
����Ŀ��ij�Ȼ�����Ʒ����FeCl2���ʡ���Ҫ�ⶨ������Ԫ�ص�����������ʵ�鰴���²�����У�
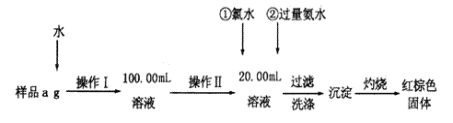
(1)����I���õ��IJ����������ձ�����Ͳ��100mL������ƿ�⣬��������_________(����������)������ƿʹ��ǰ������еIJ�����_________(�����)
A.���� B.��© C.��ʪ
(2)д��������ˮ������Ӧ�����ӷ���ʽ_______________________________________���÷�Ӧ��������ˮ�����������Լ��е�______________����(�����)��
A��H2O2 B����ˮ C��NaClO
(3)��������Ѿ�ϴ�Ӹɾ��IJ�����������__________________________��
(4)��������ΪW1g�����Ⱥ����������ɫ����������ΪW2g������Ʒ����Ԫ�ص�����������____________________(�г�ԭʼ��ʽ�����軯��)
���𰸡�����������ͷ�ι� B 2Fe 2++Cl2=2Fe3++2Cl- AC ȡ���һ��ϴ��Һ���Թܣ��μ����������ữ���ټ�����������Һ�����ް�ɫ�������ɣ���֤���Ѿ�ϴ�Ӹɾ� 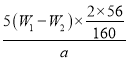 ��100%
��100%
��������
��ʵ��Ŀ���Dzⶨ����������������ȡ�ķ�����ʹ��Ʒ�ܽ⡢��Ӧ������������������Ȼ��ͨ������������������������������
(1)��ͼ��֪������I�ǽ���ˮ�ܽ�����Һϡ�ͳ�100.00mL��Һ����Ҫ�ձ��ܽ⣬�ò��������裬�����ȣ�������Ҫ��ͷ�ιܣ�����ƿʹ��ǰ��Ҫ��©��
(2)����ˮ������+2������Ϊ+3�ۣ�������ˮ�����ù������⡢�������ƴ���������������ˮ�еⵥ�ʲ��������������ӣ�
(3)��Һ�д����Ȼ�泥������������������Һ�������һ��ϴ��Һ���Ƿ���������ӣ����жϳ����Ƿ�ϴ����
(4)���ȷֽ����õ�������Fe2O3��������Ϊ(W2-W1)g������ʹ��20.00mL��Һ����ʵ�飬���100.00mL��Һ���Եõ�Fe2O3����Ϊ5(W2-W1)g�����ݻ�ѧʽ������Ԫ�ص����������������������Ķ������ԭ�Ȼ�����Ʒ����Ԫ�ص�����������
��1����ͼ��֪������I�ǽ���ˮ�ܽ�����Һϡ�ͳ�100.00mL��Һ��������������Ͳ��100mL������ƿ�⣬���������ܽ���ձ��Ͳ�����������ƿ��������̶���1~2cmʱ��Ҫ�ý�ͷ�ιܶ��ݣ�����ȱ�ٵ������Dz������ͽ�ͷ�ιܣ�����ƿʹ��ǰ������еIJ����Ǽ���ܷ�©ˮ����˺���ѡ����B��
(2)����ˮ����ʹ+2��Fe2+����Ϊ��Ϊ+3��Fe3+���÷�Ӧ�����ӷ���ʽΪ��2Fe 2++Cl2=2Fe3++2Cl-����������ˮ�����ù������⡢�������ƴ�������������������ˮ�еⵥ�ʲ��������������ӣ���˺���ѡ����AC��
(3)Fe(OH)3�����Ǵ�NH4Cl��Һ�й��˳����ģ�Fe��OH��3�������������Ȼ�泥������������������Һ�������һ��ϴ��Һ���Ƿ���������ӣ����鷽���ǣ�ȡ���һ��ϴ��Һ���Թ��У��μ�ϡ�����ữ���ټ���������������Һ�����ް�ɫ�������ɣ���֤����ϴ�Ӹɾ���
(4)����Ԫ�������غ㣬������ɫ�����е���������Ʒ������Fe2O3������ΪW2-W1g�����ڲμӷ�Ӧ����Һֻȡ������Һ��![]() �������Ԫ�ص�����Ϊ5(W2-W1)g��
�������Ԫ�ص�����Ϊ5(W2-W1)g��![]() ������Ʒ����Ԫ�ص�����������
������Ʒ����Ԫ�ص����������� ��100%��
��100%��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ҩ��F���п���������Ѫ�ǡ���Ѫѹ�ȶ���������ԣ���ϳ�·�����£�

��֪��M�Ľṹ��ʽΪ��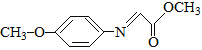 ��
��
��ش��������⣺
��1��A�Ļ�ѧ������_______________��B�Ļ�ѧʽ��________________��
��2��C�й����ŵ�������__________________________��
��3��д��F�Ľṹ��ʽ__________________________��
��4����֪A��һ�������������ɿɽ���ľ�������д���÷�Ӧ��ѧ����ʽ��______________________________��
��5����������������M��ͬ���칹����_______��(���������칹)��
�� �ܹ�����������Ӧ��
�� ��������(�CNO2)��������ֱ�����ڱ����ϡ�
�� ���б����ұ�����ֻ������ȡ������
���к˴Ź�������Ϊ������ҷ����֮��Ϊ6��2��2��1�Ľṹ��ʽΪ________��д��һ�ּ��ɣ���
��6��д������ȩΪԭ���Ʊ��߷��ӻ�����۱�ϩ��ĺϳ�·�ߣ����Լ���ѡ����______________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������P(s)��Cl2(g)������Ӧ����PCl3(g)��PCl5(g)����Ӧ���̺�������ϵ��ͼ��ʾ(ͼ�е���H��ʾ����1mol���������)��
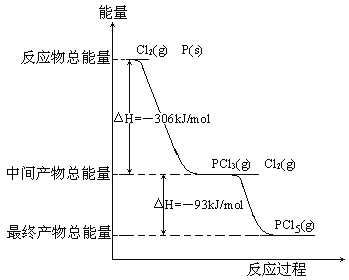
������ͼ�ش��������⣺
(1)P��Cl2��Ӧ����PCl3���Ȼ�ѧ����ʽ_________________________________��
(2)PCl5�ֽ��PCl3��Cl2���Ȼ�ѧ����ʽ_________________________________�������ֽⷴӦ��һ�����淴Ӧ���¶�T1ʱ�����ܱ������м���0.80molPCl5����Ӧ�ﵽƽ��ʱPCl5��ʣ0.60mol����ֽ�����1����_________������Ӧ�¶���T1���ߵ�T2��ƽ��ʱPCl5�ķֽ���Ϊ��2����2_______��1(��������������С��������������)��
(3)��ҵ���Ʊ�PCl5ͨ�����������У��Ƚ�P��Cl2��Ӧ�����м����PCl3��Ȼ���£��ٺ�Cl2��Ӧ����PCl5��ԭ����________________________________________��
(4)P��Cl2��������Ӧ����1molPCl5����H3��_________��P��Cl2һ����Ӧ����1molPCl5����H4______��H3(��������������С��������������)��
(5)PCl5������ˮ��ַ�Ӧ���������������ᣬ�仯ѧ����ʽ��______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������(SO2Cl2)����������(SOCl2)�������Ȼ�������������ҽҩ��ũҩ��Ⱦ�Ϲ�ҵ���л��ϳɹ�ҵ���������Ȼ����������������(Li/SOCl2)��ء�
�й����ʵIJ����������±���
���� | �۵�/�� | �е�/�� | �������� |
SO2Cl2 | -54.1 | 69.1 | ����ˮ�⣬������������ ���ֽ⣺SO2Cl2 |
H2SO4 | 10.4 | 338 | ������ˮ�����ѷֽ� |
ʵ�����ø���������Ķ�������������ϳ������ȣ���Ӧ���Ȼ�ѧ����ʽΪ��SO2(g)+Cl2(g) ![]() SO2Cl2(l) ��H= - 97.3 kJ��mol-1����Ӧװ����ͼ��ʾ���г�������ʡ�ԣ�����ش��й����⣺
SO2Cl2(l) ��H= - 97.3 kJ��mol-1����Ӧװ����ͼ��ʾ���г�������ʡ�ԣ�����ش��й����⣺

��1������A������Ϊ___________��
��2������B��������_____________________��
��3��װ�ñ���ʢ�ŵ��Լ�Ϊ____________����ʵ�����������������ն��������ȷ�Ӧ�����ӷ���ʽΪ___________________��
��4��Ϊ��߱�ʵ���������ȵIJ��ʣ���ʵ���������Ҫע���������_________(�����)
����ͨ����ˮ����ͨ���� �ڿ����������ʣ��������˿�
����������ƿ���̣����ʵ����� �ܼ���������ƿ
��5������������Ҳ�����Ȼ���(ClSO3H)�ֽ��ã��÷�Ӧ�Ļ�ѧ����ʽΪ��2ClSO3H��H2SO4+SO2Cl2���˷����õ��IJ�Ʒ�л�������ᡣ
�ٴӷֽ�����з���������ȵ�ʵ���������Ϊ__________________��
�����ʵ�鷽�������Ȼ���ֽ���ȡ�����Ȳ�Ʒ�л������ᣬ���з����������ǣ�_____(����ĸ��
A.ȡ��Ʒ����ˮ���μ���ɫʯ����Һ��죺��ȡ������Һ������BaCl2��Һ������ɫ������˵������H2SO4��
B.ȡ��Ʒ�ڸ��������¼�������ȫ��Ӧ����ȴ��ֱ�Ӽ�BaCl2��Һ���а�ɫ�������ٵμ���ɫʯ����Һ��죬˵������H2SO4��
��6��Li��SOCl2��ؿ����������������õ�صĵ缫���Ϸֱ�Ϊ﮺�̼���������Һ��LiAlCl4��SOCl2����ص��ܷ�Ӧ�ɱ�ʾΪ��4Li��2SOCl2===4LiCl��S��SO2��
��д���õ�����������ĵ缫��Ӧʽ_______________________________
����װ�õ�ر�������ˮ�������������½��У�ԭ����__________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������ƾ���(Na2S2O3��5H2O)�ֳƺ���������ɫ��״��������ˮ���������Ҵ�����������֯��Ư������ȼ������������еĻ�ԭ����
��. Na2S2O3��5H2O���Ʊ�
Na2S2O3��5H2O���Ʊ������ж��֣������������Ʒ��ǹ�ҵ��ʵ�����е���Ҫ������Na2SO3 + S + 5H2O ![]() Na2S2O3��5H2O
Na2S2O3��5H2O
�Ʊ��������£�
�ٳ�ȡ12.6g Na2SO3��100mL�ձ��У���50 mLȥ����ˮ�����ܽ⡣
����ȡ4.0 g�����200mL�ձ��У���6 mL�Ҵ���ֽ�����Ƚ�����ʪ���ټ���Na2SO3��Һ����ʯ��С�������У����Ͻ�������ۼ���ȫ����Ӧ��
��ֹͣ���ȣ�����Һ����ȴ���2 g����̿���������2����(��ɫ)��
�ܳ��ȹ��ˣ�����Һ���������У� ______________��____________________��
�ݹ��ˡ�ϴ�ӣ�����ֽ���ɺ��أ�������ʡ�
��1�������������Ҵ���ʪ��Ŀ����____________________________��
��2������ܳ��ȹ��˵�ԭ��_____________________���ո�Ӧ��ȡ�IJ�����_________________��____________________��
��3�������ϴ�ӹ����У�Ϊ��ֹ�в��ֲ�Ʒ��ʧ��Ӧѡ�õ��Լ�Ϊ___________��
��4����Һ�г�Na2S2O3��δ��Ӧ��ȫ��Na2SO3�⣬����ܴ��ڵ���������________________�����ɸ����ʵ�ԭ�������____________________________��
��.��Ʒ���ȵIJⶨ
ȷ��ȡ1.00 g��Ʒ(��������ƾ����Ħ������Ϊ248 g/mol)������������ˮ�ܽ⣬�Ե�����ָʾ������0.1000 mol/L�����Һ�ζ����յ㣬����21.00 mL����Ӧ�����ӷ���ʽΪ��2S2O32-+I2=S4O62-+2I-��
��5���������ò�Ʒ�Ĵ���Ϊ___________(������λ��Ч����)�������ݵĺ������Ϳ�����__________(������ʵ�������������)��
��.��Ʒ��Ӧ��
��6��Na2S2O3���������ȼ�������Һ���ױ�Cl2����ΪSO42-���÷�Ӧ�����ӷ���ʽΪ ____________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������������ˮ���������м���ǿ���������������ʵ��ǣ� ��
A.Cl2B.NH3C.NO2D.SO2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���������ӷ���ʽ����д��ȷ����
A. ʳ�׳�ȥƿ���ڵ�ˮ����CaCO3+2H+===Ca2++CO2��+H2O
B. Ư����Һ��ͨ������SO2��Ca2++2ClO-+SO2+H2O===CaSO3��+2HClO
C. Al����NaOH��Һ��2Al+2OH-+2H2O===2 AlO+3H2��
D. �ö��Ե缫�������ͭ��Һ��2Cu2++4OH-![]() 2Cu+O2��+2H2O
2Cu+O2��+2H2O
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���Ը���������(��Ҫ�ɷ�ΪAl2O3��Fe2O3��SiO2������FeS2)Ϊԭ�ϣ�����Fe3O4�IJ��ֹ����������£�

����˵���������
A. ��NaOH��Һ���ձ��չ��̲�����SO2�����ڱ�����������Դ������
B. �Ӹ����������п��Ƶ�Al��Fe�Ļ�����
C. ���ˡ��õ�����Һ��ͨ�����CO2����Ԫ�ش��ڵ���ʽ��AlOת��ΪAl3+
D. FeS2��Fe2O3��Ϻ������������±��յ�Ŀ��������Fe3O4��SO2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ԭ�Ӻ˴Ź�����(NMR)���о��л�������ṹ�������ֶ�֮һ�������о�������ķ����У�ÿһ�ṹ�еĵ�����ԭ�ӣ���NMR���ж�������Ӧ�ķ�(�ź�)�����з��ǿ����ṹ�е�Hԭ���������ȡ����磬��ȩ�ĽṹʽΪ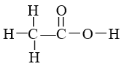 ����NMR�����������źţ���ǿ��֮��Ϊ3��1��
����NMR�����������źţ���ǿ��֮��Ϊ3��1��
��1������ʽΪC3H6O2�Ķ�Ԫ���������NMR���й۲���ԭ�Ӹ����ķ��������������һ��������ǿ�ȱ�Ϊ3��3���ڶ���������ǿ�ȱ�Ϊ3��2��1���ɴ˿��ƶϸû�������ɿ�����(д�ṹ��ʽ)_____________________��
��2���ڲ�õ�CH3CH2CH2CI������ģ�NMR���Ͽɹ۲쵽���ַ壬���ⶨ������CH3CH=CHClʱ��ȴ�õ���ԭ�Ӹ�����6���źŷ塣����ԭ�ӿռ����з�ʽ��ͬ����д��CH3CH=CHCl���ӵĿռ��칹____��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com