
����Ŀ�����⻯��(NaAlH4)���л��ϳɵ�һ����Ҫ��ԭ����һ����Ʊ������ǽ�AlC13�����л��ܼ����ٰ�������Һ�μӵ�NaH��ĩ�ϣ����Ƶ����⻯�ơ�ʵ��Ҫ���װ�����£��ش��������⣺
(1)��ȡ���⻯��Ҫ�ڷ�ˮ��Һ�н��У���Ҫԭ����_______(�û�ѧ����ʽ��ʾ)��������װ����ȡ�������⻯��(�������ﴦ��)��Ϊ��������__________��

(2)����ԭ��A1C13����ȡ��ij��ȤС�����������װ�ã�
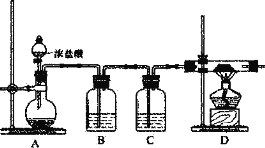
�����Ӻ�װ�ú�Ӧ���еĵ�һ��������_________��Ϊ��֤����Ĵ�����Ӧ��D��__________(������)�ټ��Ⱦƾ��ơ�
��Bװ����ʢװ����NaCl��Һ��ʵ�鿪ʼ��B�в�����������_________��ѡ��NaCl��Һ��������_____��
��Cװ�õ�������_________������Cװ�ã���Ԥ��ʵ���D���������A1C13����ܻ�����_________��(�ѧʽ)
������װ�ô��ڵ�����ȱ����_________��
���𰸡�NaAlH4+2H2O=NaAlO2+4H2��(��дΪNaH+H2O=NaOH+ H2��) D ���װ�õ������� ��������ɫ����ʱ ��ɫ�������� ���ܳ�ȥHCl�����ܽ���Cl2���ܽ�� ���� Al2O3 û��β������װ�ã�����ɴ�����Ⱦ
��������
(1)���⻯�ƺ��⻯����-1�۵�H������ˮ��Ӧ���ݴ˷��������ݷ�Ӧ���״̬����Ӧ����ѡ����ʵ�װ�ã�
(2)��Ũ������MnO2��ϼ�����ȡ��ȡCl2���Ƶõ�Cl2�к�������HCl��H2O����ͨ������ʳ��ˮ��ȥ�Ȼ������ʣ�Ȼ��ͨ��Ũ������и��Ȼ��ʹ�����������������ڼ���ʱ��Ӧ��ȡAlCl3���ݴ˷������
(1)���⻯�ƺ��⻯����-1�۵�H������ˮ��Ӧ����Ӧ����ʽΪ��NaAlH4+2H2O=NaAlO2+4H2��(��дΪNaH+H2O=NaOH+ H2��)��Ϊ��ֹ���ʣ���ȡ���⻯�Ʊ����ڷ�ˮ��Һ�н��У�AlC13���л��ܼ��γɵ���Һ��NaH�����������·�����ӦAlCl3+4NaH=NaAlH4+3NaCl��װ��A��B���ǹ������ʼ�����ȡ�����ʵ�״̬����Ӧ���������ϣ�C��D������״̬��������ȡNaAlH4������AlCl3�к��������ᾧˮ��ʪ��ˮ���ø��Ȼ�����ȡNaAlH4ʱ���ͻ�ͬʱ�����ܶȱȿ���С������������ֻ���������ſ����ķ����ռ���װ��C���õ��������ſ������������������������ռ���װ��D���ʣ��ʺ���ѡ����D��
(2)��������μӵĻ�ѧ��Ӧ��������װ�ú�Ӧ���еĵ�һ�������Ǽ��װ�õ������ԣ�Ϊ��֤����Ĵ�����Ӧ��D�г�������ɫ����ʱ�ټ��Ⱦƾ��ƣ��Ը���װ���п�������ֹAl��װ���ڿ����е�O2��Ӧ��
�ڱ���ʳ��ˮ�д����ܽ�ƽ�⣬������ˮ�д��ڻ�ѧƽ�⡣��HCl��������ˮ������ˮ��c(Cl-)����NaCl���ܽ�ƽ���Cl2��ˮ�����Ŀ��滯ѧ��Ӧ��ƽ����������ƶ����Ӷ��������������а�ɫ�����������ò���ͬʱ�ֽ������������ܽ�ȣ�
��Cװ��ʢ��Ũ���ᣬ�������Ǹ�������������Cװ�ã���ʪ��������Al�ڷ�Ӧʱ��������AlCl3��������ˮ��Ӧ�����ֱ�ΪAl(OH)3��HCl������ˮ�ֵ�������HCl�ӷ��������ΪAl(OH)3������Al(OH)3�����ֽⷴӦ����Al2O3�����Ԥ��ʵ���D���������A1C13����ܻ�����Al2O3��
(3)Cl2���ж����壬������������������������ɴ�����Ⱦ����˸�װ�ô��ڵ�����ȱ����û��β������װ�ã�����ɴ�����Ⱦ��


 Сѧ�̲�ȫ��ϵ�д�
Сѧ�̲�ȫ��ϵ�д� Сѧ��ѧ������ѿڶ���ϵ�д�
Сѧ��ѧ������ѿڶ���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����ԭ���ڻ�ѧ��ҵ���й㷺Ӧ�á�
��1���ȼ��ⱥ��ʳ��ˮ��ȡ�ռ�,��������ͼװ���Ʊ�NaOH�����缫�����Һ�ֱ�ΪNaOH��NaCl��Һ��
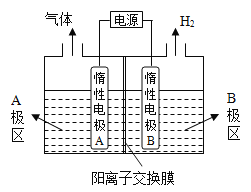
��B�������ҺΪ_____________��Һ���ѧʽ����A��Ҫ���ӵ�Դ��______________(���������)����
�������缫��ӦʽΪ_____________________________����������Na+��_____________�缫Ǩ�ƣ��A����B������
�۵���ܷ�Ӧ�����ӷ���ʽ��________________________________��
��2��������ͼװ�ã�����ģ�����ĵ绯ѧ������
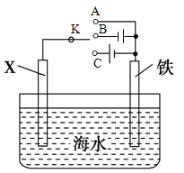
����XΪ̼�缫��Ϊ�������ĸ�ʴ������KӦ����_____________�����A������B����C������
����XΪп�缫������K����A�����õ绯ѧ��������Ϊ______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������ⷨ���Ѱ۲����ķ�Һ[���д���FeSO4��H2SO4������Fe2(SO4)3��TiOSO4]����������Ͳ�Ѫ�������������������������£�
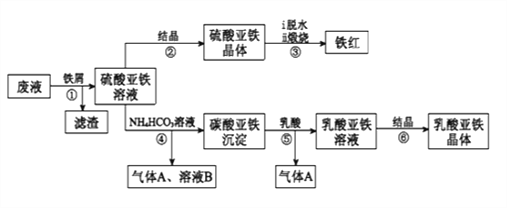
��֪��TiOSO4������ˮ����ˮ�п��Ե���ΪTiO2+��SO42-��TiOSO4ˮ���TiO2xH2O����Ϊ���淴Ӧ������ṹ��ʽΪCH3CH(OH)COOH��
��ش�
��1���������з�������������Һ�������IJ�����________________________��
��2��������м��Ŀ��һ�ǻ�ԭ����Fe2(SO4)3������ʹ����TiOSO4ת��ΪTiO2xH2O��������ƽ���ƶ���ԭ�����͵õ�������ԭ��___________________________��
��3�����������ڿ�������������������������÷�Ӧ���������ͻ�ԭ�������ʵ���֮��Ϊ_________________��
��4�������ӷ���ʽ���Ͳ������м������ܵõ�����������ԭ��_________________��
��5�������������ӷ���ʽ��_________________________________________��
��6��Ϊ�ⶨ�����������þ�����FeSO4��7H2O������������ȡ������Ʒa g������ϡ�������100.00 mL��Һ��ȡ��20.00 mL��Һ����KMnO4��Һ�ζ���������KMnO4����Ӧ����������0.1000 molL-1 KMnO4��Һ20.00 mL�����þ�����FeSO4��7H2O����������Ϊ______________����a��ʾ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����ӻ�����A�Ļ�ѧʽΪNH5����������ԭ�ӵ�����㶼������Ӧ��ϡ������ԭ�ӵ������ṹ����ش�
(1)��д��A�������Ӽ������ӵĻ�ѧʽ________��________
(2)�ʵ����ȣ�A�ͷֽ�Ϊ�������壬����һ�������ǻ�����÷�Ӧ�Ļ�ѧ����ʽΪ________��
(3)A����ˮ������һ�ֻ������һ�ֵ��ʣ���A��ˮ��Һ��________��(�ᣯ���)��ԭ��________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ˮ�к��д���Na+��C1-������Ca2+��Mg2+��SO42-���õ��������Ըú�ˮ��Ʒ���е�������������ͼ��ʾ������˵����ȷ����

A. bĤ�������ӽ���Ĥ
B. A���Ҳ������ݲ�����������������
C. ����������ɺ�A��B��C������pH��СΪpHA<pHB<pHC
D. B���Ҳ����������ʹʪ���KI������ֽ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���Ҵ�(C2H5OH)ȼ�ϵ��(DEFC)���кܶ��ŵ㣬���������ǵ��о���Ȥ���������������Ҵ�ȼ�ϵ�ء�

(1)�����Ҵ�ȼ�ϵ����������Ӧ���Ϊ_________________��(�ѧʽ)
(2)�������Ҵ�ȼ�ϵ������ѡ������̼���Ϊ���ʣ���ع���ʱ��CO32����缫___(����a������b��)�ƶ���
(3)�����Ҵ�ȼ�ϵ���У���������ı�״����2.24L O2�����·��ͨ���˵ĵ�����ĿΪ___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ijѧ����0.1000mol/L��NaOH����Һ�ζ�δ֪Ũ�ȵ����ᣬ������ɷ�Ϊ���¼�����
A��ȡ��NaOH��Һע���ʽ�ζ������̶���0����2cm��3cm��
B����ȡ25.00mL������������Һע��ྻ����ƿ��������2��3�η�̪��
C���ñ���Һ��ϴ�ζ���2��3�Σ�
D������Һ������0������0�����¿̶Ȳ����¶�����
E����ʢ�б���Һ�ļ�ʽ�ζ����ܶ��ã����ڵζ��ܼ���ʹ֮������Һ��
F������ƿ���ڵζ��ܵ����棬�ñ�NaOH��Һ�ζ����յ㲢�ǵζ���Һ��Ŀ̶ȡ�
�ݴ�ʵ������գ�
��1����ȷ���������˳����__��__��__��__��B��__�����������ĸ��д��
��2���ζ���ϴ��֮ǰ����___���ζ�������ʱ�������յζ��ܵĻ���������ҡ����ƿ���۾�ע��___���ζ��ﵽ�յ�ı�־��___��
��3������B�������֮ǰ�����ô�����Һ��ϴ��ƿ����Եζ������Ӱ����___������ƫ������ƫС����������������
��4�������Ĵεζ���ʼ�ͽ���ʱ���ζ����е�Һ����ͼ��ʾ��������NaOH��Һ�����Ϊ___mL��

��5��ijѧ�������Ĵ�ʵ���¼���ݼ����������Һ�����ʵ���Ũ�ȣ�c��HCl��=___��
�ζ����� | ���ᣨmL�� | 0.1000mol/LNaOH�������mL�� | ||
�ζ�ǰ | �ζ��� | ��Һ��� | ||
��һ�� | 25.00 | 0.05 | 26.17 | 26.12 |
�ڶ��� | 25.00 | 1.58 | 30.31 | 28.73 |
������ | 25.00 | 0.22 | 26.30 | 26.08 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������£�0.1mol/LijһԪ�ᣨHA����Һ��![]() =1��10-8������������ȷ���ǣ� ��
=1��10-8������������ȷ���ǣ� ��
A.��һԪ����Һ��pH=1
B.����Һ����ˮ�������=c(H+)=1��10-11mol/L
C.����Һ��ˮ�����ӻ�����Ϊ1��10-22
D.��pH=11������������ҺV1L��V2L0.1mol/L��һԪ�ᣨHA����Һ��ϣ��������Һ��pH=7����V1<V2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ijС����CoCl36H2O��NH4Cl��H2O2��Ũ��ˮΪԭ�ϣ��ڻ���̿���£��ϳ��˳Ȼ�ɫ����X��Ϊȷ������ɣ���������ʵ�顣

�ٰ��IJⶨ����ȷ��ȡw g X��������ˮ�ܽ⣬ע����ͼ��ʾ������ƿ�У�Ȼ����μ�������10%NaOH��Һ��ͨ��ˮ����������ƷҺ�еİ�ȫ����������V1 mLc1 molL-1���������Һ���ա�����������ȡ�½���ƿ����c2 molL-1NaOH����Һ�ζ���ʣ��HCl�����յ�ʱ����V2 mL NaOH��Һ��
���ȵIJⶨ��ȷ��ȡ��ƷX�������Һ����AgNO3����Һ�ζ���K2CrO4��ҺΪָʾ���������ֵ���ɫ����������ʧΪ�յ�(Ag2CrO4Ϊש��ɫ)��
�ش��������⣺
(1)װ���а�ȫ�ܵ�����ԭ����___��
(2)��NaOH����Һ�ζ���ʣ��HClʱ��Ӧʹ��______ʽ�ζ��ܣ���ʹ�õ�ָʾ��Ϊ_______��
(3)��Ʒ�а���������������ʽΪ_______��
(4)�ⶨ��ǰӦ�ö�װ�ý��������Լ��飬�������Բ��òⶨ�����_____(�ƫ�ߡ���ƫ�͡�)��
(5)�ⶨ�ȵĹ����У�ʹ����ɫ�ζ��ܵ�ԭ����_____���ζ��յ�ʱ������Һ��c(Ag+)=2.0��10-5 molL-1��c(CrO42-)Ϊ_____ molL-1��(��֪��K sp(Ag2CrO4)=1.12��10-12)
(6)���ⶨ����ƷX���ܰ��ȵ����ʵ���֮��Ϊ1��6��3���ܵĻ��ϼ�Ϊ_____���Ʊ�X�Ļ�ѧ����ʽΪ______��X���Ʊ��������¶Ȳ��ܹ��ߵ�ԭ����_____��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com