
 H2(g) + CO2(g)��ƽ�ⳣ�����¶ȵı仯���±���
H2(g) + CO2(g)��ƽ�ⳣ�����¶ȵı仯���±���| �¶�/�� | 400 | 500 | 850 |
| ƽ�ⳣ�� | 9.94 | 9 | 1 |
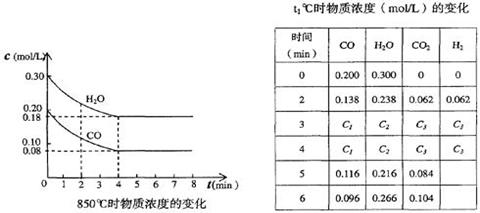
| ʱ �䣨min�� | CO | H2O | CO2 | H2 |
| 0 | 0.200 | 0.300 | 0 | 0 |
| 2 | 0.138 | 0.238 | 0.062 | 0.062 |
| 3 | C1 | C2 | C3 | C3 |
| 4 | C1 | C2 | C3 | C3 |
| 5 | 0.116 | 0.216 | 0.084 | |
| 6 | 0.096 | 0.266 | 0.104 | |
 1/2N2O4(g) ��H = ��26.35 kJ��mol��1
1/2N2O4(g) ��H = ��26.35 kJ��mol��1 H2(g) + CO2(g)
H2(g) + CO2(g)
 H2(g) + CO2(g)
H2(g) + CO2(g)

 �����Ծ���Ԫ���Ծ�ϵ�д�
�����Ծ���Ԫ���Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ӦCaCO3(s)=CaO(s)+CO2(g)�ڳ����²��ܷ�������÷�Ӧ�ġ�H��0 |
B��0.1 mol��L��1CH3COOH��Һ��ˮϡ�ͺ���Һ��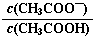 ��ֵ��С ��ֵ��С |
| C��Ǧ�����ڷŵ�����У������������ӣ������������� |
| D����¯�г�����CaSO4���ñ���Na2CO3��Һ���ݣ��ٽ���������ϡ�����ܽ��ȥ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 H2��I2
H2��I2
 2HI(g)��ƽ�ⳣ��K��__________________
2HI(g)��ƽ�ⳣ��K��__________________�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����֪���³�ѹ��4Fe(s)+3O2(g)=2Fe2O3(s)���Է���Ӧ, ��÷�Ӧ�����ȷ�Ӧ |
| B��Ǧ���طŵ�ʱ�ĸ����ͳ��ʱ������������������Ӧ |
C��һ��������2molPCl3��2molCl2������ӦPCl3(g)+Cl2(g)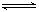 PCl5(g) ��H= -93kJ?mol��1 ,��ƽ��ʱ����139.5kJ,��PCl3��ת����Ϊ75% PCl5(g) ��H= -93kJ?mol��1 ,��ƽ��ʱ����139.5kJ,��PCl3��ת����Ϊ75% |
| D����pH�ֱ�Ϊ2��3�Ĵ����к͵�����NaOH,���Ĵ��������ֱ�ΪV1��V2����V1>10V2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
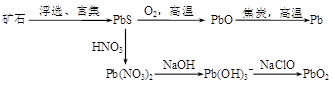
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����H��0�ķ�Ӧ��������һ�������Է����� |
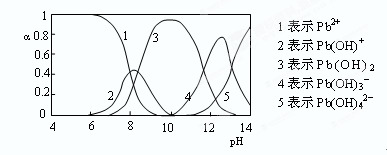
B����0.1mol/LNaOH��Һ�ֱ�ζ���ͬ���ʵ���Ũ�Ⱥ���ͬ���������ʹ��ᣬ����ʵ�߱�ʾ���ǵζ����������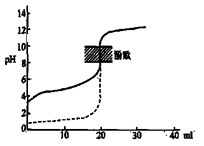 |
| C�������£���0.1mol/L��ˮ�У���������NH4Cl���壬��Һ��pH��С |
D�������ܱ������н��еķ�Ӧ3A(g) 2B(g)��C(s)���������������������£��ٳ���һ������A���壬A��ת���ʲ��� 2B(g)��C(s)���������������������£��ٳ���һ������A���壬A��ת���ʲ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��H3BO3��������H2CO3������ |
| B����Ag2CrO4����Һ�м�NaCl��Һ����AgCl�������ɣ�Ksp (Ag2CrO4) ��Ksp(AgCl) |
| C��I2��KI��Һ�к���ˮ�е��ܽ�� |
| D����ͬ�¶��£�lO ml O.l mol��lһ�Ĵ�����100 ml O.01 mol��Lһ�Ĵ����е�H+���ʵ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 N2(g
N2(g )��O2(g)
)��O2(g) 2NO(g) ��H��a kJ��mol-1��ƽ�ⳣ��K���±���
2NO(g) ��H��a kJ��mol-1��ƽ�ⳣ��K���±���| �¶�/�� | 1538 | 1760 | 2404 |
| ƽ�ⳣ��K | 0��86��10��4 | 2��6��10��4 | 64��10��4 |
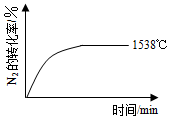
 ��ʽ���£�
��ʽ���£� (g)��3O2(g) ��H����1530kJ��mol-1
(g)��3O2(g) ��H����1530kJ��mol-1 �о����ø����ӵ����Ե�SCY�մɣ��ܴ���H+��ʵ�ֵ��Ĺ̶��������ĵ�ⷨ�ϳɣ��������˵�����������ת���ʡ��ܷ�ӦʽΪ��N2��3H2
�о����ø����ӵ����Ե�SCY�մɣ��ܴ���H+��ʵ�ֵ��Ĺ̶��������ĵ�ⷨ�ϳɣ��������˵�����������ת���ʡ��ܷ�ӦʽΪ��N2��3H2 2NH3�����ڵ�ⷨ�ϳɰ��Ĺ����У�Ӧ����2���ϵ�ͨ�� ������������������� ������ ��������������һ�缫ͨ��N2���õ缫��ӦʽΪ ��
2NH3�����ڵ�ⷨ�ϳɰ��Ĺ����У�Ӧ����2���ϵ�ͨ�� ������������������� ������ ��������������һ�缫ͨ��N2���õ缫��ӦʽΪ ��
 ��4���ϳɰ��ķ�ӦN2(g)+3H2(g)
��4���ϳɰ��ķ�ӦN2(g)+3H2(g)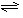 2NH3(g) ��H = ��92��4 kJ��mol��1��һ�������µ��ܱ������У��÷�Ӧ�ﵽƽ�⣬Ҫ���N2��ת���ʣ����Բ�ȡ�Ĵ�ʩ��__________������ĸ���ţ���
2NH3(g) ��H = ��92��4 kJ��mol��1��һ�������µ��ܱ������У��÷�Ӧ�ﵽƽ�⣬Ҫ���N2��ת���ʣ����Բ�ȡ�Ĵ�ʩ��__________������ĸ���ţ��� ��5����25���£���a mol��L-1�İ�ˮ��0��01 mol��L-1������������ϣ���Ӧ����Һ��c(NH4+)=c(Cl-)������Һ��_____________�ԣ���ᡱ������С��������ƶ�a 0��01������ڡ����ڻ�С�ڣ���
��5����25���£���a mol��L-1�İ�ˮ��0��01 mol��L-1������������ϣ���Ӧ����Һ��c(NH4+)=c(Cl-)������Һ��_____________�ԣ���ᡱ������С��������ƶ�a 0��01������ڡ����ڻ�С�ڣ����鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com