

���� ��1����ͼ������������֪XΪԲ����ƿ��
��2���ζ�ǰ���ȼ��ζ����Ƿ�©ˮ���ٽ�����ϴ��Ȼ���ñ�Һ��ϴ����ע���Һ���ž��ζ���������ݲ�����Һ�棬��������¼���ζ�ǰ����ɣ�
��3�����������������Ƽ���Bװ����ʣ���HCl���μӷ�Ӧ��HCl���շֽ����ɵ�NH3��������Ӧ��NH3+HCl=NH4Cl��������������NH3�����ʵ�����
��4��ȡD��Һ���Թ��У���������BaCl2��Һ���ټ������ᣬ��ɫ������ȫ�ܽ������ɴ̼�����ζ�����壬˵��D����Һ����SO32-����SO42-��
��5��װ��D����Һ����SO32-��˵���ֽ�����SO2��װ��A��D֮��ĵ���������������ɫ���壬��ɫ����Ӧ�Ƕ�����������ˮ�γɵ��Σ�װ��B����Һ���յ������ǰ�����
��6���ɣ�5���з�����֪����NH4��2SO4��400��ֽ�ʱ����NH3��SO2��H2O���ɣ�SԪ�ػ��ϼ۽��ͣ����ݵ���ת���غ㣬ֻ��ΪNԪ�ػ��ϼ����ߣ�����������������˵������N2����ƽ��д����ʽ��
��� �⣺��1��������X�Ľṹ��֪��XΪԲ����ƿ���ʴ�Ϊ��Բ����ƿ��
��2���ζ�ǰ���ȼ��ζ����Ƿ�©ˮ���ٽ�����ϴ��Ȼ���ñ�Һ��ϴ����ע���Һ���ž��ζ���������ݲ�����Һ�棬��������¼���ζ�ǰ����ɣ�����ȷ��˳��Ϊ��dbaec��
�ʴ�Ϊ��dbaec��
��3���ζ�ʣ�����ᣬ�յ�ʱ����NaOHΪ0.025L��0.2mol/L=0.005mol����ʣ��HClΪ0.005mol����μӷ�Ӧ��HClΪ0.07L��0.5mol/L-0.005mol=0.03mol���μӷ�Ӧ��HCl���շֽ����ɵ�NH3��������Ӧ��NH3+HCl=NH4Cl��������NH3�����ʵ���Ϊ0.03mol��
�ʴ�Ϊ��0.03mol��
��4�����װ��D����Һ����SO32-����SO42-��ʵ������������ǣ�ȡ����D��Һ���Թ��У���������BaCl2��Һ���а�ɫ�������ɣ��ټ������ᣬ��ɫ������ȫ�ܽ⣬���ɴ̼�����ζ�����壬˵��D����Һ����SO32-����SO42-��
�ʴ�Ϊ��ȡ����D��Һ���Թ��У���������BaCl2��Һ���а�ɫ�������ɣ��ټ������ᣬ��ɫ������ȫ�ܽ⣬���ɴ̼�����ζ�����壬˵��D����Һ����SO32-����SO42-��
��5��װ��D����Һ����SO32-��˵���ֽ�����SO2��װ��A��D֮��ĵ���������������ɫ���壬��ɫ����Ӧ�Ƕ�����������ˮ�γɵ��Σ�װ��B����Һ���յ������ǰ�����
�ʴ�Ϊ��NH3��
��6���ɣ�5���з�����֪����NH4��2SO4��400��ֽ�ʱ����NH3��SO2��H2O���ɣ�SԪ�ػ��ϼ۽��ͣ����ݵ���ת���غ㣬ֻ��ΪNԪ�ػ��ϼ����ߣ�����������������˵������N2���ֽⷴӦ����ʽΪ3��NH4��2SO4$\frac{\underline{\;400��\;}}{\;}$4NH3��+3SO2��+6H2O��+N2����
�ʴ�Ϊ��3��NH4��2SO4$\frac{\underline{\;400��\;}}{\;}$4NH3��+3SO2��+6H2O��+N2����
���� ���⿼������ʵ�鷽������ƣ�Ϊ��Ƶ���㣬���շ�Ӧԭ����ʵ�鲽�輰ʵ��װ�õ�����Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬��Ŀ�ѶȲ���


 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Na202������H20��Ӧ���ɱ����11.2L 02��ת�Ƶ��ӵ���ĿΪ2NA | |
| B�� | ��״���£�2.24L S03������������Ϊ4NA | |
| C�� | �ö��Ե缫���CuS04��Һ���������0.1mol Cu��0H��2��ʹ��Һ��ԭ�����·��ת�Ƶ��ӵ���ĿΪ0.2NA | |
| D�� | 0���101kp�������£�1.12L������ȫȼ�����ɵ�ˮ������Ϊ0.1NA |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
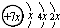 ��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
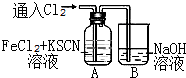 ��ѧ����Cl2��FeCl2��KSCN�����Һ�ķ�Ӧ����ʵ��̽������A��ͨ������������������A����Һ�ȳʺ�ɫ����Ϊ��ɫ��
��ѧ����Cl2��FeCl2��KSCN�����Һ�ķ�Ӧ����ʵ��̽������A��ͨ������������������A����Һ�ȳʺ�ɫ����Ϊ��ɫ��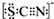 ����̼Ԫ�صĻ��ϼ���+4
����̼Ԫ�صĻ��ϼ���+4�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��ɫʳƷ�Dz����κλ�ѧ���ʵ�ʳƷ | |
| B�� | Na2O2��CO2��Ӧ��Na2O2����������CO2�ǻ�ԭ�� | |
| C�� | �����Ƶ�FeSO4��Һ�е���������NaOH��Һ������Ƭ�̣�������Ӧ���̵���ɫ�仯�ǣ�dz��ɫ��Һ����ɫ����������ɫ���������ɫ���� | |
| D�� | ��ȡ���������������ۣ��ֱ���������ϡ���������NaOH��Һ����ų������������ͬ��ͬѹ�£������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
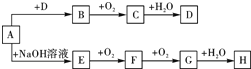 A��H��8�����ʴ�������ת����ϵ����Ӧ���������ֲ���δ���������֪��A�����Σ�B����ʹƷ����Һ��ɫ�����壬G�Ǻ���ɫ���壮��Ҫ��ش����⣺
A��H��8�����ʴ�������ת����ϵ����Ӧ���������ֲ���δ���������֪��A�����Σ�B����ʹƷ����Һ��ɫ�����壬G�Ǻ���ɫ���壮��Ҫ��ش����⣺�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 3.0g��ȩ�ͼ�������Ļ�����к��е�ԭ����Ϊ4NA | |
| B�� | �ȼҵ�У�������������״���µ�����22.4Lʱ����·��ת�Ƶĵ�����Ϊ2NA | |
| C�� | ��״���£�0.1mol���к��еĦҼ�����ĿΪ0.6NA | |
| D�� | ����200ml0.1mol/L��ϡ���ᣬ��Ҫ98%��Ũ����0.02NA |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Fe2O3+3CO�T2Fe+3CO2 | B�� | Ca��OH��2+CO2�TCaCO3��+H2O | ||
| C�� | CuO+H2�TCu+H2O | D�� | 2Na+Cl2�T2NaCl |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
| A�� | CH2=CH-CH2OH | B�� | HO-CH2-CH2-CH2-COOH | ||
| C�� | HNO3 | D�� | �Ҵ� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com