

| | �������↑ʼ����ʱ��pH | �������������ȫʱ��pH |
| Fe3+ | 1.9 | 3.2 |
| Fe2+ | 7.0 | 9.0 |
| Mg2+ | 9.5 | 11.0 |
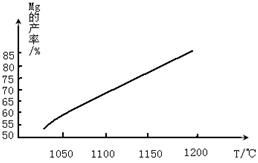
| A��KMnO4 | B��Cl2 | C��MgO | D��CaO |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
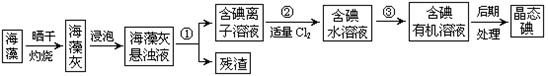
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
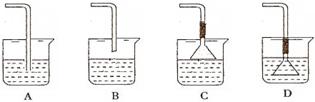
| A���ױ����ƾ� | B�����Ȼ�̼���� | C�����͡����� | D�����͡����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
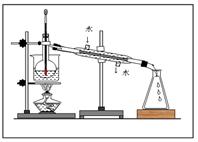
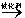 R-Cl(Һ)+HCl��������ȡ����Ʒ��������뼺��Ϊ��ʵ�����������ģ��װ��ͼ�ش��������⣺
R-Cl(Һ)+HCl��������ȡ����Ʒ��������뼺��Ϊ��ʵ�����������ģ��װ��ͼ�ش��������⣺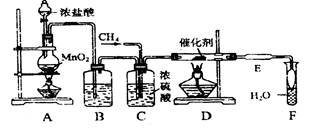
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
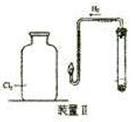
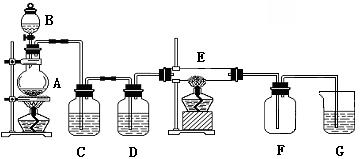
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
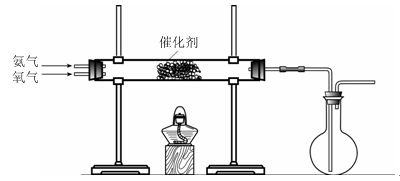
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�����ε��ܽ��һ����������ÿһ���ζ�С |
| B���Ͻ���۵�һ����������ÿһ�ֽ������� |
| C����NaOH��Һ�����η�Ӧ�ò���Al��OH��3���������һ���ð�ˮ�����η�Ӧ�Ʊ� Al��OH��3 |
| D����ȡ��ѡ��ı�֮һ�Dz����뱻��ȡ�����ʷ�Ӧ�������ܶ�Ҫ��ԭ�ܼ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

A������������������Ҵ������ñ���Na2CO3��Һϴ�Ӻ��Һ |
B�����飨��ϩ������Ũ��ˮϴ�� |
C���Ҵ������ᣩ����NaOH��Һ�кͺ��Һ |
D���Ҵ���ˮ��������ʯ����ˮ������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
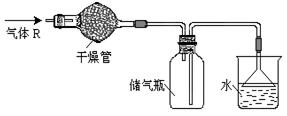
| A��HCl | B��Cl2 | C��CO | D��NH3 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com