
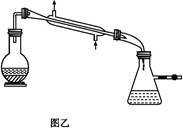
�������ȣ�ClO2����һ�ֻ���ɫ�д̼�����ζ�����壬���۵�Ϊ-59�棬�е�Ϊ11.0�棬������ˮ����ҵ�����Գ�ʪ��KClO3�Ͳ��ᣨH2C2O4����60��ʱ��Ӧ�Ƶá�ijѧ����������ͼ��ʾװ��ģ�ҵ��ȡ���ռ�ClO2��
��1��A�з�Ӧ������K2CO3��ClO2��CO2�ȣ���д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
��2��A���������¶ȿ���װ�ã����ƾ��ơ��ձ��⣬����Ҫ�IJ��������� ��BҲ���������¶ȿ���װ�ã�Ӧ���� (ѡ���ˮԡ������ˮԡ��)װ�á�
��3����Ӧ����װ��C�пɵ�NaClO2��Һ����֪NaClO2������Һ�����¶ȵ���38��ʱ����������NaClO2��3H2O�����¶ȸ���38��ʱ����������NaClO2��������ͼ��ʾ��NaClO2���ܽ�����ߣ��벹���NaClO2��Һ���Ƶ�NaClO2����IJ������裺
�� ���� ����ϴ�ӣ��ܸ��
��4��ClO2�ܲ��ȶ������������ƣ���ˮ���յõ�ClO2��Һ��Ϊ�ⶨ������Һ��ClO2�ĺ���������������ʵ�飺
����1��ȷ��ȡClO2��Һ10 mL��ϡ�ͳ�100 mL��������ȡV1mL����������ƿ�У�
����2������������pH��2.0������������KI���壬����Ƭ�̣���ʱ���������ӷ���ʽΪ�� ��
����3���������ָʾ������c mol/L Na2S2O3��Һ�ζ����յ㣬����Na2S2O3��ҺV2 mL����ԭClO2��Һ��Ũ��Ϊ g��L���ú���ĸ�Ĵ���ʽ��ʾ����
����֪2 Na2S2O3+I2= Na2S4O6+2NaI��
��1��2KClO3 + H2C2O4  K2 CO3 + CO2��+ 2ClO2��+ H2O��2�֣�
K2 CO3 + CO2��+ 2ClO2��+ H2O��2�֣�
��2���¶ȼƣ�1�֣� ��ˮԡ��1�֣�
��3���������ᾧ��1�֣� �ڳ��ȹ��ˣ�1�֣�
��4����2ClO2 + 10I�� �� 8H+ = 2Cl����5I 2+ 4H2O��2�֣�
��135cV2/ V1��2�֣�
���������������1�����ݻ��ϼ��������ͽ��ͼ��������ƽ�ɵû�ѧ����ʽ��2KClO3 + H2C2O4  K2 CO3 + CO2��+ 2ClO2��+ H2O
K2 CO3 + CO2��+ 2ClO2��+ H2O
��2���÷�Ӧ�¶�Ϊ60�棬ˮԡ���ȣ�����Ҫ�¶ȼƣ�B����ȴClO2��Ϊ ��ˮԡװ�á�
��3����ΪNaClO2������Һ�����¶ȵ���38��ʱ����������NaClO2��3H2O�����¶ȸ���38��ʱ����������NaClO2������Ӧ�������ᾧ���ٳ��ȹ��ˡ�
��4����ClO2���������ԣ��ɰ�I?����ΪI2����ƽ�ɵ����ӷ���ʽ��2ClO2 + 10I�� �� 8H+ = 2Cl����5I 2+ 4H2O
��ClO2��Һ��⻯�ط�Ӧ�����ӷ���ʽ8H+ + 2ClO2 + 10I������H2O + 5I2 + 2Cl����I2��2S2O32����2I����S4O62��������ClO2��5S2O32����ԭClO2��Һ��Ũ��Ϊ��cV2/5V1����10��67.5=135cV2/ V1��
���㣺���⿼�黯ѧ����ʽ����ƽ������ʵ���������������������ݷ���ʽ�ļ��㡣


 ��У��������ĩ��̾�ϵ�д�
��У��������ĩ��̾�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�Ʊ���ˮ�Ҵ���ʵ��ԭ��:�����Ҵ�95.6%��ˮ4.4%���һ�ֻ����,��ѹ��,����һ���ķе㣨78.17 �棩,����е����ˮ�ģ�100 �棩Ҳ���ڴ��Ҵ��ķе㣨78.3 �棩,����ֻ��������ƾ�,�����Ҵ��������ֻ�ܴﵽ95.6%,�к���4.4%��ˮ�����Ҫ�õ������ϸߵ��Ҵ�,��ʵ�����г��õļ�㷽���Ǽ�����ʯ�һ�������������һ�����Լ��ȵķ�Ӧ������,��Һ�����ʹ����ת�����ɵ�����ͨ�������������ֻ�����ԭ�����IJ�������ͼ�ף�,����֮��������,�ɵ�99.5%���Ҵ�,ͨ����Ϊ��ˮ�Ҵ������Ҫ�õ����ȸ��ߵľ����Ҵ������Ҵ�99.95%��,��Ҫ�ý����ƽ��д���,���Գ�ȥ�������ˮ�֡�
ʵ�鲽��:ȡ�����250 mLԲ����ƿһ��,����95%�Ҵ�100 mL��С����ʯ��30 g,��ҡ������Ƥ������,���ù�ҹ������ƿ��װ�ϻ���������,�ܿڽ�һ֧�Ȼ��Ƹ���ܣ�װ�ü�ͼ�ף�,��ˮԡ�ϼ��Ȼ���1��2Сʱ,����,ȡ��������,��װ������װ�ã���ͼ�ң�,�ٷ�����ˮԡ������,�����������5 mL���Һ�������,Ȼ���ú�ɵ�����ƿ��Ϊ������,���ܽ�һ֧�Ȼ��Ƹ����,ʹ���������ͨ,������Һ�γ���Ϊֹ,��ȡ�����Ҵ������,��������ʡ���ش�:
��1��Ϊʲô���ջ�����ʱ��Ҫװ���Ȼ��Ƹ����?
����
��2��Ϊ�˷�ֹ����ϵͳ�ⲿʪ������,���������Ӱ��������϶�����,�������Ƿ����,Ϊʲô? ����
��3����ˮ�Ȼ��Ƴ�������ˮ��,������ˮ�Ȼ��ƴ���������Ͷ��ƾ���,���ù�ҹ,Ȼ����ˡ��ô˷������б�ʵ����ȡ�Ҵ��Ƿ����,Ϊʲô?
����
��4��һ���ø���������л���ʱ,������ǰӦ�ȹ���,�����������Ʊ���ˮ�Ҵ�ʱ,Ϊʲô�ɲ�����,����ƿ��һ������?
����
��5��Ϊʲô���������5 mL���Һ�����ռ�,���Ĵ����Ƿ��Ѵ�99.5%?
����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�����к�̼�����Ԫ�ء���ѧ��ȤС���ij������Ʒ������̽��������Ҫ��ش��������⡣
��.������̼����Ԫ�صĶ��Լ���
����ͼװ�ý���ʵ��(�г���������ȥ,��ӿ���ĸ����)����ʵ��̼����Ԫ�صļ��顣
(1)����X��������������;װ�âۢ����Լ���ͬ,װ�â���ʢ�ŵ��Լ����� ������
(2)д�����з�Ӧ�Ļ�ѧ����ʽ����������������������������������������
(3)�������װ�âۢܢ�,�ܷ�ȷ��������Ʒ��̼Ԫ�صĴ���?��������,������������������������������������������������
��.������̼�������������IJⶨ
(4)��ͬѧ��Ϊ,����װ�ÿ��Դ��Բⶨ��Ʒ��̼�ĺ�������ȡ��Ʒw1 g����ʵ��,��ַ�Ӧ��,���װ�â������ɵij���Ϊw2 g,����Ʒ��̼����������Ϊ������(�ú�w1��w2��ʽ�ӱ�ʾ)��
(5)��ͬѧ��Ϊ,��һ������Ʒ��ַ�Ӧ��,��װ�â��м�������Ȼ�����Һ,���ݳ����������Լ�����Ʒ�������������,�˷�����õĽ��������(�ƫ��ƫС��);��Ҫ�����Ԫ�غ����IJⶨ����,�ڲ��ı�ʵ��ԭ����ǰ����,���Բ�ȡ��һ�ִ�ʩ������������������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��̼���ƣ�2Na2CO3��3H2O2����һ�ּ�ϴ�ӡ�Ư�ס�ɱ����һ�����ϵƯ�����þ������Na2CO3��H2O2��˫�����ʡ�����ͼ-2װ���Ʊ���̼���ƣ�����ˮԡ�г�ַ�Ӧ��ͼ-1���̿ɻ�ù�̼���Ʋ�Ʒ��

��1����ѹ��Һ©����֧�ܵ������� ��
��2���Ʊ���̼���ƵĹؼ��� ��
��3��������ƹ�̼���Ƶ�ˮ�к��������ӣ�����������ϴ�Ӽ���ȥ��������������ȫʧȥɱ�����á��Է������е�ԭ��д������һ�ּ��ɣ��÷���ʽ��ʾ����________________________________��
��4��ij��ѧѧϰС��Ϊ�˶���̽�������Ӷ���������Ư���IJ���Ӱ�죬ȡ��Ư��100mL������25g FeCl3���壬����������ɫ��ζ���壬������ƿ�ռ����塣��ѡ�������Լ���ʵ����Ʒ�������ɷֵ�̽�����̣�0��1mol/LNaOH��Һ��8��0mol/LNaOH��Һ������ʯ��ˮ��0��01mol/LKMnO4��Һ��BaCl2ϡ��Һ��Ʒ����Һ������ˮ��ľ�����ƾ��ơ����ϴ��ƿ��
��������裺�Ը�����ɷ�����������衣
����1��������O2�� ����2��������______________�� ����3��������CO2��
����Ʒ��������ʵ�鷽��֤����ļ��裬���±������ʵ�鲽�衢Ԥ����������ۣ�
| ʵ�鲽�� | Ԥ����������� |
| ����������ͨ��ʢ��_______��________��ϴ��ƿ�У�________________________�� | ��________________________ ��________________________ ��________________________ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����(�Ҷ���)������ԭ���ͳ����������ڽ������⡢֯��Ư��ϡ��������һ���Ʊ�����(��2���ᾧˮ)�Ĺ����������ң�
�ش��������⣺
��1��CO��NaOH��һ�������ºϳɼ����ơ������Ƽ�������Ļ�ѧ��Ӧ����ʽ�ֱ�Ϊ�� �� ��
��2�����Ʊ������������ι��˲��������˲����ٵ���Һ�� �������� �����˲����ڵ���Һ��_ �� �������� ��
��3�����չ����Тۺܵ͢�Ŀ���� ��
��4�����˽�������������ֱ���������ữ�Ʊ����ᡣ�÷�����ȱ���Dz�Ʒ���������к��е�������Ҫ�� ��
��5���ᾧˮ�ϲ����Ʒ�Ĵ����ø�����ط��ⶨ��
���������Ʒ0.250 g����ˮ����0.0500 mol��L-1������KMnO4��Һ�ζ�����dz�ۺ�ɫ�����ʣ�����KMnO4��Һ15.00 mL����Ӧ�����ӷ���ʽΪ ��
��ʽ����ó�Ʒ�Ĵ��� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij�о���ѧϰС����̽���ں�Mg2+��Al3+ �Ļ����Һ�еμ�NaOH��Һʱ������������������Ĺ��̡�
��ʵ�顿��0.1 mol?L��1 MgSO4��0.05 mol?L��1Al2(SO4)3�Ļ����Һ�еμ�0.5 mol?L-1NaOH��Һ�����������Ӵ���������Ӧ���������������Һ��pH��NaOH��Һ�ļ���仯�����ͼ��ʾ��
��1��Ϊ��ȷ���Ƽ���NaOH��Һ��������ɽ�NaOH��Һ���� �����������ƣ��еμӡ�
��2��ͼ��������pH���ӻ����ĽΣ���һ�Σ�a��ǰ����Ӧ��ʵ�������� ��
��3���Եڶ��Σ�b��c֮�䣩��ҺpH�仯�����ı��ʣ�С��ͬѧ���������Ʋ⣬�벹���Ʋ�2��3��
�Ʋ�1������Mg(OH)2����������OH����
�Ʋ�2�� ��
�Ʋ�3�� ��
���Ʋ�1����ʵ���������a��֮ǰ��Ӧ�����ӷ���ʽΪ ����ݴ�����Mg(OH)2��Al(OH)3����������ˮ��Һ���ܽ��ԵIJ��� ��
��4�������e�����Һ�нϴ������ڵĺ�����Ԫ�ص����Ӳ����ʵ����飨�ɲ���������
| �ϴ������ڵĺ�����Ԫ�ص����� | ���鷽�� |
| | |
| | |
| | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��̼����(2Na2CO4��3H2O)��һ������Ч����Ư��ɱ������������������������Ⱦ���ص㣬������Ӧ����ϴ�ӡ�ӡȾ����֯����ֽ��ҽҩ�����������С����д���Ʊ���̼���ƿɽ��������ɱ����������������£�
�Իش��������⣺
��1����̼��������ˮ��������Һ�� (����ԡ��������ԡ������ԡ�)���������ӷ���ʽ��ʾ��______________________��
��2����������Ҫ�IJ��������� (��д��������)��
��3����ҵ�����г���������NaCl��ijУ��ѧ����С�������ͼ��ʾװ�ã��ⶨ��ҵ������Na2CO3�ĺ�����
��Ҫ���鹤ҵ���������ʵĴ��ڣ����ѡ�������Լ��е�__________(ѡ�����)��
a. ����������Һ b. ϡ���� c. ���軯����Һ d. ��������Һ
�ڼ���װ��B�����Եķ����ǣ�����������©���������������н����ɼк�©��ע��һ������ˮ��ʹ©���ڵ�ˮ�����ƿ�ڵ�ˮ�棬ֹͣ��ˮ�����۲쵽 ˵��װ�ò�©����
��װ��A�������� ��װ��C�е��Լ�Ϊ ��
��ijͬѧ��Ϊ��Dװ�ú�Ӧ������Eװ��(װ���ʵ��Լ�)������Ϊ�Ƿ��Ҫ? (ѡ���Ҫ������Ҫ��)���жϵ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ʽ̼��ͭ��Cu2(OH)2CO3����һ����;�㷺�Ļ���ԭ�ϣ�ʵ�����Է�ͭмΪԭ����ȡ��ʽ̼��ͭ�IJ������£�
����һ����ͭм������ͭ
��ͼ���ý�ͷ�ι���ȡŨHNO3�����ӵ���ƿ�ڵķ�ͭм��(��ͭм����)����ַ�Ӧ����ˣ��õ�����ͭ��Һ��
���������ʽ̼��ͭ���Ʊ�
����Թ��м���̼������Һ������ͭ��Һ��ˮԡ������70�����ң���0��4 mol��L��NaOH��Һ����pH��8��5�������ã����ˣ�����ˮϴ�ӣ���ɣ��õ���ʽ̼��ͭ��Ʒ��
���������գ�
��1��д��Ũ������ͭ��Ӧ�����ӷ���ʽ�� ��
��2����ͼװ����NaOH��Һ��������_ ����Ӧ�������ƿ�ڵ���Һ�У����˺���NaOH�⣬����___ _(��д��ѧʽ)��
��3��������У�ˮԡ��������������__ _��_ (���ȡ��г�������ʯ��������)��
��4�����������Һ�п��ܺ���CO32-��д������CO32-�ķ�����
��5��Ӱ���Ʒ��������Ҫ������__ __��
��6����ʵ��õ�2��42 g��Ʒ(ֻ��CuO����)��ȡ����Ʒ�������ֽ���ȫ�õ�1��80 g���壬����Ʒ�м�ʽ̼��ͭ������������___ _��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ʵ�����Ʊ������飨C2H5Br����װ�úͲ�����ͼ������֪������ķе�38.4�棩
�ټ��װ�õ������ԣ���װ��ͼ��ʾ��U�ܺʹ��ձ��м����ˮ��
����Բ����ƿ�м���10mL95%�Ҵ���28mLŨ���ᣬȻ�������ϸ��13g�廯�ƺͼ������Ƭ��
��С����ȣ�ʹ���ַ�Ӧ��
�ش��������⣺
��1����ʵ����ȡ������Ļ�ѧ����ʽΪ�����ɵ���ΪNaHSO4����______________________��
��2����Ӧʱ���¶ȹ��ߣ��ɿ����к���ɫ��������������廯ѧʽΪ__________��
��3��Ϊ�˸��õĿ��Ʒ�Ӧ�¶ȣ�����ͼʾ��С����ȣ����õļ��ȷ�ʽ��__________��
��4��U���ڿɹ۲쵽��������_____________________________��
��5����Ӧ������U�ι��д��Ƶ�C2H5Br���ػ�ɫ��Ϊ�˳�ȥ�ֲ�Ʒ�е����ʣ���ѡ�������Լ��е�_________________������ţ�
a���� b��H2O c��Na2SO3��Һ d��CCl4
�������Ҫ����������______________�����������ƣ���
��6�����м���ʵ�鲽�裬�����ڼ�������������Ԫ�أ�����ȷ�IJ���˳���ǣ�ȡ���������飬Ȼ��__________________������ţ���
�ټ��ȣ��ڼ���AgNO3��Һ���ۼ���ϡHNO3�ữ���ܼ���NaOH��Һ������ȴ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com