
���ͷ���һ����������Ʒ����ʳƷ�Ļ�ѧ���ɼ�����С�մ���(̼�����)�������е�����������ɡ�ij�о���ѧϰС��Ϊ̽����ͬƷ�Ƶķ��ͷ۵Ļ�ѧ�ɷ֣���������ʵ�顣
��������衿
��1������1����С�մ�ͳ�����ɣ�
����2����С�մ��������ɣ�
����3����________________��ɡ�
�����������̡�
Ϊ̽��ijƷ�Ƶķ��ͷ۵Ļ�ѧ�ɷ֣�ijͬѧ�������ʵ�飬�õ���������
��2���÷��ͷ۵ijɷ�Ϊ________ (�ѧʽ����
��3����һƷ�Ƶķ��ͷ۵Ļ�ѧ��ɿ���Ϊ����2������������ʵ����֤��д��ʵ�鲽�衢Ԥ������ͽ��ۡ�
| ʵ�鲽�� |  Ԥ��������� Ԥ��������� |
| 1��ȡ������Ʒ����ϡ�������Һ�ֳ����� | |
| 2�� _______________________________________ | |
| 3�� ________________________________________ | |
��17�֣�
��1�����ۺ����� ��2�֣�
��2��NH4HCO3��NaHCO3 ��2�֣�
��3����8�֣�ʵ�鲽��  Ԥ���������
Ԥ���������1��ȡ������Ʒ����ϡ�������Һ�ֳ����� 2���ù���������˿��1�֣�պȡ����һ����Һ�ھƾ����������գ��۲������ɫ��1�֣�____ ����ʻ�ɫ��1�֣����÷��ͷ��к���NaHCO3��1�֣� 3�� ������һ����Һ�еμ�������1�֣�BaCl2��Һ��1�֣���
��������1��2�ξ��ɵ�1�֣�
�Լ�BaCl2�÷֣�Ba��NO3��2��Ba��0H��2���÷֣���д����HCl�ữ���۷֣� �а�ɫ�������ɣ�1�֣�����ϲ���2�Ľ��ۼ������1�֣�����
��4��100mL����ƿ�� 2�֣� 2NaHCO3��3NH4HCO3 (3��)
���������������1�����������֪����ͬƷ�Ƶķ��ͷۿ���С�մ�ͳ�����ɣ�Ҳ������С�մ��������ɣ��������ɳ��ۺ�������ɣ�������֪�ļ���1������2�ƶϼ���3Ϊ���ۺ���������2������NaHCO3��NH4HCO3��Al2(SO4)2?12H2O����Ҫ���ʿ�֪��NH4HCO3����Σ�����ʯ�һ����ĥ���Էų���������������������ʯ����ĥ�����ܷų����壬��AΪNH3�����ͷۼ�һ�����г��ۣ�NH4HCO3�����ּ��Ⱥ���ȫ��Ϊ�����ݳ���NaHCO3���������ȱ�ΪNa2CO3�����CO2��H2O��Na2CO3������������Ӧ���ɶ�����̼���塢NaCl��H2O����Al2(SO4)2?12H2O����ˮ�����Al(OH)3�������϶�������������ɴ��ƶ�BΪCO2���÷��ͷ�һ������С�մ����Լ�Ʒ�Ƶķ��ͷ۵���Ҫ�ɷ���NaHCO3��NH4HCO3����3������ʵ�鷽���в���2�Ľ������ƿ�֪������2���ýྻ�IJ�˿պȡA�е���Һ���پƾ������������գ��������ɫ�ʻ�ɫ��֤����Na+�����ͷ�����NaHCO3�����ڼ���2�Ƿ��ͷ���С�մ��������ɣ�����3�Ľ�����֤�����ͷ���Al2(SO4)2?12H2O�����������ʼ��ṩ�Լ��������ƶϣ�����3�����ʵ�鷽��֤�����ͷ��к���Al3+�����Ӧ��B�Թ�����εμ�0.1mol/LNaOH��Һ���۲쵽��ɫ�������Ȳ�����ɫ����������ܽ⣬֤����Al3+�����ͷ�������������4��������Һ��Ҫ���������������ܽ⡢ת�ơ�ϴ�ӡ����ݵȲ��裬������100 mL��Һ��Ҫ���ܽ⡢��ȴ�����Һ����100mL����ƿ�У�25.00mL������ȫ��Ӧ������CO2+Ca(OH)2=CaCO3��+H2O�����ɫ����Ϊ̼��ƣ�����n=m/M����n(CaCO3)=1.25g��100g/mol=0.0125mol������100mL������Һ�������25.00mL������Һ֮��Ϊ100/25.00����100mL������Һ���������ᷴӦ�ų��Ķ�����̼Ϊ0.0125mol��100/25.00=0.05mol���跢�ͷ���NaHCO3��NH4HCO3�ֱ�Ϊxmol��ymol������m=n��M����84x+79y=4.05����������������NaHCO3+HCl=NaCl+CO2��+H2O��NH4HCO3+HCl=NH4Cl+CO2��+H2O����xmolС�մ���ȫ��Ӧ�ų�xmol������̼���壬ymol̼�������ȫ��Ӧ�ų�ymol������̼����x+y=0.05����������������⣬��x=0.02��y=0.03��˵���÷��ͷ���С�մ��̼����淋����ʵ���֮��Ϊ0.2��0.3=2��3�����Ը÷��ͷ۵���ɿ��Ա�ʾΪ2NaHCO3��3NH4HCO3��
���㣺����̽��ʵ�鷽������Ƽ���ѧ���㣬�漰Ԫ�ػ���������ʡ�������衢���ʵ�鷽����֤���衢��ѧ������ɵĻ�ѧ���������Һ�������������ʵ����ڻ�ѧ����ʽ�����е�Ӧ�õȡ�


 �ŵ������ϵ�д�
�ŵ������ϵ�д� 53������ϵ�д�
53������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ʵ������ȡ���ᶡ����ʵ��װ�������¼ס�������װ�ÿɹ�ѡ�á�

���ף� ���ң�
�Ʊ����ᶡ�����漰���й����ʵ��������ʼ��±�
| | ���� | 1������ | ���ᶡ�� |
| �۵�(��) | 16��6 | ��89��5 | ��73��5 |
| �е�(��) | 117��9 | 117 | 126��3 |
| �ܶ�(g/cm3) | 1��05 | 0��81 | 0��88 |
| ˮ���� | ���� | ���� (9g/100gˮ) | �� |





�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
̼�����׳ƴ������;�ܹ㡣ʵ�����У���̼����狀ͱ���ʳ��ˮ���Ƶô���������ڲ�ͬ�¶��µ��ܽ�ȼ�����
  �¶ȡ� �¶ȡ��ܽ�� ���� g/100gˮ | 10 | 20 | 30 | 40 | 50 | 60 | 70 |
| NaCl | 35.8 | 36.0 | 36.3 | 36.6 | 37.0 | 37.3 | 37.8 |
| NH4HCO3 | 15.8 | 21.0 | 27.0 | | | | |
| NaHCO3 | 8.2 | 9.6 | 11.1 | 12.7 | 14.4 | 16.4 | |
| NH4Cl | 33.3 | 37.2 | 41.4 | 45.8 | 50.4 | 55.2 | 60.2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�ִ�п��Ʒ�ӹ���ҵ���յķ���������ZnO��FeO��Fe2O3��CuO��Al2O3�����ʣ�����ȡ����п���������£�
�й�����������ȫ������pH���±���
| ������ | Al(OH)3 | Fe(OH)3 | Fe(OH)2 | Cu(OH)2 | Zn(OH)2 |
| pH | 5.2 | 3.2 | 9.7 | 6.7 | 8.0 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
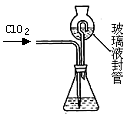
NaCl��NaClO�����������¿ɷ�����Ӧ��ClO��+Cl��+2H+ = Cl2��+H2O��ijѧϰС�����о�����Һ(��Ҫ�ɷ�ΪNaCl��NaClO)�ı��������
��1��������Һ��NaClO�����տ����е�CO2����NaHCO3��HClO�����ʡ�д����ѧ��Ӧ����ʽ ��
��2��ȡ��������Һ�����Թ��У���������һ��Ũ�ȵ����ᣬ������ų���ͨ������װ�ü�������ijɷֿ����ж�����Һ�Ƿ���ʡ�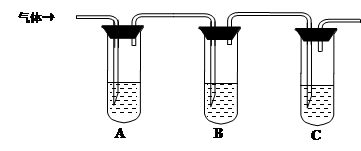
��ѡ�Լ���98%Ũ���ᡢ1%Ʒ����Һ��1.0 mol��L��1 KI-������Һ��1.0 mol��L��1NaOH������ʯ��ˮ������NaCl��Һ
���������ʵ�鷽����
| �����Լ� | Ԥ������ͽ��� |
| �Թ�A�м������� �� �Թ�B�м�1%Ʒ����Һ�� �Թ�C�мӢ� �� | ��A����Һ����ɫ��B����Һ����ɫ��C����Һ����ǡ�������Һ���ֱ��ʣ� �� ������Һδ���ʣ� �� ������Һ��ȫ���ʡ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
FeSO4��7H2O�׳��̷����㷺����ҽҩ��������
��1����֪FeSO4��7H2O�����ڼ��������·������·�Ӧ��
 ��������ͼװ�ü���÷�Ӧ��������
��������ͼװ�ü���÷�Ӧ��������
����д���пհף�
������������˳��Ϊ ����a��i����ĸ��ʾ����
��װ��C�е��Լ�X��ѧʽΪ ����װ������ˮ�������� ��
��2��������FeSO4��7H2O��ʵ�����Ʊ�����ͼ��
�����������������գ�
�ٷ�Ӧ����Ҫl00mL l��5mol��L-1l��ϡ�����ܽ�ྻ����м������������Ϊ98%���ܶ�Ϊ1.84g��cm-3��Ũ�������ơ����õ���������Ͳ���ձ�������������ͷ�ιܼ� ����ȡŨ��������Ϊ ��
�ڷ�Ӧ1��Ҫ���������ӣ���ԭ���� ������AΪ ��
�۲ⶨFeSO4��7H2O��Ʒ��Fe2+�����ij��÷�����KMnO4��Һ�ζ�������֪��ȡ3.0g FeSO4��7H2O��Ʒ�����Ƴ���Һ���������ữ��0.01000moL��L-1 KMnO4��Һ�ζ�������KMnO4��Һ�����Ϊ200.00mL��������Ӧ�����ӷ���ʽΪ ������������Ʒ��FeSO4��7H2O����������Ϊ ��������λ��Ч���֣���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������ȣ�ClO2����Ŀǰ�����Ϲ��ϵĵ��Ĵ���Ч�����Ĺ�������������һ�ֻ���ɫ�����壬������ˮ��ʵ���ҿ���NH4Cl�����ᡢNaClO2���������ƣ�Ϊԭ�����Ʊ�ClO2�����������£�
��1��д�����ʱ������Ӧ�Ļ�ѧ����ʽ�� ��
��2����ȥClO2�е�NH3��ѡ�õ��Լ��� ��������ĸ��
| A������ʳ��ˮ | B����ʯ�� | C��Ũ���� | D��ˮ |
| ʵ�鲽�� | ʵ������ | ʵ����� |
| �� | | ��ҺX�к���Na+ |
| �� | | ��ҺX�к���Cl�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�����±��ṩ��������ҩƷ�����ܴﵽ��Ӧʵ��Ŀ�ĵ���
| ��� | ���� | ҩƷ | ʵ��Ŀ�� |
| A | ������ƽ�������룩��250mL����ƿ����Ͳ���ձ���ҩ�ס������� | NaOH���塢����ˮ | ����250mLһ�����ʵ���Ũ�ȵ�NaOH��Һ |
| B | ��Һ©������ƿ�����ܼ���Ƥ�� | ϡ���ᡢ̼���ơ���������Һ | ֤���ǽ����ԣ�S��C��Si |
| C | ��ʽ�ζ��ܡ���ʽ�ζ��ܡ���ͷ�ιܡ�����̨�������У�����ƿ | ��֪Ũ�ȵ�NaOH��Һ���������ᡢ����ˮ����ֽ | �ⶨϡ��������ʵ���Ũ�� |
| D | ����̨�������У����ƾ��ơ����Թܡ�����ƿ�����ܼ���Ƥ�� | �Ȼ�� | ��ȡ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��ʵ����������м������ͭ��ϡ����Ϊԭ���Ʊ�ͭ������������;����
��1��Fe H2
H2 Cu
Cu
��2��CuO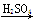 CuSO4
CuSO4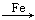 Cu
Cu
���������ַ����Ƶõ�����ͭ���������й�˵������ʵ��������ǣ� ��
| A����������������ͬ |
| B�����������������ͬ |
| C����������������������ͬ |
| D����������ͭ��������ͬ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com