ��ʯ����Һ����̪��Һ������ˮ��
| ʵ�鲽�� | Ԥ����������� |
| ����1��ȡ����Ư��������������ˮ����ֽ��裬���ã����ˣ��ó�������Һ�� | |
| ����2���������������2mol��L��1HCl��Һ��������������ͨ�� | ���� ���ۣ� |
| ����3��ȡ��Һ��װA��B��֧�Թܡ���A�Թܣ� | ������Һ�ȱ��ɫ��Ȼ����ɫ�� ���ۣ� |
| ����4����B�Թܣ� | ��������ɫ������ ���ۣ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
NaCl��NaClO�����������¿ɷ�����Ӧ��ClO��+Cl��+2H+ = Cl2��+H2O��ijѧϰС�����о�����Һ(��Ҫ�ɷ�ΪNaCl��NaClO)�ı��������
��1��������Һ��NaClO�����տ����е�CO2����NaHCO3��HClO�����ʡ�д����ѧ��Ӧ����ʽ ��
��2��ȡ��������Һ�����Թ��У���������һ��Ũ�ȵ����ᣬ������ų���ͨ������װ�ü�������ijɷֿ����ж�����Һ�Ƿ���ʡ�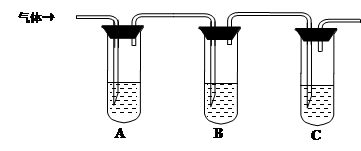
��ѡ�Լ���98%Ũ���ᡢ1%Ʒ����Һ��1.0 mol��L��1 KI-������Һ��1.0 mol��L��1NaOH������ʯ��ˮ������NaCl��Һ
���������ʵ�鷽����
| �����Լ� | Ԥ������ͽ��� |
| �Թ�A�м������� �� �Թ�B�м�1%Ʒ����Һ�� �Թ�C�мӢ� �� | ��A����Һ����ɫ��B����Һ����ɫ��C����Һ����ǡ�������Һ���ֱ��ʣ� �� ������Һδ���ʣ� �� ������Һ��ȫ���ʡ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijͭ��ʯ��ͭԪ�غ����ϵͣ��Һ�������þ���Ƶ��������ӡ�ijС����ʵ�������ý���-��ȡ���Ʊ�����ͭ��
��1������IΪ_______������II�õ��IJ����������ձ�_______
��2������II������III����ҪĿ����_______������ͭԪ�ء�
��3��С���Ա����CuSO4��Һ��Na2CO3��Һ��Ϸ�Ӧ���Ʊ�������ľ�ķ�����Cu2(OH)2CO3����Һ�����ʵ�鷢��������ɫ����Һ��ɫ���в��죬�������ϱ��������������������Ʋ�ͬʹ���л��н϶�Cu(OH)2��Cu4(OH)6SO4��
��֪Cu(OH)2��Cu2(OH)2CO3��Cu4((OH)6SO4��������ˮ����������ֽ��¶�����Ϊ 80�桢200�桢300�档
���ʵ���������Һ�ɷ֣���ɱ������ݡ�
��ѡ�Լ���2mol?L��1HCl��1 mol?L��1H2SO4��0.1 mol?L��1NaOH��0.1 mol?L��1 BaCl2������ˮ����������Ʒ��ѡ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ��������Һ�����ˣ����ϴ�Ӻ�ȡ�������Թ��У�_______________________________________________________________ | ˵������Һ�л�__________________________________________,��Cu4( OH)6SO4�� |
| ����2����ȡ��������Һ���Թ��У�____________________________ | ______________�� ˵������Һ�л���Cu( OH) 2�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
ʵ������ֻ���ռ���ᡢͭƬ������ʯ������ˮ�����Լ�����Ƿȱ�Լ��ĽǶ�����(ʵ��������ȫ)�������е�ʵ����Ŀ��( )
| A����ȡ���� | B����ȡ̼���� | C����ȡ�Ȼ�ͭ | D���ⶨ�����Ũ�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com