
ͼʾ����ͼ���dz��õĿ�ѧ�о�������
��1����ѧ��ͨ��X�����Ʋ���мȺ�����λ���ֺ����������ṹʾ��ͼ�ɼ�ʾ���£�������λ����������������߱�ʾ��
��д����̬Cuԭ�ӵĺ�������Ų�ʽ____________________��
��д������������ˮ��ͭ���ӵĽṹ��ʽ�����뽫��λ����ʾ������________��
��2����ͼ���о�����Ԫ���⻯��ķе�仯���ɵ�ͼ������c���Ա������________��Ԫ���⻯��ķе�仯���ɡ���λͬѧ��ij����Ԫ���⻯��ķе�ı仯���ƻ������������ߡ�������a������b������Ϊ��ȷ����________���a����b������������______________________________________________________________��
��3����������Ԫ�ص���̬�����Ի�̬ԭ��ʧȥ�����һ������ת��Ϊ��̬��̬������������������������һ�����ܣ���ΪE������ͼ��ʾ��
��ͬ�����ڣ�����ԭ������������Eֵ�仯����������________��
�ڸ���ͼ���ṩ����Ϣ�����ƶ�E��________E�������������������������ͬ����
�۸��ݵ�һ�����ܵĺ����Ԫ�������ɣ����ƶ�Eþ________E����
 �ǻ�С��ϰϵ�д�
�ǻ�С��ϰϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Ԫ��R��Q��M��T��Ԫ�����ڱ��е����λ�����±�����֪Rԭ�����������������������֮��Ϊ2��1��
��1���˵ĺ�Һ��̨��T�ļ����ӣ������ӽṹʾ��ͼΪ_________________��Ԫ��M��Ԫ�����ڱ��е�λ����___________________��
��2��R����������������Ļ�ѧ��������__________��(ѡ����ӡ����ۡ�)��
��3������ʱ��Q������������Ӧˮ�����Ũ��Һ�뵥��R��Ӧ�Ļ�ѧ����ʽΪ__________(�þ���Ļ�ѧʽ��ʾ)��
��4����һ�������¼ס��ҡ���������ת������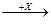 ��
��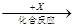 ���������м��ǵ��ʣ��ҡ���Ϊ�����x�Ǿ��������Ե���ɫ���嵥�ʣ���Ļ�ѧ��ɲ�������________(ѡ����ţ���ͬ)��
���������м��ǵ��ʣ��ҡ���Ϊ�����x�Ǿ��������Ե���ɫ���嵥�ʣ���Ļ�ѧ��ɲ�������________(ѡ����ţ���ͬ)��
��R ��Q2 ��M ��T2
��5����ҵ�ϣ������á�RO��MO2��Ӧ���ɹ�̬M���ʺ�RO2���Ӷ���������������Դ�������Ⱦ��
��֪��2RO(g)+O2(g)��2RO2(g) ��H��-akJ��mol-l
M(s)+O2(g)��MO2(g) ��H��-bkJ��mol-l
��Ӧ2RO(g)+MO2(g)��2RO2(g)+M(s) ��H��___________��
��6��Ԫ��T�ĺ�����HTO����Ư���ԡ���20mL 0.0lmol��L-l��HTO��Һ����μ���һ��Ũ�ȵ��ռ���Һ����û����Һ���¶ȱ仯����ͼ��ʾ���ݴ��жϣ����ռ���Һ��Ũ��Ϊ______________mol��L-l�������й�˵����ȷ����_______________��
��HTO�ĵ��볣����b�㣾a��
����ˮ�������c(OH��)��b�㣼c��
�۴�a�㵽b�㣬�����Һ�п��ܴ��ڣ�c(TO��)= c(Na+)
�ܴ�b�㵽c�㣬�����Һ��һֱ���ڣ�c(Na��)��c(TO��)��c(OH��)��c(H��)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ��Ԫ�����ڱ��н�ȡ��һ��Ƭ��,����X��Y��Z��W�����ڶ�����Ԫ�ء���ش���������:
(1)��X����Ϊ��������Ҫ�ɷ�֮һ,��Wԭ�ӽṹʾ��ͼΪ����������
(2)��Y��W��Z������������Ӧ��ˮ��������ΪһԪ�ᡢ��Ԫ�ᡢ��Ԫ��,������������εĺ������������,�����ֵĵ��������,�����������ӵķ�������������������������
(3)��Na��Y��Z�ĵ��ʷֱ�Ӧ��������Ħ��������ͬ�����ֻ�����,����Na��Y�γɵĻ�����ĵ���ʽΪ��������������,������ѧ��������Ϊ��
(4)YԪ�������ڱ��п��ܴ��ڵ���������������������(д�����п���������)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ֶ����ڷǽ���Ԫ��A��B��C��Dԭ��������������A��ԭ�Ӱ뾶��С��Ԫ��,Bԭ�ӵ������������Ǵ���������������,D�ڵؿ��к��������ش�:
(1)BԪ�ص�ԭ�ӽṹʾ��ͼ����
(2)������CA3�ĵ���ʽ����������������,BD2���ӵĽṹʽΪ��������������
(3)��Ϊ������������Ԫ�صij�������ʽ��,����Ļ�ѧʽΪ��������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
п��Zinc���ǵ��ġ��������Ľ�������������������ͭ�����ִ���ҵ�ж��ڵ�������в���ĥ��Ĺ��ס�
��.ʪ����п
ij��ұ�����̿�����ͼ���Ա�ʾ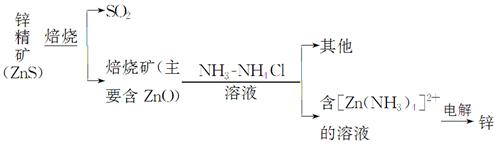
��1��ZnS���շ�Ӧ�Ļ�ѧ����ʽΪ_____________________________________��
��2������������пһ���ĵ缫��ӦʽΪ_______________________________��
��3������п��������һ�����������������������п���������Ʒ�Ӧ�����ӷ���ʽΪ_________________________________________________________��
����֪����п�ᡱ�Ļ�ѧʽ��д��H2[Zn��OH��4]��
��.��
��п�������ĵ���������洦�ɼ�����п�̸ɵ�ء�п�̼��Ե�ء�п�յ�صȡ�
��4��п�̼��Ե�أ��Զ�������Ϊ������п��Ϊ����������������ҺΪ���Һ������������ŵ��������ص㣬����õ��㷺Ӧ�á�����ܷ�ӦʽΪZn��2MnO2��2H2O=2MnO��OH����Zn��OH��2��
���Ե���У�����п��Ƭ״�ı����״���ŵ���_______________________��������ӦʽΪ________________________________________________________________________��
��5������п�յ�أ���ͼ����﮵����ȣ�п��������صĴ�������������������ɱ���﮵�ص�һ�룬������ȫû�й��ȱ�ը�İ�ȫ�������õ�ص��ܷ�ӦΪ2Zn��O2=2ZnO���������ҺΪKOH��Һ�����ĵ缫��ӦʽΪ__________________________�����Ըõ��Ϊ��Դ���ö��Ե缫�����������Һ��Ϊ��֤������10.8 g��������������Ҫ________L����������ɱ�״��������õ�ء�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Ԫ���γɵij����ǽ������嵥��A�볣����������B���ڼ��������·�Ӧ���ɻ�����C��C��ˮ��Ӧ���ɰ�ɫ����D������E��D��������ǿ�ᣬҲ������ǿ�E������������ȼ�ղ����̼�������G��G�ڴ������ܵ���������γɡ�E����������������Һϴ��õ���ɫ��ҺF����ҺF�ڿ����г��ڷ��÷�����Ӧ��������֮һΪH��H��������ƵĽṹ�ͻ�ѧ�������ƣ�����Һ�Ի�ɫ����ش��������⣺
��1����ɵ���A��Ԫ�������ڱ��е�λ���� ��
��2����ɵ���B��Ԫ�ص�ԭ�ӽṹʾ��ͼΪ ��
��3��G�������������������·�Ӧ����������ɱ�����������ȣ��÷�Ӧ��������Ϊ ��
��4����ҺF�ڿ����г��ڷ�������H�Ļ�ѧ����ʽΪ ��
��5��H����Һ��ϡ���ᷴӦ����������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)�������߷ֱ��ʾԪ�ص�ij��������˵�����Ĺ�ϵ(ZΪ�˵������YΪԪ�ص��й�����)����������Ԫ���йص�������������߱��������Ӧ�Ŀո��У�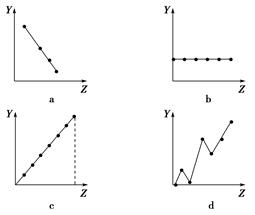
�ٵڢ�A��Ԫ�صļ۵�����________��
�ڵ�������Ԫ�ص��������________��
��F����Na����Mg2����Al3�������Ӱ뾶________��
(2)Ԫ��X��Y��Z��M��N��Ϊ����������Ԫ�أ���ԭ����������������֪Yԭ������������������������֮��Ϊ3��4��MԪ��ԭ�ӵ���������������Ӳ���֮��Ϊ4��3��N����Z����X�����ӵİ뾶��С��������XN������Ϊ���塣�ݴ˻ش�
��N������������Ӧ��ˮ����Ļ�ѧʽΪ_______________��
�ڻ�����A��B��Ϊ����������Ԫ���е���������Ԫ����ɵ�ǿ����ʣ�����������ˮ��Һ���������ͬ�����Ԫ�ص�ԭ����Ŀ֮�Ⱦ�Ϊ1��1��1��A��Һ��ˮ�ĵ���̶ȱ�B��Һ��ˮ�ĵ���̶�С������A�еĻ�ѧ������Ϊ________��B�Ļ�ѧʽΪ________��
�۹�ҵ����ȡ����M�Ļ�ѧ����ʽΪ____________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��Y��Z��W��R��Ԫ�����ڱ�ǰ�������еij���Ԫ��,�������Ϣ���±�:
| Ԫ�� | �����Ϣ |
| X | ��ɵ����ʵĻ���Ԫ��,����������ϼ���������ϼ۵Ĵ�����Ϊ2 |
| Y | �ؿ��к�����ߵ�Ԫ�� |
| Z | ����������Ϊ23,������Ϊ11�ĺ��� |
| W | �����д���ʹ����Ͻ���Ʒ,��ҵ�Ͽ��õ������������ķ����Ʊ��䵥�� |
| R | �ж��ֻ��ϼ�,���ɫ���������ڿ����л�Ѹ�ٱ�ɻ���ɫ,����ɺ��ɫ |
 W�Ͻ�(Z17W12)��һ��DZ�ڵ��������,��Z��W������һ���������������ɡ��úϽ���һ����������ȫ����ķ�Ӧ����ʽΪ:Z17W12+17H2
W�Ͻ�(Z17W12)��һ��DZ�ڵ��������,��Z��W������һ���������������ɡ��úϽ���һ����������ȫ����ķ�Ӧ����ʽΪ:Z17W12+17H2 17ZH2+12W,�õ��Ļ����Q(17ZH2+12W)��6.0 mol��L-1 HCl��Һ������ȫ�ͷų�H2��1 mol Z17W12��ȫ�����õ��Ļ����Q������������ȫ��Ӧ,�ͷų�H2�����ʵ���Ϊ��������
17ZH2+12W,�õ��Ļ����Q(17ZH2+12W)��6.0 mol��L-1 HCl��Һ������ȫ�ͷų�H2��1 mol Z17W12��ȫ�����õ��Ļ����Q������������ȫ��Ӧ,�ͷų�H2�����ʵ���Ϊ�������� �鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Ԫ�����ڱ��У�ͬһ����Ԫ�ػ�ѧ�������ơ�ĿǰҲ������ЩԪ�صĻ�ѧ���ʺ��������ڱ������Ϸ������·�����һ����Ԫ���������ƣ����Ϊ�Խ��߹��ݴ���ش�
��1����ڿ�����ȼ�գ�������______________�⣬Ҳ��������________________��
��2������֪��ӦBe2C��4H2O��2Be(OH)2��CH4������Al4C3������ǿ����Һ��Ӧ�����ӷ���ʽΪ_____________________________________________________________��
��3����ѧ��֤ʵ��BeCl2�ǹ��ۻ�����������һ����ʵ��֤������ʵ�鷽���ǣ�____________________________________________________________________________��
�õ���ʽ��ʾBeCl2���γɹ��̣�______________________________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com