
��ˮAlCl3�������л��ϳɵĴ�����ʳƷ���ɼ��ȡ���ҵ������������Ҫ�ɷ���A12O3��Fe2O3����ʯ�ͽ�����Ҫ�ɷ���C������ͼ��ʾ���̽���һϵ�з�Ӧ���Ʊ���ˮAlCl3��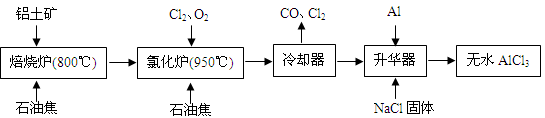
��1���Ȼ����ڼ�������������������̬�Ȼ����Ļ�ѧʽΪAl2Cl6��ÿ��Ԫ�ص�ԭ���������ﵽ8�����ȶ��ṹ����AlCl3�ǣ� ���壬��ṹʽΪ�� ��
��2���Ȼ�¯��Al2O3��Cl2��C��Ӧ�Ļ�ѧ����ʽ�� ��
��3����ȴ���ų���β���к��д���CO������Cl2������Na2SO3��Һ��ȥCl2���˷�Ӧ�����ӷ���ʽΪ�� ��
��4������������Ҫ����AlCl3��FeCl3�����������Al���������ǣ� ��
��5��AlCl3��Ʒ��FeԪ�غ���ֱ��Ӱ����Ʒ�ʣ�Ϊ�ⶨ��Ʒ��FeԪ�صĺ������ֳ�ȡ16.25g��ˮAlCl3��Ʒ�����ڹ�����NaOH��Һ�����˳�����������ᆳϴ�ӡ����ա���ȴ�����ز�����������Ϊ0.32g�����Ʒ��FeԪ�صĺ���Ϊ�� ��
��1������ 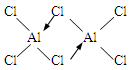
��2��A12O3+3C12+3C 2 A1C13+3CO
2 A1C13+3CO
��3��SO32��+C12+H2O��SO42��+2C1��+2H+
��4����ȥFeCl3
��5��1.38%����1.4%��
���������������1�������Ȼ�������������������ȷ����Ϊ���Ӿ��壻��������ԭ���������ﵽ8�����ȶ��ṹ��ȷ���Ȼ����ṹʽ����2����Ӧ����Al2O3��Cl2��C����������ȷ���������Ȼ�����CO����3��Na2SO3Ϊ��ԭ����Cl2����Ϊ+6�۵������Σ��ȱ���ԭΪ-1�ۣ���4�����Ļ����Ա���ǿ����������FeCl3�����û�������ͬʱ�������������Ȼ�����
��5���������������������n(Fe)=2��n(Fe2O3)= 2��0.32��160=0.004mol
W(Fe)= 0.004��56��16.25=0.014
���㣺���鹤ҵ�����з�Ӧԭ�������ʽṹ��ʵ��������й����⡣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij�о�С��������¹���·���ᴿ��ҵ̼���ơ���֪��ҵ̼����(����ԼΪ98%)�к���Ca2����Mg2����Fe3����Cl������SO42�������ʡ�
��.̼���Ƶı�����Һ�ڲ�ͬ�¶�����������������ͼ��ʾ��
��.�й����ʵ��ܶȻ����£�
| ���� | CaCO3 | MgCO3 | Ca(OH)2 | Mg(OH)2 | Fe(OH)3 |
| Ksp | 4.96��10��9 | 6.82��10��6 | 4.68��10��6 | 5.61��10��12 | 2.64��10��39 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Һ�žú���������������AgN3��������ը��ֱ���ŷŻ���Ⱦ���������������Դ���˷ѡ�ij�о�С������˴�������Ӧ��ķ�Һ�У���������������Һ�����費��������������������������ʵ�����̣�
����֪��[Ag��NH3��2]������Һ�д���ƽ�⣺[Ag��NH3��2]��??Ag����2NH3��
��1��д�������ڢٲ���Һ��ϡHNO3��Ӧ�����ӷ���ʽ ��
��2�������ڢڲ����������Ҫ������Ŀ���� ��
�������̿��ܲ����Ĵ�����Ⱦ���� ��
��3���ҷ��������յõ����۵�����ƫ���ų�δϴ�Ӹɾ������أ����ܵ�ԭ���� ��
��4��ʵ��������������Һ�IJ��������� ��
��5����֪�ҷ����ڢ۲���Ӧ��H2S��������������յõ�����21.6 g��������������ʧ�������ϸò���Ҫ�������� g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������������ҡ������Դ�ı��⡣�й�Ҫʵʩ����ǿ��ս�ԣ�ʵ���ɺ���������ǿ�����������롣�����Ѿ���Ϊ�����ҹ����÷�չ����Ҫ���棬��ˮ���ۺϿ����������Ǻ��õ�һ���֣���ˮ�п���ȡ���ֻ���ԭ�ϣ������ǹ�ҵ�϶Ժ�ˮ�ļ����ۺ����õ�ʾ��ͼ������������ͼ��ʾ��
��1��д���١��ڷ�Ӧ�����ӷ���ʽ��
��______________________����______________________��
��2����ҵ�����õ�ⱥ��ʳ��ˮ������������������ȡ���ᣬΪ��������ɫ��ѧ���ʹ������ַ�Ӧ����ȡ��������������ȼ�յİ취���ɱ�������ȼ�ղ���ȫ��Ⱦ��������д��������������ȼ�յ�ʵ������______________________��
��3�������к���Ca������Mg������SO4���������ʣ����ƺ�ɵõ�����NaC1��Һ���������г����Լ���A������ B������������Һ C��̼������Һ������ʱ������������Լ�����ȷ˳����______________��������ţ�
��4������þ�ڿ�����ȼ��ʱ��������MgO�⣬��������Mg3N2���ɡ��ѵ����ʵ����Ľ���þ�ֱ���ڣ�A����������O2���У�B��������̼�����У�C�������С���ȫȼ�պõ��Ĺ������ʵ������ɴ�С��˳����______________��������ţ�
��5������ⱥ��NaCl��Һ���ɵ�����ͨ������������Һ�п��Եõ�NaClO��ij��ѧ��ȤС��̽��NaClO������CO(NH2)2�ķ�Ӧ���ͨ��ʵ�鷢�ֲ����ij�����⣬������ﶼ���ܲ������ѭ�������ʣ���÷�Ӧ�Ļ�ѧ����ʽΪ____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ʯ���顢ʯ�ҵ�(CaCN2)��������(��H2S)��Ӧ�����ܾ���β�������ܻ��Ӧ�ù㷺��CS(NH2)2(����)���䲿�ֹ����������£�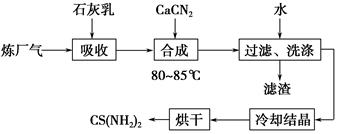
(1)�����£�H2S�������з�Ӧ��2H2S(g)??2H2(g)��S2(g)����ƽ�ⳣ������ʽΪK��________________��
(2)��ʯ��������H2S��ȡCa(HS)2��Ҫ�ڵ����½��У���ԭ����_____________________________________________________��
���˵õ��������������ã���������Ҫ�ɷ���________(�ѧʽ)��
(3)�ϳ������賤ʱ����裬���ڽϸ��¶�(80��85 ��)�½��У���Ŀ����_______________________________________��
Ca(HS)2��CaCN2��ˮ��Һ�кϳ�����Ļ�ѧ����ʽΪ________________________________��
(4)������X�����廥Ϊͬ���칹�壬X����FeCl3��Һ�У���Һ�Ժ�ɫ��X�Ļ�ѧʽΪ________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ˮ����Ҫ���ӵĺ������£�
| �ɷ� | ����/(mg/L) | �ɷ� | ����/(mg/L) |
| Cl- | 18980 | Ca2+ | 400 |
| Na+ | 10560 | HCO3- | 142 |
| SO42- | 2560 | Mg2+ | 1272 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ƿ��ɫ��Һ�ı�ǩ���ˣ�ֻ֪�����Ƿֱ������ᡢBaCl2��Һ��Na2CO3��Һ���ס�����λͬѧ����һ���Լ���һ���Լ���ȡ���˳ɹ������õ�һ��ָʾ����_________�����õ�һ������Һ��__________����ͬѧû���κ��Լ�Ҳ����ɹ��ˣ����ļ�������ǣ�ȡ�����Թ��У����Ϊa��b��c��Ȼ��������Һ������ϣ�����a����b�������ݲ�����a����c���а�ɫ������������a��b��c�ֱ���________��________��__________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
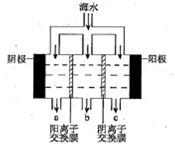
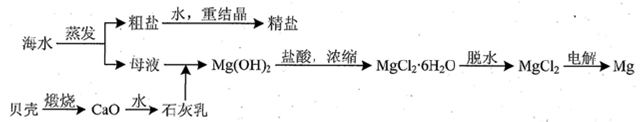
��ˮ���зḻ����Դ�����ֶ����ĺ������ֲ������зḻ�Ŀ�ء�ʯ�͡���Ȼ���ȣ����⣬��ˮ�л����д����ĵ���ʣ����ǵ������Cl����Br���������ӣ���SO42����Na����Mg2����Ca2���ȣ�������Ҫ��Դ��
��1��д������١��ڡ��ܷ����ᴿ�ķ�����
�� �� �� �� �� ��
��2������ڷ����ᴿ��������Ҫѡ����Ҫ�������������� ��
��3������ȥ�����ᴿ��Ĵ����е�MgCl2��CaCl2��Na2SO4��Ӧ��ô�ʳ��ˮ�����μ���NaOH��Һ�� ��Һ�� ��Һ��Ȼ����ˣ�Ϊ�����ܳ�ȥ���ʣ�ÿ�μ�����Լ�Ӧ ����������Һ�еμ� �������ݲ������پ������ᾧ�õ�ʳ�ξ��塣
��4�����鵭ˮ���Ƿ���Cl-�IJ��������� ��
��5��д�������Լ�a������Ӧ�����ӷ���ʽ ��
��6����ҵ���õ�ⱥ��ʳ��ˮ�ķ��������������������ռ��д������������ʯ������ȡƯ�۵Ļ�ѧ����ʽ ________________________________________________ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������棨CeO2����һ����Ҫ��ϡ�������ƽ�������ʾ�����������в��������ķϲ�����ĩ����SiO2��Fe2O3��CeO2�Լ���������������ϡ������ʣ���ij�������Դ˷�ĩΪԭ�ϻ����棬���ʵ���������£�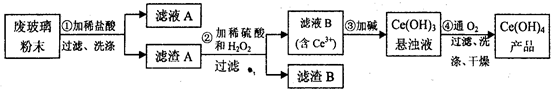
��1��ϴ�ӵ�Ŀ����Ҫ��Ϊ�˳�ȥCl����___________�������ӷ��ţ������������Aϴ���ķ�����_____________________��
��2���ڢڲ���Ӧ�����ӷ���ʽ��____________________________��
��3��ȡ���������еõ���Ce��OH��4��Ʒ0��536 g���������ܽ����0��1000mol· L��1FeSO4����Һ�ζ��յ�ʱ���汻��ԭΪCe3����������25��00mL����Һ���ò�Ʒ��Ce��OH��4����������Ϊ_____________��
��������þ��ҽҩ����������ҵӦ�ù㷺������þ��ԭ�Ƚ��Ʊ��ߴ�����þ��һ���µ�̽��������þ����Ҫ�ɷ�ΪMgCO3��������FeCO3��Ϊԭ���Ʊ��ߴ�����þ��ʵ���������£�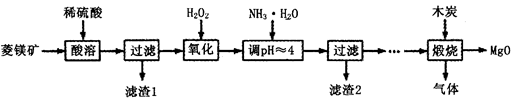
��1������H2O2����ʱ��������Ӧ�Ļ�ѧ����ʽΪ________________��
��2������2�ijɷ���______________���ѧʽ����
��3�����չ��̴������·�Ӧ��2MgSO4��C=2MgO��2SO2����CO2��
MgSO4��C=MgO��SO2����CO�� MgSO4��3C=MgO��S����3CO��
������ͼװ�ö����ղ�����������зֲ����ջ��ռ���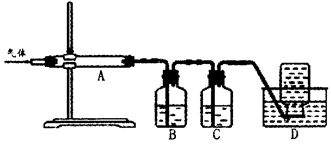
��D���ռ������������______________���ѧʽ����
��B��ʢ�ŵ���Һ������______________������ĸ����
| A��NaOH��Һ | B��Ca��OH��2��Һ | C��ϡ���� | D��KMnO4��Һ |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com